Ro 5-3335CAS# 30195-30-3 |
- BS-181
Catalog No.:BCC1439
CAS No.:1092443-52-1
- WHI-P180 hydrochloride
Catalog No.:BCC4243
CAS No.:153437-55-9
- SNS-032 (BMS-387032)
Catalog No.:BCC1152
CAS No.:345627-80-7
- Dinaciclib (SCH727965)
Catalog No.:BCC3765
CAS No.:779353-01-4
- RGB-286638
Catalog No.:BCC5519
CAS No.:784210-87-3
- AT7519 Hydrochloride
Catalog No.:BCC1376
CAS No.:902135-91-5
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 30195-30-3 | SDF | Download SDF |
| PubChem ID | 64983 | Appearance | Powder |
| Formula | C13H10ClN3O | M.Wt | 259.63 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in DMSO > 10 mM | ||
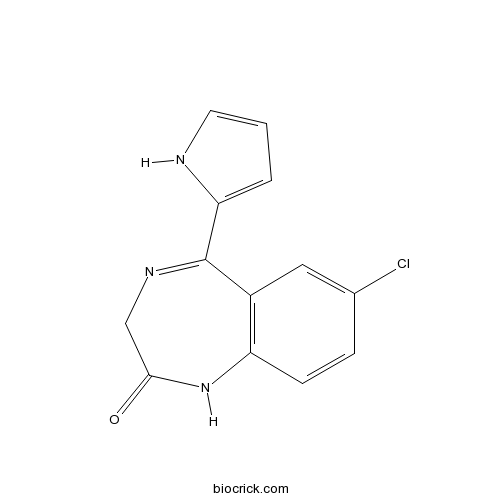
| Chemical Name | 7-chloro-5-(1H-pyrrol-2-yl)-1,3-dihydro-1,4-benzodiazepin-2-one | ||
| SMILES | C1C(=O)NC2=C(C=C(C=C2)Cl)C(=N1)C3=CC=CN3 | ||
| Standard InChIKey | XWNMORIHKRROGW-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C13H10ClN3O/c14-8-3-4-10-9(6-8)13(11-2-1-5-15-11)16-7-12(18)17-10/h1-6,15H,7H2,(H,17,18) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Core binding factor (CBF) inhibitor; preferentially kills human leukemia cell lines with CBF fusion proteins (IC50 = 1.1 μM). Represses RUNX1/CBFβ-dependent transactivation in reporter assays and inhibits transcriptional regulation by RUNX1 and CBFβ. Reduces leukemia burden in a mouse model. Attenuates RUNX1-dependent hematopoiesis in zebrafish embryos. Also a Tat antagonist; inhibits HIV-1 replication in vitro. |

Ro 5-3335 Dilution Calculator

Ro 5-3335 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.8516 mL | 19.2582 mL | 38.5164 mL | 77.0327 mL | 96.2909 mL |
| 5 mM | 0.7703 mL | 3.8516 mL | 7.7033 mL | 15.4065 mL | 19.2582 mL |
| 10 mM | 0.3852 mL | 1.9258 mL | 3.8516 mL | 7.7033 mL | 9.6291 mL |
| 50 mM | 0.077 mL | 0.3852 mL | 0.7703 mL | 1.5407 mL | 1.9258 mL |
| 100 mM | 0.0385 mL | 0.1926 mL | 0.3852 mL | 0.7703 mL | 0.9629 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Boc-Lys(Boc)-OSu
Catalog No.:BCC3415
CAS No.:30189-36-7
- D4476
Catalog No.:BCC1508
CAS No.:301836-43-1
- SB 431542
Catalog No.:BCC3658
CAS No.:301836-41-9
- Jasminin
Catalog No.:BCN7468
CAS No.:30164-93-3
- Compstatin control peptide
Catalog No.:BCC6067
CAS No.:301544-78-5
- Seneganolide
Catalog No.:BCN5209
CAS No.:301530-12-1
- H-Val-NH2.HCl
Catalog No.:BCC3144
CAS No.:3014-80-0
- P7C3
Catalog No.:BCC6524
CAS No.:301353-96-8
- CH 223191
Catalog No.:BCC3896
CAS No.:301326-22-7
- TCS 359
Catalog No.:BCC1183
CAS No.:301305-73-7
- Tianeptine sodium
Catalog No.:BCC2506
CAS No.:30123-17-2
- Malvidin-3-O-galactoside chloride
Catalog No.:BCN3030
CAS No.:30113-37-2
- Desoxyrhaponticin
Catalog No.:BCN2954
CAS No.:30197-14-9
- Hydroxyprogesterone acetate
Catalog No.:BCC8997
CAS No.:302-23-8
- Aconitine
Catalog No.:BCN1014
CAS No.:302-27-2
- DL-Alanine
Catalog No.:BCN8539
CAS No.:302-72-7
- Retinoic acid
Catalog No.:BCN2185
CAS No.:302-79-4
- Arenobufagin 3-hemisuberate
Catalog No.:BCN7837
CAS No.:30219-16-0
- Effusanin A
Catalog No.:BCN5210
CAS No.:30220-43-0
- Ingenol
Catalog No.:BCN2333
CAS No.:30220-46-3
- 1-Hydroxybaccatin I
Catalog No.:BCN5211
CAS No.:30244-37-2
- Potassium 7-hydroxynaphthalene-1-sulfonate
Catalog No.:BCN8289
CAS No.:30252-40-5
- Rosiglitazone HCl
Catalog No.:BCC2269
CAS No.:302543-62-0
- Bisline
Catalog No.:BCN2062
CAS No.:30258-28-7
[Therapeutic potential of Ro 5-3335. New active benzodiazepine in vitro against HIV-1 viruses resistant or non-resistant to AZT].[Pubmed:8351676]
Therapie. 1993 Mar-Apr;48(2):105-7.
Ro 5-3335 is a new benzodiazepine highly active in vitro (IC50 = 0.1-1.0 microM [corrected]) against HIV-1 viruses of AIDS resistant or non-resistant to zidovudine (AZT). It is also active against HIV-2. Ro 5-3335 is original by its mechanism of action, acting on the trans-activation factor of transcription (TAT) and non on the reverse transcriptase. Such as, it could prevent proviral DNA to express in both evolutive and silent AIDS resistant or non-resistant to AZT or to other anti-reverse transcriptase series. In addition, in antagonizing extracellular TAT's actions, Ro 5-3335 could alleviate the syndrome commonly associated with AIDS as Kaposi's syndrome. In rodent test, Ro 5-3335 has no diazepam-like central effects and presents in comparison to AZT a more favorable therapeutic index. In dog, the elimination half-life, peak concentration and availability are 2 h, 0.8 microM and 85% respectively, after a 1 mg.kg-1 oral dose of Ro 5-3335. Theoretically, Ro 5-3335 and now its analogue Ro 24-7429 seem to possess all virtues to antagonize evolutive and latent AIDS. Its arrival is timely to cope with the ever increasing resistance phenomena, lengthy development of AIDS vaccines, exponential contamination of populations worldwide and last but not least possibly to impede evolutions of the disease. Ability to manipulate TAT-mediated activation of HIV-1 genes paves the ways to study conceivable corrections of abnormal gene expressions of neurotransmitters, hormones, oncogenes and key enzymes.
The human immunodeficiency virus type 1 Tat antagonist, Ro 5-3335, predominantly inhibits transcription initiation from the viral promoter.[Pubmed:7884917]
J Virol. 1995 Apr;69(4):2640-3.
Tat, the transcriptional transactivator protein of the human immunodeficiency virus type 1 (HIV-1), is required for viral replication in vitro. The Tat antagonist, Ro 5-3335, and its analog, Ro 24-7429, have been shown to inhibit replication of HIV-1 and to reduce steady-state viral RNA in infected cells (M.-C. Hsu et al., Science 254:1799-1802, 1991, and M.-C. Hsu et al., Proc. Natl. Acad. Sci. USA 90:6395-6399, 1993). Analysis of HIV-1 long terminal repeat-driven reporter gene transcription in a recombinant adenovirus by nuclear run-on assay indicated that the drug predominantly inhibits Tat-dependent initiation and also exerts a measurable effect on elongation. This result may imply a common mechanism for Tat-mediated transcription initiation and elongation.
HIV replication in chronically infected macrophages is not inhibited by the Tat inhibitors Ro-5-3335 and Ro-24-7429.[Pubmed:8083610]
J Leukoc Biol. 1994 Sep;56(3):369-73.
Human immunodeficiency virus infects different cell types including CD4+ lymphocytes and monocyte-derived macrophages (MDMs). We have examined the activity of the HIV-1 Tat inhibitors Ro-5-3335 and Ro-24-7429 in cultured human peripheral MDMs. Monocytes were isolated from HIV-seronegative donors by gradient centrifugation and plastic adherence. MDMs and unfractionated peripheral blood mononuclear cells (PBMCs) were infected with HIV Ba-L and then treated with drug either immediately (acute infection) or after 4 days (PBMCs) or 14 days (MDMs) (chronic infection). Inhibition of HIV replication by each drug was assessed by quantitation of HIV p24 antigen in culture supernatant using an enzyme immunoassay. In acutely infected MDMs, Ro-5-3335 (10 microM) and Ro-24-7429 (10 microM) resulted in 77% and 99% mean inhibition, respectively, of HIV replication with a clear dose response at lower concentrations; chronically infected MDMs were much less susceptible to these drugs, with both compounds inhibiting p24 antigen production by less than 50% at 10 microM. The drugs had no deleterious effect on cell viability at any concentration tested. In acutely infected PBMCs Ro-5-3335 and Ro-24-7429 resulted in 68% and 68.5% mean inhibition at 10 microM; when the compounds were added 4 days after infection inhibition was less than 50% compared with controls. Thus, the Tat inhibitors were effective in inhibiting acute HIV infection in MDMs but not in chronically infected cells, findings that differ from those of published studies using continuous lymphoblastoid cell lines.


