TetraacetylphytosphingosineCAS# 13018-48-9 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 13018-48-9 | SDF | Download SDF |
| PubChem ID | 10972946.0 | Appearance | Powder |
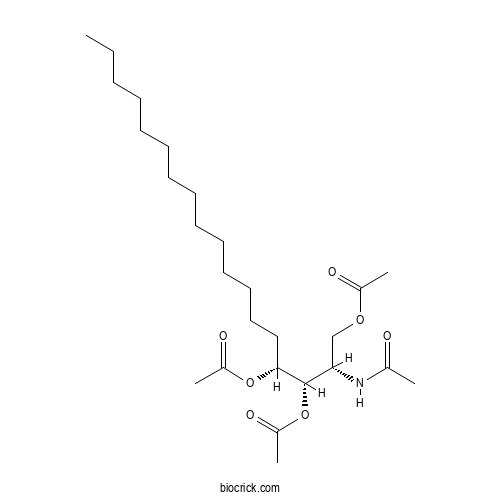
| Formula | C26H47NO7 | M.Wt | 485.66 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | [(2S,3S,4R)-2-acetamido-3,4-diacetyloxyoctadecyl] acetate | ||
| SMILES | CCCCCCCCCCCCCCC(C(C(COC(=O)C)NC(=O)C)OC(=O)C)OC(=O)C | ||
| Standard InChIKey | SGTYQWGEVAMVKB-NXCFDTQHSA-N | ||
| Standard InChI | InChI=1S/C26H47NO7/c1-6-7-8-9-10-11-12-13-14-15-16-17-18-25(33-22(4)30)26(34-23(5)31)24(27-20(2)28)19-32-21(3)29/h24-26H,6-19H2,1-5H3,(H,27,28)/t24-,25+,26-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Tetraacetylphytosphingosine Dilution Calculator

Tetraacetylphytosphingosine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.0591 mL | 10.2953 mL | 20.5905 mL | 41.1811 mL | 51.4763 mL |
| 5 mM | 0.4118 mL | 2.0591 mL | 4.1181 mL | 8.2362 mL | 10.2953 mL |
| 10 mM | 0.2059 mL | 1.0295 mL | 2.0591 mL | 4.1181 mL | 5.1476 mL |
| 50 mM | 0.0412 mL | 0.2059 mL | 0.4118 mL | 0.8236 mL | 1.0295 mL |
| 100 mM | 0.0206 mL | 0.103 mL | 0.2059 mL | 0.4118 mL | 0.5148 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- L-Guluronic Acid Sodium Salt
Catalog No.:BCX1131
CAS No.:15769-56-9
- Ostruthine
Catalog No.:BCX1130
CAS No.:148-83-4
- Agigenin
Catalog No.:BCX1129
CAS No.:55332-76-8
- Salidroside pentaacetate
Catalog No.:BCX1128
CAS No.:39032-08-1
- 2,3,4,6-tetraacetate Salidroside
Catalog No.:BCX1127
CAS No.:28251-63-0
- (-)-Epitaxifolin
Catalog No.:BCX1126
CAS No.:114761-89-6
- Zymosterol
Catalog No.:BCX1125
CAS No.:128-33-6
- Jaligonic acid B
Catalog No.:BCX1124
CAS No.:2375176-78-4
- Monensin B
Catalog No.:BCX1123
CAS No.:30485-16-6
- Beta-Tomatine
Catalog No.:BCX1122
CAS No.:17406-46-1
- Orchioside B
Catalog No.:BCX1121
CAS No.:851780-22-8
- Abyssinone II
Catalog No.:BCX1120
CAS No.:77263-08-2
- β-Sitosteryl acetate
Catalog No.:BCX1133
CAS No.:915-05-9
- 3-Feruloyl-4-caffeoylquinic acid
Catalog No.:BCX1134
CAS No.:96990-65-7
- Avenanthramide B
Catalog No.:BCX1135
CAS No.:108605-69-2
- Avenanthramide A
Catalog No.:BCX1136
CAS No.:108605-70-5
- Ganosporeric acid A
Catalog No.:BCX1137
CAS No.:135357-25-4
- N-methyltyramine
Catalog No.:BCX1138
CAS No.:370-98-9
- 3α-Hydroxymogrol
Catalog No.:BCX1139
CAS No.:1343402-73-2
- Ligustrosidic acid
Catalog No.:BCX1140
CAS No.:96382-89-7
- Presenegenin
Catalog No.:BCX1141
CAS No.:2163-40-8
- Hydroxypropyl tetrahydropyrantriol
Catalog No.:BCX1142
CAS No.:439685-79-7
- Euphornin
Catalog No.:BCX1143
CAS No.:80454-47-3
- Reptoside
Catalog No.:BCX1144
CAS No.:53839-03-5
Differences in the Fatty Acid Profile, Morphology, and Tetraacetylphytosphingosine-Forming Capability Between Wild-Type and Mutant Wickerhamomyces ciferrii.[Pubmed:34178960]
Front Bioeng Biotechnol. 2021 Jun 9;9:662979.
One Tetraacetylphytosphingosine (TAPS)-producing Wickerhamomyces ciferrii mutant was obtained by exposing wild-type W. ciferrii to gamma-ray irradiation. The mutant named 736 produced up to 9.1 g/L of TAPS (218.7 mg-TAPS/g-DCW) during batch fermentation in comparison with 1.7 g/L of TAPS (52.2 mg-TAPS/g-DCW) for the wild type. The highest production, 17.7 g/L of TAPS (259.6 mg-TAPS/g-DCW), was obtained during fed-batch fermentation by mutant 736. Fatty acid (FA) analysis revealed an altered cellular FA profile of mutant 736: decrease in C16:0 and C16:1 FA levels, and increase in C18:1 and C18:2 FA levels. Although a significant change in the cellular FA profile was observed, scanning electron micrographs showed that morphology of wild-type and mutant 736 cells was similar. Genetic alteration analysis of eight TAPS biosynthesis-related genes revealed that there are no mutations in these genes in mutant 736; however, mRNA expression analysis indicated 30% higher mRNA expression of TCS10 among the eight genes in mutant 736 than that in the wild-type. Collectively, these results imply that the enhancement of TAPS biosynthesis in mutant 736 may be a consequence of system-level genetic and physiological alterations of a complicated metabolic network. Reverse metabolic engineering based on system-level omics analysis of mutant 736 can make the mutant more suitable for commercial production of TAPS.
Engineering Yarrowia lipolytica for de novo production of tetraacetyl phytosphingosine.[Pubmed:33190388]
J Appl Microbiol. 2021 Jun;130(6):1981-1992.
AIMS: To genetically engineer the oleaginous yeast Yarrowia lipolytica for de novo production of Tetraacetylphytosphingosine (TAPS), a precursor of phytosphingosine, and optimization of fermentation conditions for high yield. METHODS AND RESULTS: We successfully constructed a TAPS-producing Y. lipolytica CE3 strain by co-expression of Wickerhamomyces ciferrii-derived acetyl transferases, Sli1p and Atf2p. Next, we optimized several environmental factors including temperature, initial pH and C/N ratio for TAPS production in a shake culture. Deletion of LCB4 in CE3 strain increased the volumetric TAPS titre and cell-specific yield to 142.1 +/- 10.7 mg(TAPS) l(-1) and 3.08 +/- 0.11 mg(TAPS) g(DCW) (-1) , respectively, in a shake flask culture incubated for 120 h at 28 degrees C with glycerol as the carbon source. Finally, we developed a 5-l fed-batch process with NaOH-mediated pH control and olive oil as a carbon source, exhibiting 650 +/- 24 mg(TAPS) l(-1) of TAPS production within 56 h of the fermentation. CONCLUSIONS: The introduction of codon-optimized Sli1p and Atf2p, deletion of LCB4 gene and sexual hybridization, accompanied by specific fermentation conditions, enhanced TAPS yield in Y. lipolytica. SIGNIFICANCE AND IMPACT OF THE STUDY: Our results highlight Y. lipolytica as a promising candidate for the industrial production of TAPS, an important component of cosmetic formulations.
Combined application of targeted and untargeted proteomics identifies distinct metabolic alterations in the tetraacetylphytosphingosine (TAPS) producing yeast Wickerhamomyces ciferrii.[Pubmed:23500128]
J Proteomics. 2013 Apr 26;82:274-87.
The Wickerhamomyces ciferrii strain NRRL Y-1031 F-60-10A is a well-known producer of Tetraacetylphytosphingosine (TAPS) and used for the biotechnological production of sphingolipids and ceramides. It was our aim to gain new biological insights into the sphingolipid metabolism by employing a dual platform mass spectrometry strategy. The first step comprised metabolic (15)N-labeling in combination with label-free proteomics using high resolution LTQ Orbitrap mass spectrometry. Then selected reaction monitoring tandem mass spectrometry served for the targeted quantification of sphingoid base biosynthesis enzymes. The non-producer strain NRRL Y-1031-27 served as a reference strain. In total, 1697 proteins were identified, and 123 enzymes of main metabolic pathways were observed as differentially expressed. Major findings were: 1) the likely presence of higher carbon flux in NRRL Y-1031 F-60-10A, resulting in higher availability of pyruvate and acetyl-CoA; 2) indications of oleaginous yeast-like behavior in NRRL Y-1031 F-60-10A; and 3) approx. 2-fold higher abundance of eight sphingolipid biosynthesis enzymes in NRRL Y-1031 F-60-10A. Taken together, in NRRL Y-1031 F-60-10A, the levels of glycolytic enzymes apparently gear carbon flux towards higher acetyl-CoA synthesis rates, while simultaneously reducing protein biosynthesis in the process. Thereby, C2 building blocks for acyl-moieties and downstream sphingoid base acetylation are provided to maintain TAPS production. BIOLOGICAL SIGNIFICANCE: First quantitative proteome study for a phytosphingosine-producing yeast.
Phytoceramide shows neuroprotection and ameliorates scopolamine-induced memory impairment.[Pubmed:22037667]
Molecules. 2011 Oct 28;16(11):9090-100.
The function and the role phytoceramide (PCER) and phytosphingosine (PSO) in the central nervous system has not been well studied. This study was aimed at investigating the possible roles of PCER and PSO in glutamate-induced neurotoxicity in cultured neuronal cells and memory function in mice. Phytoceramide showed neuro-protective activity in the glutamate-induced toxicity in cultured cortical neuronal cells. Neither phytosphingosine nor Tetraacetylphytosphingosine (TAPS) showed neuroproective effects in neuronal cells. PCER (50 mg/kg, p.o.) recovered the scopolamine-induced reduction in step-through latency in the passive avoidance test; however, PSO did not modulate memory function on this task. The ameliorating effects of PCER on spatial memory were confirmed by the Morris water maze test. In conclusion, through behavioral and neurochemical experimental results, it was demonstrated that central administration of PCER produces amelioration of memory impairment. These results suggest that PCER plays an important role in neuroprotection and memory enhancement and PCER could be a potential new therapeutic agent for the treatment of neurodegenerative diseases such as Alzheimer's disease.
Quantification of the wound healing using polarization-sensitive optical coherence tomography.[Pubmed:16965152]
J Biomed Opt. 2006 Jul-Aug;11(4):041124.
We use polarization-sensitive optical coherence tomography (PS-OCT) to monitor the wound healing process in vitro and in vivo, which are affected by various drugs. Five rabbit subjects are used for in vitro studies and another five are used for in vivo studies. The in vitro studies are conducted to compare the PS-OCT images with histopathology. For each subject, three biopsy lesions are created on each ear: one site is not treated (control); the second site is treated with sphingosylphosphorylcholine, which is expected to promote healing; and the last is administered with Tetraacetylphytosphingosine, which negatively affects the healing process. Each site is examined with a PS-OCT system at 1, 4, 7, 10, and 14- days after wound generation. The variations of phase retardation values caused by the collagen morphology changes on wound sites are quantified for all cases. Our results suggest that PS-OCT may be a useful tool for visualization of collagen fiber regeneration and for quantification of various drug effects during the wound healing process.
Formation of Extracellular Sphingolipids by Microorganisms: IV. Pilot-Plant Production of Tetraacetylphytosphingosine by Hansenula ciferrii.[Pubmed:16349621]
Appl Microbiol. 1962 Sep;10(5):401-6.
Tetraacetylphytosphingosine (TAPS) formation by the F-60-10 mating type strain of the yeast Hansenula ciferrii, previously observed on agar plates, has been shown to take place in submerged cultures. The optimal conditions for TAPS formation, and the correlation of TAPS production and sugar utilization under aerobic conditions, were studied in 10-liter fermentors. For each gram of glucose consumed, 5 mg of TAPS were formed; for each gram of yeast solids produced, 15 mg of TAPS were synthesized. A 750-liter pilot-plant run yielded 175 g of crude TAPS, which were obtained by hexane extraction of centrifuged yeast cells.


