SCH 221510Potent and selective NOP agonist CAS# 322473-89-2 |
- MLN8237 (Alisertib)
Catalog No.:BCC2166
CAS No.:1028486-01-2
- VX-680 (MK-0457,Tozasertib)
Catalog No.:BCC2167
CAS No.:639089-54-6
- Reversine
Catalog No.:BCC1892
CAS No.:656820-32-5
- AZD1152
Catalog No.:BCC1393
CAS No.:722543-31-9
- XL228
Catalog No.:BCC2058
CAS No.:898280-07-4
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 322473-89-2 | SDF | Download SDF |
| PubChem ID | 9887077 | Appearance | Powder |
| Formula | C28H31NO | M.Wt | 397.55 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in DMSO | ||
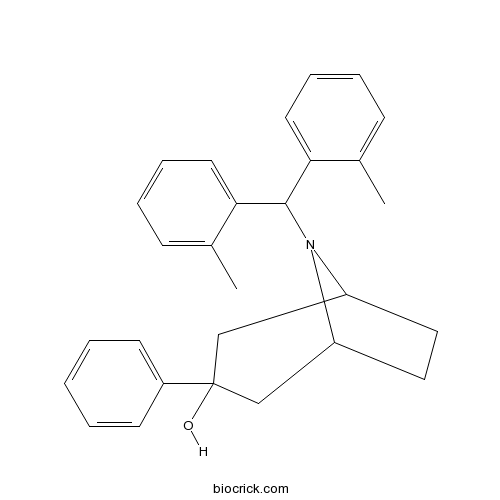
| Chemical Name | 8-[bis(2-methylphenyl)methyl]-3-phenyl-8-azabicyclo[3.2.1]octan-3-ol | ||
| SMILES | CC1=CC=CC=C1C(C2=CC=CC=C2C)N3C4CCC3CC(C4)(C5=CC=CC=C5)O | ||
| Standard InChIKey | LOSJNRBXNQTUNT-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C28H31NO/c1-20-10-6-8-14-25(20)27(26-15-9-7-11-21(26)2)29-23-16-17-24(29)19-28(30,18-23)22-12-4-3-5-13-22/h3-15,23-24,27,30H,16-19H2,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Potent and selective nociceptin opioid receptor (NOP) agonist (EC50 values are 12, 693, 683 and 8071 nM, and Ki values are 0.3, 65, 131 and 2854 nM at NOP, μ-, κ- and δ-opioid receptors respectively). Exhibits anxiolytic-like activity at doses that have no effect on overt behaviors such as locomotion in vivo. Orally active. |

SCH 221510 Dilution Calculator

SCH 221510 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5154 mL | 12.577 mL | 25.1541 mL | 50.3081 mL | 62.8852 mL |
| 5 mM | 0.5031 mL | 2.5154 mL | 5.0308 mL | 10.0616 mL | 12.577 mL |
| 10 mM | 0.2515 mL | 1.2577 mL | 2.5154 mL | 5.0308 mL | 6.2885 mL |
| 50 mM | 0.0503 mL | 0.2515 mL | 0.5031 mL | 1.0062 mL | 1.2577 mL |
| 100 mM | 0.0252 mL | 0.1258 mL | 0.2515 mL | 0.5031 mL | 0.6289 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 2alpha,7beta,13alpha-Triacetoxy-5alpha-cinnamoyloxy-9beta-hydroxy-2(3->20)abeotaxa-4(20),11-dien-10-one
Catalog No.:BCN7208
CAS No.:322471-42-1
- Calcitriol
Catalog No.:BCC4950
CAS No.:32222-06-3
- Lupeol palmitate
Catalog No.:BCN7133
CAS No.:32214-80-5
- H-Asp(OMe)-OMe.HCl
Catalog No.:BCC2890
CAS No.:32213-95-9
- Heraclenol 3'-O-beta-D-glucopyranoside
Catalog No.:BCN1459
CAS No.:32207-10-6
- Triflusal
Catalog No.:BCC4443
CAS No.:322-79-2
- p-3-Methylamino propyl phenol
Catalog No.:BCN1802
CAS No.:32180-92-0
- 1,10:4,5-Diepoxy-7(11)-germacren-8-one
Catalog No.:BCN1460
CAS No.:32179-18-3
- Pilloin
Catalog No.:BCN6817
CAS No.:32174-62-2
- L002
Catalog No.:BCC8000
CAS No.:321695-57-2
- Poloxin
Catalog No.:BCC1867
CAS No.:321688-88-4
- BIBR 1532
Catalog No.:BCC1147
CAS No.:321674-73-1
- Methyl 3-cyclopropyl-3-oxopropionate
Catalog No.:BCC9038
CAS No.:32249-35-7
- 4-Methoxy-1-methylquinolin-2-one
Catalog No.:BCN4824
CAS No.:32262-18-3
- H-Threoninol
Catalog No.:BCC2705
CAS No.:3228-51-1
- Triphosgene
Catalog No.:BCC2848
CAS No.:32315-10-9
- SCS
Catalog No.:BCC7266
CAS No.:3232-36-8
- p-Cresyl sulfate
Catalog No.:BCC4013
CAS No.:3233-58-7
- 4-Nitrobenzyl carbamate
Catalog No.:BCN3286
CAS No.:32339-07-4
- 9,9-Bis(4-hydroxyphenyl)fluorene
Catalog No.:BCC8795
CAS No.:3236-71-3
- Medicarpin
Catalog No.:BCN5241
CAS No.:32383-76-9
- Imetit dihydrobromide
Catalog No.:BCC6768
CAS No.:32385-58-3
- Dexfenfluramine hydrochloride
Catalog No.:BCC5927
CAS No.:3239-45-0
- AKTide-2T
Catalog No.:BCC5908
CAS No.:324029-01-8
Anti-inflammatory and antinociceptive action of an orally available nociceptin receptor agonist SCH 221510 in a mouse model of inflammatory bowel diseases.[Pubmed:24345466]
J Pharmacol Exp Ther. 2014 Mar;348(3):401-9.
The nociceptin receptors (NOPs) are expressed in the gastrointestinal (GI) tract on muscle cell membranes and neurons, as well as the immune cells that infiltrate the mucosa. The involvement of NOPs in the pathophysiology of GI inflammation has been suggested, but due to the lack of selective NOP agonists, it never fully elucidated. Our aim was to characterize the anti-inflammatory and antinociceptive effect of the NOP agonist, SCH 221510 [3-endo-8-[bis(2-methylphenyl)methyl]-3-phenyl-8-azabicyclo [3.2.1]octan-3-ol], as a potential therapeutic strategy in the treatment of inflammatory bowel diseases (IBD). The anti-inflammatory action of SCH 221510 was determined after intraperitoneal, oral, and intracolonic administration of SCH 221510 (0.1-3.0 mg/kg once or twice daily) in mice treated with 2,4,6-trinitrobenzenesulfonic acid (TNBS). Antinociceptive action of SCH 221510 was evaluated in the mouse model of mustard oil (MO)-induced abdominal pain. Relative NOP mRNA expression was assessed in patients with IBD using real-time reverse transcriptase-polymerase chain reaction. We found that the expression of NOP mRNA was significantly decreased in patients with IBD. The administration (0.1 and 1.0 mg/kg i.p. twice daily and 3 mg/kg p.o. twice daily) of SCH 221510 attenuated TNBS colitis in mice. This effect was blocked by a selective NOP antagonist [J-113397 [(+/-)-1-[(3R*,4R*)-1-(cyclooctylmethyl)-3-(hydroxymethyl)-4-piperidinyl]-3-ethyl -1,3-dihydro-2H-benzimidazol-2-one]]. The intracolonic injections of SCH 221510 did not improve colitis in mice. The antinociceptive effect of SCH 221510 was observed after oral administration of SCH 221510 in MO-induced pain tests in mice with acute colitis. In conclusion, our results show a potent anti-inflammatory and antinociceptive effect upon selective activation of NOP receptors and suggest that the NOP agonist SCH 221510 is a promising drug candidate for future treatment of IBD.
The anxiolytic-like effects of the novel, orally active nociceptin opioid receptor agonist 8-[bis(2-methylphenyl)methyl]-3-phenyl-8-azabicyclo[3.2.1]octan-3-ol (SCH 221510).[Pubmed:18492950]
J Pharmacol Exp Ther. 2008 Aug;326(2):672-82.
Orphanin FQ/nociceptin (OFQ/N) is the endogenously occurring peptide ligand for the nociceptin opioid receptor (NOP) that produces anxiolytic-like effects in mice and rats. The present study assessed the anxiolytic-like activity of 8-[bis(2-methylphenyl)-methyl]-3-phenyl-8-azabicyclo[3.2.1]octan-3-ol (SCH 221510), a novel potent piperidine NOP agonist (EC(50) = 12 nM) that binds with high affinity (K(i) = 0.3 nM) and functional selectivity (>50-fold over the mu-, kappa-, and delta-opioid receptors). The anxiolytic-like activity and side-effect profile of SCH 221510 were assessed in a variety of models and the benzodiazepine, chlordiazepoxide (CDP), was included for comparison. The effects of chronic dosing of SCH 221510 were also assessed. Furthermore, the specificity of the anxiolytic-like effect of SCH 221510 was investigated with the NOP receptor antagonist 1-[(3R,4R)-1-cyclooctylmethyl-3-hydroxymethyl-4-piperidyl]-3-ethyl-1,3-dihydro-2H -benzimidazol-2-one (J-113397) and the opioid receptor antagonist naltrexone. Like CDP (1-30 mg/kg i.p.), SCH 221510 (1-30 mg/kg p.o.) produced anxiolytic-like effects in the elevated plus-maze (rat and gerbil), Vogel conflict (rat), conditioned lick suppression (rat), fear-potentiated startle (rat), and pup separation-induced vocalization (guinea pig) assays. In the Vogel conflict, the anxiolytic-like effect of SCH 221510 (10 mg/kg) was attenuated by J-113397 (3-10 mg/kg p.o.), but not naltrexone (3-30 mg/kg i.p.). Additionally, the anxiolytic-like effects of SCH 221510 did not change appreciably following 14-day b.i.d. dosing in rats (10 mg/kg). Furthermore, unlike CDP, SCH 221510 (3-30 mg/kg) produced anxiolytic-like activity at doses that did not disrupt overt behavior. Collectively, these data suggest that NOP agonists such as SCH 221510 may have an anxiolytic-like profile similar to benzodiazepines, with a reduced side-effect liability.
Activation of the endogenous nociceptin system by selective nociceptin receptor agonist SCH 221510 produces antitransit and antinociceptive effect: a novel strategy for treatment of diarrhea-predominant IBS.[Pubmed:25041572]
Neurogastroenterol Motil. 2014 Nov;26(11):1539-50.
BACKGROUND: Diarrhea-predominant irritable bowel syndrome (IBS-D) is a functional gastrointestinal (GI) disorder, defined by the presence of loose stools and abdominal pain. In search for a novel anti-IBS-D therapy, here we investigated the nociceptin receptor (NOP)-dependent effects in the GI tract. METHODS: A novel potent and selective NOP agonist SCH 221510 was used in the study. The effect of NOP activation on mouse intestinal motility was characterized in vitro and in vivo, in physiological conditions and in animal models of hypermotility and diarrhea. Well-established mouse models of visceral pain were used to characterize the antinociceptive effect of the NOP activation. To provide additional evidence that the endogenous nociceptin system is a relevant target for IBS, NOP expression and nociceptin levels were quantified in serum and colonic biopsies from IBS-D patients. KEY RESULTS: SCH 221510 produced a potent NOP-mediated inhibitory effect on mouse intestinal motility in vitro and in vivo in physiological conditions. The NOP agonist displayed an antidiarrheal and analgesic action after oral administration in animal models mimicking the symptoms of IBS-D. Studies on human samples revealed a strong decrease in endogenous nociceptin system expression in IBS-D patients compared with healthy controls. CONCLUSIONS & INFERENCES: Collectively, mouse and human data suggest that the endogenous nociceptin system is involved in IBS-D and may become a target for anti-IBS-D treatments using potent and selective synthetic NOP agonists.
Identification of 3-substituted N-benzhydryl-nortropane analogs as nociceptin receptor ligands for the management of cough and anxiety.[Pubmed:19332374]
Bioorg Med Chem Lett. 2009 May 1;19(9):2482-6.
A series of nortropane analogs based on previously reported compound 1 have been synthesized and shown to bind to the nociceptin receptor with high affinity. The synthesis and structure-activity relationships around the C-3 nortropane substitution are described. From the SAR study and hPXR screening effort, compound 15 was identified to possess potent oral antitussive and anxiolytic-like activities in the guinea pig models.
The nociceptin/orphanin FQ receptor: a target with broad therapeutic potential.[Pubmed:18670432]
Nat Rev Drug Discov. 2008 Aug;7(8):694-710.
Identification of the enigmatic nociceptin/orphanin FQ peptide (N/OFQ) in 1995 represented the first successful use of reverse pharmacology and led to deorphanization of the N/OFQ receptor (NOP). Subsequently, the N/OFQ-NOP system has been implicated in a wide range of biological functions, including pain, drug abuse, cardiovascular control and immunity. Although this could be considered a hurdle for the development of pharmaceuticals selective for a specific disease indication, NOP represents a viable drug target. This article describes potential clinical indications and highlights the current status of the very limited number of clinical trials.


