SitostenoneCAS# 1058-61-3 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1058-61-3 | SDF | Download SDF |
| PubChem ID | 241573 | Appearance | Cryst. |
| Formula | C29H48O | M.Wt | 412.7 |
| Type of Compound | Steroids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
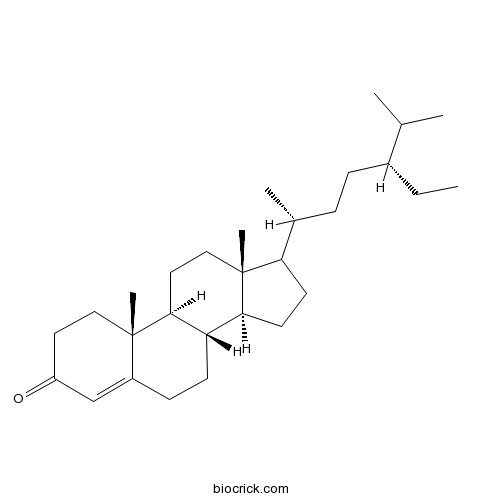
| Chemical Name | (8S,9S,10R,13R,14S)-17-[(2R,5R)-5-ethyl-6-methylheptan-2-yl]-10,13-dimethyl-1,2,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-3-one | ||
| SMILES | CCC(CCC(C)C1CCC2C1(CCC3C2CCC4=CC(=O)CCC34C)C)C(C)C | ||
| Standard InChIKey | RUVUHIUYGJBLGI-VCVIHXEESA-N | ||
| Standard InChI | InChI=1S/C29H48O/c1-7-21(19(2)3)9-8-20(4)25-12-13-26-24-11-10-22-18-23(30)14-16-28(22,5)27(24)15-17-29(25,26)6/h18-21,24-27H,7-17H2,1-6H3/t20-,21-,24+,25?,26+,27+,28+,29-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. β-Sitostenone has antioxidation activity. 2. Stigmasta-4-en-3-one(Sitostenone) can be used for the treatment of androgen-dependent diseases, especially for the treatment of benign prostatic hyperplasia. |

Sitostenone Dilution Calculator

Sitostenone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.4231 mL | 12.1153 mL | 24.2307 mL | 48.4614 mL | 60.5767 mL |
| 5 mM | 0.4846 mL | 2.4231 mL | 4.8461 mL | 9.6923 mL | 12.1153 mL |
| 10 mM | 0.2423 mL | 1.2115 mL | 2.4231 mL | 4.8461 mL | 6.0577 mL |
| 50 mM | 0.0485 mL | 0.2423 mL | 0.4846 mL | 0.9692 mL | 1.2115 mL |
| 100 mM | 0.0242 mL | 0.1212 mL | 0.2423 mL | 0.4846 mL | 0.6058 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Fmoc-Ser(tBu)-OPfp
Catalog No.:BCC3545
CAS No.:105751-13-1
- Methyl ganoderate C6
Catalog No.:BCN3259
CAS No.:105742-81-2
- Ganoderic acid C6
Catalog No.:BCN3257
CAS No.:105742-76-5
- AL 8697
Catalog No.:BCC8037
CAS No.:1057394-06-5
- Androstanolone 17-benzoate
Catalog No.:BCC8825
CAS No.:1057-07-4
- AT13148
Catalog No.:BCC5360
CAS No.:1056901-62-2
- CYT387 sulfate salt
Catalog No.:BCC1506
CAS No.:1056636-06-6
- CYT387
Catalog No.:BCC2196
CAS No.:1056634-68-4
- Mizolastine dihydrochloride
Catalog No.:BCC4132
CAS No.:1056596-82-7
- ML 9 hydrochloride
Catalog No.:BCC6644
CAS No.:105637-50-1
- Hydroxyfasudil
Catalog No.:BCC1635
CAS No.:105628-72-6
- Fasudil (HA-1077) HCl
Catalog No.:BCC2542
CAS No.:105628-07-7
- E-3810
Catalog No.:BCC1541
CAS No.:1058137-23-7
- Nateglinide
Catalog No.:BCC5005
CAS No.:105816-04-4
- Tropisetron Hydrochloride
Catalog No.:BCC4027
CAS No.:105826-92-4
- TSTU
Catalog No.:BCC2828
CAS No.:105832-38-0
- Obtusilin
Catalog No.:BCN2697
CAS No.:105870-59-5
- (tert-Butoxycarbonyl)oxycefcapene pivoxil
Catalog No.:BCC8403
CAS No.:105889-80-3
- Taraxasterol
Catalog No.:BCN5869
CAS No.:1059-14-9
- Doxycycline HCl
Catalog No.:BCC3772
CAS No.:10592-13-9
- STEARDA
Catalog No.:BCC7288
CAS No.:105955-10-0
- OLDA
Catalog No.:BCC7138
CAS No.:105955-11-1
- Clinafloxacin CI96 AM1091
Catalog No.:BCC3754
CAS No.:105956-97-6
- Sulfocostunolide B
Catalog No.:BCN5870
CAS No.:1059671-65-6
A New Dihydrochromone Dimer and Other Secondary Metabolites from Cultures of the Marine Sponge-Associated Fungi Neosartorya fennelliae KUFA 0811 and Neosartorya tsunodae KUFC 9213.[Pubmed:29194412]
Mar Drugs. 2017 Dec 1;15(12). pii: md15120375.
A previously unreported dihydrochromone dimer, paecilin E (1), was isolated, together with eleven known compounds: beta-Sitostenone, ergosta-4,6,8 (14), 22-tetraen-3-one, cyathisterone, byssochlamic acid, dehydromevalonic acid lactone, chevalone B, aszonalenin, dankasterone A (2), helvolic acid, secalonic acid A and fellutanine A, from the culture filtrate extract of the marine sponge-associated fungus Neosartorya fennelliae KUFA 0811. Nine previously reported metabolites, including a chromanol derivative (3), (3beta, 5alpha, 22E), 3,5-dihydroxyergosta-7,22-dien-6-one (4), byssochlamic acid, hopan-3beta,22-diol, chevalone C, sartorypyrone B, helvolic acid, lumichrome and the alkaloid harmane were isolated from the culture of the marine-sponge associated fungus Neosartorya tsunodae KUFC 9213. Paecilin E (1), dankasterone A (2), a chromanol derivative (3), (3beta, 5alpha, 22E)-3,5-dihydroxyergosta-7,22-dien-6-one (4), hopan-3beta,22-diol (5), lumichrome (6), and harmane (7) were tested for their antibacterial activity against Gram-positive and Gram-negative reference and multidrug-resistant strains isolated from the environment. While paecilin E (1) was active against S. aureus ATCC 29213 and E. faecalis ATCC 29212, dankastetrone A (2) was only effective against E. faecalis ATCC 29212 and the multidrug-resistant VRE E. faecalis A5/102. Both compounds neither inhibit biofilm mass production in any of the strains at the concentrations tested nor exhibit synergistic association with antibiotics.
Uncovering the Molecular Mechanism of Anti-Allergic Activity of Silkworm Pupa-Grown Cordyceps militaris Fruit Body.[Pubmed:28367714]
Am J Chin Med. 2017;45(3):497-513.
Cordyceps militaris has been widely used as an herbal drug and tonic food in East Asia and has also been recently studied in the West because of its various pharmacological activities such as antitumoral, anti-inflammatory and immunomodulatory effects. In this study, we examined the molecular mechanism underlying the anti-allergic activity of ethanol extract prepared from silkworm pupa-cultivated Cordyceps militaris fruit bodies in activated mast cells. Our results showed that ethanol extract treatment significantly inhibited the release of [Formula: see text]-hexosaminidase (a degranulation marker) and mRNA levels of tumor necrosis factor-[Formula: see text] as well as interleukin-4 in RBL-2H3 cells. The cells were sensitized with 2,4-dinitrophenol specific IgE and then stimulated with human serum albumin conjugated with 2,4-dinitrophenol. Oral administration of 300[Formula: see text]mg/kg ethanol extract significantly ameliorated IgE-induced allergic reaction in mice with passive cutaneous anaphylaxis. Western immunoblotting results demonstrated that ethanol extract incubation significantly inhibited Syk/PI3K/MEKK4/JNK/c-jun biochemical cascade in activated RBL-2H3 cells, which activated the expression of various allergic cytokines. In addition, it suppressed Erk activation and PLC[Formula: see text] evocation, which would respectively evoke the synthesis of lipid mediators and Ca[Formula: see text] mobilization to induce degranulation in stimulated RBL-2H3 cells. A compound, identified as [Formula: see text]-Sitostenone, was shown to inhibit [Formula: see text]-hexosaminidase secretion from activated mast cells. Our study demonstrated that ethanol extract contained the ingredients, which could inhibit immediate degranulation and de novo synthesis of allergic lipid mediators and cytokines in activated mast cells.
Aristolic Acid Derivatives from the Bark of Antidesma ghaesembilla.[Pubmed:28499305]
Planta Med. 2017 Aug;83(12-13):1097-1102.
Antidesma ghaesembilla is an important medicinal and food plant in many Asian countries. Ten substances could be isolated from the dichloromethane and methanol extract: Sitostenone (3), daucosterol (4), chavibetol (5), asperphenamate (6), protocatechuic acid (7), vanillic acid-4-O-beta-D-glucoside (8), 1-O-beta-D-glucopyranosyl-3-O-methyl-phloroglucinol (9), and aristolic acid II-8-O-beta-D-glucoside (10), and two new aristolic acid derivatives, 10-amino-5,7-dimethoxy-aristolic acid II (= 6-amino-9,11-dimethoxyphenanthro[3,4-d]-1,3-dioxole-5-carboxylic acid; 1) and 5,7-dimethoxy-aristolochic acid II (= 9,11-dimethoxy-6-nitrophenantro[3,4-d]-1,3-dioxole-5-carboxylic acid; 2). Exposure to humans of some of these compounds is associated with a severe disease today known as aristolochic acid nephropathy. Therefore, the traditional usage of this plant has to be reconsidered carefully.
Acetylcholinesterase inhibitory active metabolites from the endophytic fungus Colletotrichum sp. YMF432.[Pubmed:29397775]
Nat Prod Res. 2018 Feb 4:1-4.
An endophytic fungus, Chaetomium sp. YMF432, was isolated from Huperzia serrata (Thunb. ex Murray) Trev. and subjected to phytochemical investigation based on its special environment. From the extracts of fermentation solid of strain YMF 432, eight compounds including 1-O-methylemodin (1), 5-methoxy-2-methyl-3-tricosyl-1,4-benzoquinone (2), 4,8-dihydroxy-1-tetralone (3), (3beta,5alpha,6alpha, 22E)-3-hydroxy-5,6-epoxy-7-one-8(14),22-dien-ergosta (4), ergosta-4,6,8(14),22-tetraen-3-one (5), beta-Sitostenone (6), beta-sitosterol (7) and (22E,24R)-ergosta-5,7,22 -trien-3beta-ol (8) were obtained. Their structures were elucidated on the basis of their spectroscopic data. These compounds were evaluated for acetylcholinesterase inhibitory activities in vitro. Compounds 1, 2, and 4 showed moderate acetylcholinesterase inhibitory activities (IC50 from 37.7 +/- 1.5 to 370.0 +/- 2.9 muM).


