Avenanthramide ACAS# 108605-70-5 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 108605-70-5 | SDF | Download SDF |
| PubChem ID | 5281157.0 | Appearance | Powder |
| Formula | C16H13NO5 | M.Wt | 299.28 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
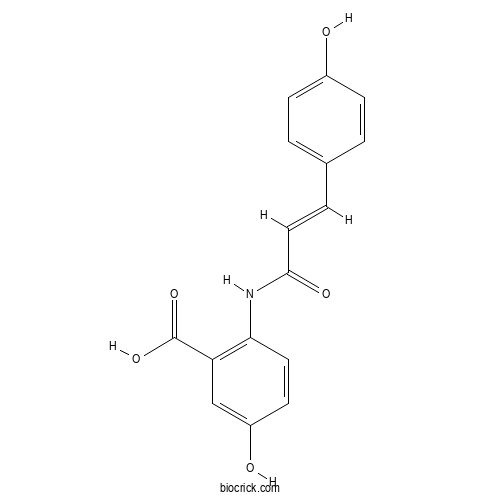
| Chemical Name | 5-hydroxy-2-[[(E)-3-(4-hydroxyphenyl)prop-2-enoyl]amino]benzoic acid | ||
| SMILES | C1=CC(=CC=C1C=CC(=O)NC2=C(C=C(C=C2)O)C(=O)O)O | ||
| Standard InChIKey | QGUMNWHANDITDB-FPYGCLRLSA-N | ||
| Standard InChI | InChI=1S/C16H13NO5/c18-11-4-1-10(2-5-11)3-8-15(20)17-14-7-6-12(19)9-13(14)16(21)22/h1-9,18-19H,(H,17,20)(H,21,22)/b8-3+ | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Avenanthramide A Dilution Calculator

Avenanthramide A Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.3414 mL | 16.7068 mL | 33.4135 mL | 66.8271 mL | 83.5338 mL |
| 5 mM | 0.6683 mL | 3.3414 mL | 6.6827 mL | 13.3654 mL | 16.7068 mL |
| 10 mM | 0.3341 mL | 1.6707 mL | 3.3414 mL | 6.6827 mL | 8.3534 mL |
| 50 mM | 0.0668 mL | 0.3341 mL | 0.6683 mL | 1.3365 mL | 1.6707 mL |
| 100 mM | 0.0334 mL | 0.1671 mL | 0.3341 mL | 0.6683 mL | 0.8353 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Avenanthramide B
Catalog No.:BCX1135
CAS No.:108605-69-2
- 3-Feruloyl-4-caffeoylquinic acid
Catalog No.:BCX1134
CAS No.:96990-65-7
- β-Sitosteryl acetate
Catalog No.:BCX1133
CAS No.:915-05-9
- Tetraacetylphytosphingosine
Catalog No.:BCX1132
CAS No.:13018-48-9
- L-Guluronic Acid Sodium Salt
Catalog No.:BCX1131
CAS No.:15769-56-9
- Ostruthine
Catalog No.:BCX1130
CAS No.:148-83-4
- Agigenin
Catalog No.:BCX1129
CAS No.:55332-76-8
- Salidroside pentaacetate
Catalog No.:BCX1128
CAS No.:39032-08-1
- 2,3,4,6-tetraacetate Salidroside
Catalog No.:BCX1127
CAS No.:28251-63-0
- (-)-Epitaxifolin
Catalog No.:BCX1126
CAS No.:114761-89-6
- Zymosterol
Catalog No.:BCX1125
CAS No.:128-33-6
- Jaligonic acid B
Catalog No.:BCX1124
CAS No.:2375176-78-4
- Ganosporeric acid A
Catalog No.:BCX1137
CAS No.:135357-25-4
- N-methyltyramine
Catalog No.:BCX1138
CAS No.:370-98-9
- 3α-Hydroxymogrol
Catalog No.:BCX1139
CAS No.:1343402-73-2
- Ligustrosidic acid
Catalog No.:BCX1140
CAS No.:96382-89-7
- Presenegenin
Catalog No.:BCX1141
CAS No.:2163-40-8
- Hydroxypropyl tetrahydropyrantriol
Catalog No.:BCX1142
CAS No.:439685-79-7
- Euphornin
Catalog No.:BCX1143
CAS No.:80454-47-3
- Reptoside
Catalog No.:BCX1144
CAS No.:53839-03-5
- Jaligonic acid
Catalog No.:BCX1145
CAS No.:51776-39-7
- 24(28)-Dehydroergosterol
Catalog No.:BCX1146
CAS No.:29560-24-5
- Eicosapentaenoic acid
Catalog No.:BCX1147
CAS No.:10417-94-4
- Toralactone
Catalog No.:BCX1148
CAS No.:41743-74-2
Ferulic Acid Derivatives and Avenanthramides Modulate Endothelial Function through Maintenance of Nitric Oxide Balance in HUVEC Cells.[Pubmed:34204635]
Nutrients. 2021 Jun 12;13(6):2026.
Wholegrain oats contain a variety of phenolic compounds thought to help maintain healthy vascular function, through the maintenance of local levels of the vasodilator nitric oxide (NO). Thus, the full molecular mechanisms involved are not yet clear. With this work we aim to understand the possible cellular mechanisms by which avenanthramides and ferulic acid derivatives, present in oats, may help maintain a healthy vascular function through the modulation of the NO pathway. Primary Human Umbilical Vein Endothelial Cells (HUVEC) were exposed to ferulic acid, isoferulic acid, hydroferulic acid, ferulic acid 4-O-glucuronide, isoferulic acid 3-O-sulfate, dihydroferulic acid 4-O-glucuronide, Avenanthramide A, avenanthramide B and avenanthramide C (1 muM) or vehicle (methanol) for 24 h. Apocynin and Nomega-Nitro-L-arginine (L-NNA) were additionally included as controls. NO and cyclic GMP (cGMP) levels, superoxide production and the activation of the Akt1/eNOS pathway were assessed. The statistical analysis was performed using one-way ANOVA followed by a Tukey post-hoc t-test. Apocynin and all phenolic compounds increased NO levels in HUVEC cells (increased DAF2-DA fluorescence and cGMP), and significantly reduced superoxide levels. Protein expression results highlighted an increase in the Akt1 activation state, and increased eNOS expression. Overall, our results indicated that the glucuronide metabolites do not enhance NO production through the Akt1/eNOS pathway, thus all compounds tested are able to reduce NO degradation through reduced superoxide formation.
Avenanthramide A triggers potent ROS-mediated anti-tumor effects in colorectal cancer by directly targeting DDX3.[Pubmed:31391454]
Cell Death Dis. 2019 Aug 7;10(8):593.
Colorectal cancer (CRC) is a common malignant gastrointestinal tumor with high mortality worldwide. Drug resistance and cytotoxicity to normal cells are the main causes of chemotherapeutic treatment failure in CRC. Therefore, extracting the bioactive compounds from natural products with anti-carcinogenic activity and minimal side-effects is a promising strategy against CRC. The present study aims to evaluate the anti-carcinogenic properties of avenanthramides (AVNs) extracted from oats bran and clarify the underlying molecular mechanisms. We demonstrated that AVNs treatment suppressed mitochondrial bioenergetic generation, resulting in mitochondrial swelling and increased reactive oxygen species (ROS) production. Further study indicated that AVNs treatment significantly reduced DDX3 expression, an oncogenic RNA helicase highly expressed in human CRC tissues. DDX3 overexpression reversed the ROS-mediated CRC apoptosis induced by AVNs. Of note, we identified Avenanthramide A (AVN A) as the effective ingredient in AVNs extracts. AVN A blocked the ATPase activity of DDX3 and induced its degradation by directly binding to the Arg287 and Arg294 residues in DDX3. In conclusion, these innovative findings highlight that AVNs extracts, in particular its bioactive compound AVN A may crack the current hurdles in the way of CRC treatment.
Avenanthramide A Induces Cellular Senescence via miR-129-3p/Pirh2/p53 Signaling Pathway To Suppress Colon Cancer Growth.[Pubmed:30888162]
J Agric Food Chem. 2019 May 1;67(17):4808-4816.
Cellular senescence is the state of irreversible cell cycle arrest that provides a blockade during oncogenic transformation and tumor development. Avenanthramide A (AVN A) is an active ingredient exclusively extracted from oats, which possesses antioxidant, anti-inflammatory, and anticancer activities. However, the underlying mechanism(s) of AVN A in the prevention of cancer progression remains unclear. In the current study, we revealed that AVN A notably attenuated tumor formation in an azoxymethane/dextran sulfate sodium (AOM/DSS) mouse model. AVN A treatment triggered cellular senescence in human colon cancer cells, evidenced by enlarging cellular size, upregulating beta-galactosidase activity, gamma-H2AX positive staining, and G1 phase arrest. Moreover, AVN A treatment significantly increased the expression of miR-129-3p, which markedly repressed the E3 ubiquitin ligase Pirh2 and two other targets, IGF2BP3 and CDK6. The Pirh2 silencing by miR-129-3p led to a significant increase in protein levels of p53 and its downstream target p21, which subsequently induced cell senescence. Taken together, our data indicate that miR-129-3p/Pirh2/p53 is a critical signaling pathway in AVN A induced cellular senescence and AVN A could be a potential chemopreventive strategy for cancer treatment.
Avenanthramides are bioavailable and have antioxidant activity in humans after acute consumption of an enriched mixture from oats.[Pubmed:17513394]
J Nutr. 2007 Jun;137(6):1375-82.
The consumption of polyphenols is associated with a decreased risk of cardiovascular disease. Avenanthramides (AV), alkaloids occurring only in oats, may have anti-atherosclerotic activity, but there is no information concerning their bioavailability and bioactivity in humans. We characterized the pharmacokinetics and antioxidant action of Avenanthramide A, B, and C in healthy older adults in a randomized, placebo-controlled, 3-way crossover trial with 1-wk washout periods. Six free-living subjects (3 mol/L, 3 F; 60.8 +/- 3.6 y) consumed 360 mL skim milk alone (placebo) or containing 0.5 or 1 g avenanthramide-enriched mixture (AEM) extracted from oats. Plasma samples were collected over a 10-h period. Concentrations of AV-A, AV-B, and AV-C in the AEM were 154, 109, and 111 micromol/g, respectively. Maximum plasma concentrations of AV (free + conjugated) after consumption of 0.5 and 1 g AEM were 112.9 and 374.6 nmol/L for AV-A, 13.2 and 96.0 nmol/L for AV-B, and 41.4 and 89.0 nmol/L for AV-C, respectively. Times to reach the C(max) for both doses were 2.30, 1.75, and 2.15 h for AV-A, AV-B, and AV-C and half times for elimination were 1.75, 3.75, and 3.00 h, respectively. The elimination kinetics of plasma AV appeared to follow first-order kinetics. The bioavailability of AV-A was 4-fold larger than that of AV-B at the 0.5 g AEM dose. After consumption of 1 g AEM, plasma reduced glutathione was elevated by 21% at 15 min (P < or = 0.005) and by 14% at 10 h (P < or = 0.05). Thus, oat AV are bioavailable and increase antioxidant capacity in healthy older adults.
Metabolism of avenanthramide phytoalexins in oats.[Pubmed:15272874]
Plant J. 2004 Aug;39(4):560-72.
Oat leaves produce phytoalexins, avenanthramides, in response to infection by pathogens or treatment with elicitors. The metabolism of avenanthramides was investigated using low molecular weight, partially deacetylated chitin as an elicitor. When oat leaf segments are floated on the elicitor solution, avenanthramides accumulate in the solution. The transfer of elicited oat leaves to solutions containing stable-isotope-labeled avenanthramides resulted in a rapid decrease in the labeled avenanthramides, suggesting the metabolism of avenanthramides. The rate of decrease was enhanced by elicitor treatment, and was dependent on the species of avenanthramides, with avenanthramide B decreasing most rapidly. The rates of biosynthesis and metabolism of avenanthramides A and B were measured using a model of isotope-labeling dynamics. Avenanthramide B was found to be more actively biosynthesized and metabolized than Avenanthramide A. Radiolabeled avenanthramide B was incorporated into the cell wall fraction and 99% of incorporated activity was released by alkaline treatment. Gel filtration indicated that high-molecular-weight compounds derived from avenanthramide B were released by alkaline treatment. The decrease in stable-isotope-labeled avenanthramides was suppressed by catalase, salicylhydroxamic acid, and sodium ascorbate, suggesting the involvement of peroxidase in the metabolism. Consistent with this, peroxidase activity that accepts avenanthramide B as a substrate was induced in apoplastic fractions by elicitor treatment. The appearance of multiple basic isoperoxidases was observed by activity staining with 3-amino-9-ethylcarbazole coupled with isoelectric focusing of proteins from elicitor-treated leaves. These findings suggest that accumulated avenanthramides are further metabolized in apoplasts in oat leaves by inducible isoperoxidases.
Induction of anthranilate synthase activity by elicitors in oats.[Pubmed:11926523]
Z Naturforsch C J Biosci. 2002 Jan-Feb;57(1-2):121-8.
Oat phytoalexins, avenanthramides, are a series of substituted hydroxycinnamic acid amides with anthranilate. The anthranilate in avenanthramides is biosynthesized by anthranilate synthase (AS, EC 4.1.3.27). Induction of anthranilate synthase activity was investigated in oat leaves treated with oligo-N-acetylchitooligosaccharide elicitors. AS activity increased transiently, peaking 6 h after the elicitation. The induction of activity was dependent on the concentration and the degree of polymerization of the oligo-N-acetylchitooligosaccharide elicitor. These findings indicate that the induction is part of a concerted biochemical change required for avenanthramide production. The elicitor-inducible AS activity was strongly inhibited by L-tryptophan and its analogues including 5-methyl-DL-tryptophan, and 5- and 6-fluoro-DL-tryptophan, while the activity was not affected by D-tryptophan. The accumulation of Avenanthramide A was also inhibited by treatment of elicited leaves with these AS inhibitors, indicating that a feedback-sensitive AS is responsible for the avenanthramide production. In elicited leaves, the content of free anthranilate remained at a steady, low level during avenanthramide production. Moreover, administration of anthranilate to elicited oat leaves resulted in an enhanced Avenanthramide Accumulation. AS may play a role as a rate-limiting enzyme in the biosynthesis of avenanthramides.
Occurrence of avenanthramides and hydroxycinnamoyl-CoA:hydroxyanthranilate N-hydroxycinnamoyltransferase activity in oat seeds.[Pubmed:10739096]
Z Naturforsch C J Biosci. 2000 Jan-Feb;55(1-2):30-6.
Oat phytoalexins, avenanthramides, occur as constitutive components in seeds. The amounts of each avenanthramide were analyzed. The composition of avenanthramides in dry seeds was different from that in elicitor-treated leaves. In seeds, avenanthramide C was most abundant with an amount two times larger than that of Avenanthramide A or B. On the other hand, Avenanthramide A was the major component in elicitor-treated leaves. The total amount of avenanthramides in seeds increased 2.5 times during imbibition for 48 h although the composition did not change. The hydroxycinnamoyl-CoA:hydroxyanthranilate N-hydroxycinnamoyltransferase (HHT, EC 2.3.1.-) activity, which is responsible for the final condensation step in the avenanthramide biosynthesis, was detected in dry seeds. The activity was localized in endosperm and scutellum, and slightly increased during 48-h imbibition. The enzyme was partially purified by anion exchange chromatography from both dry seeds and elicitor-treated leaves The activity was separated into two peaks by chromatography, indicating that HHT consists of at least two isoforms. The substrate specificities of HHT isoforms from seeds were different from each other.


