URB597FAAH inhibitor,potent and selective CAS# 546141-08-6 |
- MK-4827 tosylate
Catalog No.:BCC4174
CAS No.:1038915-73-9
- MK-4827 Racemate
Catalog No.:BCC5179
CAS No.:1038915-75-1
- BYK 204165
Catalog No.:BCC2449
CAS No.:1104546-89-5
- EB 47
Catalog No.:BCC2452
CAS No.:1190332-25-2
- BMN-673 8R,9S
Catalog No.:BCC1422
CAS No.:1207456-00-5
- A-966492
Catalog No.:BCC2211
CAS No.:934162-61-5
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 546141-08-6 | SDF | Download SDF |
| PubChem ID | 1383884 | Appearance | Powder |
| Formula | C20H22N2O3 | M.Wt | 338.4 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | KDS-4103 | ||
| Solubility | DMSO : ≥ 100 mg/mL (295.51 mM) H2O : < 0.1 mg/mL (insoluble) *"≥" means soluble, but saturation unknown. | ||
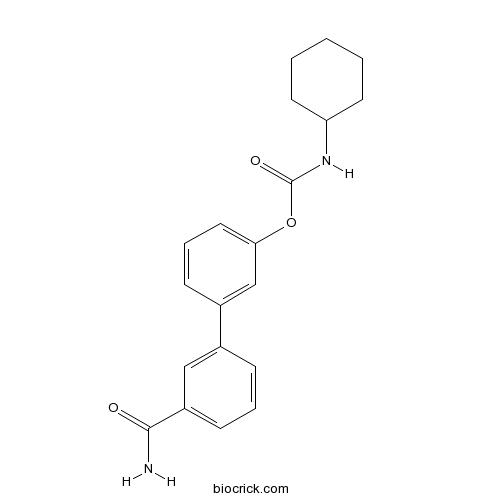
| Chemical Name | [3-(3-carbamoylphenyl)phenyl] N-cyclohexylcarbamate | ||
| SMILES | C1CCC(CC1)NC(=O)OC2=CC=CC(=C2)C3=CC(=CC=C3)C(=O)N | ||
| Standard InChIKey | ROFVXGGUISEHAM-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H22N2O3/c21-19(23)16-8-4-6-14(12-16)15-7-5-11-18(13-15)25-20(24)22-17-9-2-1-3-10-17/h4-8,11-13,17H,1-3,9-10H2,(H2,21,23)(H,22,24) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Potent and selective fatty acid amide hydrolase (FAAH) inhibitor (IC50 values are 3 and 5 nM in human liver and rat brain, respectively). Exhibits no significant inhibitory activity against a variety of receptors, ion channels and enzymes, including human cannabinoid receptors and rat monoacylglycerol lipase. Displays antiallodynic and antihyperalgesic activity in an inflammatory pain model. |

URB597 Dilution Calculator

URB597 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.9551 mL | 14.7754 mL | 29.5508 mL | 59.1017 mL | 73.8771 mL |
| 5 mM | 0.591 mL | 2.9551 mL | 5.9102 mL | 11.8203 mL | 14.7754 mL |
| 10 mM | 0.2955 mL | 1.4775 mL | 2.9551 mL | 5.9102 mL | 7.3877 mL |
| 50 mM | 0.0591 mL | 0.2955 mL | 0.591 mL | 1.182 mL | 1.4775 mL |
| 100 mM | 0.0296 mL | 0.1478 mL | 0.2955 mL | 0.591 mL | 0.7388 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
URB597, also known as KDS-4103, is a potent and selective inhibitor of fatty-acid amide hydrolase (FAAH), an enzyme catalyzing the intracellular hydrolysis of the endocannabinoid anandamide, with values of 50% inhibition concentration IC50 of 5 nM and 3 nM in rat brain membranes and human liver microsomes respectively. Study results have shown that, through intraperitoneal (i.p.) administration, URB597 potently inhibits FAAH in rat brain with a median inhibitory dose ID50 of 0.15 mh/kg. However, URB597 fails to show significant inhibition against a few other cannabinoid-related targets (such as cannabinoid receptors and anandamide transport) as well as a broad range of receptors, ion channels, transporters and enymes.
Reference
Daniele Piomelli, Giorgio Tarzia, Andrea Duranti, Andrea Tontini, Marco Mor, Timothy R. Compton, Oliver Dasse, Edward P. Monaghan, Jeff A. Parrot and David Putman. Pharmacological profile of the selective FAAH inhibitor KDS-4103 (URB597). CNS Drug Review 2006; 12(1): 21-38
- Boc-Lys(2-Cl-Z)-OH
Catalog No.:BCC3416
CAS No.:54613-99-9
- Columbin
Catalog No.:BCN2622
CAS No.:546-97-4
- α-Thujone
Catalog No.:BCC8271
CAS No.:546-80-5
- Alantolactone
Catalog No.:BCN1033
CAS No.:546-43-0
- Conessine
Catalog No.:BCC7352
CAS No.:546-06-5
- Doxercalciferol
Catalog No.:BCC4902
CAS No.:54573-75-0
- Quercetin-3-O-glucose-6'-acetate
Catalog No.:BCN6545
CAS No.:54542-51-7
- AMG 9810
Catalog No.:BCC7329
CAS No.:545395-94-6
- QNZ (EVP4593)
Catalog No.:BCC2249
CAS No.:545380-34-5
- Nicardipine HCl
Catalog No.:BCC4685
CAS No.:54527-84-3
- Methyl protogracillin
Catalog No.:BCN8177
CAS No.:54522-53-1
- Methyl protodioscin
Catalog No.:BCN6342
CAS No.:54522-52-0
- Diethyl 2-acetamido-2-phenethylmalonate
Catalog No.:BCC8940
CAS No.:5463-92-3
- Boc-Hyp(Bzl)-OH.DCHA
Catalog No.:BCC3253
CAS No.:54631-81-1
- 2-Amino-4-methoxybenzothiazole
Catalog No.:BCC8532
CAS No.:5464-79-9
- ML 204
Catalog No.:BCC6272
CAS No.:5465-86-1
- 1-O-Methyljatamanin D
Catalog No.:BCN6671
CAS No.:54656-47-2
- 2-(Acetylamino)-3-phenyl-2-propenoic acid
Catalog No.:BCN1420
CAS No.:5469-45-4
- 4'-Benzyloxyacetophenone
Catalog No.:BCC8698
CAS No.:54696-05-8
- 8-Hydroxy-7-iodo-5-quinolinesulfonic acid
Catalog No.:BCC8788
CAS No.:547-91-1
- Dodecanoic acid ingenol ester
Catalog No.:BCN8291
CAS No.:54706-70-6
- 20-Deoxyingenol
Catalog No.:BCN3770
CAS No.:54706-99-9
- 4-(4-Hydroxyphenyl)-2-butanone
Catalog No.:BCN6797
CAS No.:5471-51-2
- Fluvoxamine
Catalog No.:BCC4214
CAS No.:54739-18-3
Revealing the role of the endocannabinoid system modulators, SR141716A, URB597 and VDM-11, in sleep homeostasis.[Pubmed:27746343]
Neuroscience. 2016 Dec 17;339:433-449.
The endocannabinoid system comprises receptors (CB1 and CB2 cannabinoid receptors), enzymes (Fatty Acid Amide Hydrolase [FAAH], which synthesizes the endocannabinoid anandamide), as well as the anandamide membrane transporter (AMT). Importantly, previous experiments have demonstrated that the endocannabinoid system modulates multiple neurobiological functions, including sleep. For instance, SR141716A (the CB1 cannabinoid receptor antagonist) as well as URB597 (the FAAH inhibitor) increase waking in rats whereas VDM-11 (the blocker of the AMT) enhances sleep in rodents. However, no further evidence is available regarding the neurobiological role of the endocannabinoid system in the homeostatic control of sleep. Therefore, the aim of the current experiment was to test if SR141716A, URB597 or VDM-11 would modulate the sleep rebound after sleep deprivation. Thus, these compounds were systemically injected (5, 10, 20mg/kg; ip; separately each one) into rats after prolonged waking. We found that SR141716A and URB597 blocked in dose-dependent fashion the sleep rebound whereas animals treated with VDM-11 displayed sleep rebound during the recovery period. Complementary, injection after sleep deprivation of either SR141716A or URB597 enhanced dose-dependently the extracellular levels of dopamine (DA), norepinephrine (NE), epinephrine (EP), serotonin (5-HT), as well as adenosine (AD) while VDM-11 caused a decline in contents of these molecules. These findings suggest that SR141716A or URB597 behave as a potent stimulants since they suppressed the sleep recovery period after prolonged waking. It can be concluded that elements of the endocannabinoid system, such as the CB1 cannabinoid receptor, FAAH and AMT, modulate the sleep homeostasis after prolonged waking.
URB597 and the Cannabinoid WIN55,212-2 Reduce Behavioral and Neurochemical Deficits Induced by MPTP in Mice: Possible Role of Redox Modulation and NMDA Receptors.[Pubmed:28092019]
Neurotox Res. 2017 May;31(4):532-544.
Several physiological events in the brain are regulated by the endocannabinoid system (ECS). While synthetic cannabinoid receptor (CBr) agonists such as WIN55,212-2 act directly on CBr, agents like URB597, a fatty acid amide hydrolase (FAAH) inhibitor, induce a more "physiological" activation of CBr by increasing the endogenous levels of the endocannabinoid anandamide (AEA). Herein, we compared the pre- and post-treatment efficacy of URB597 and WIN55,212-2 on different endpoints evaluated in the toxic model produced by the mitochondrial toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in mice. MPTP (40 mg/kg, s.c., single injection) decreased locomotor activity, depleted the striatal and nigral levels of dopamine (DA), augmented the levels of lipid peroxidation and protein carbonylation in both regions, decreased the striatal protein levels of tyrosine hydroxylase, and increased the striatal protein content of the subunit 1 (NR1) of the N-methyl-D-aspartate receptor (NMDAr). Both URB597 (0.3 mg/kg, i.p., once a day) and WIN55,212-2 (10 mug/kg, i.p., twice a day), administered for five consecutive days, either before or after the MPTP injection, prevented the alterations elicited by MPTP and downregulated NMDAr. Our results support a modulatory role of the ECS on the toxic profile exerted by MPTP in mice via the stimulation of antioxidant activity and the induction of NMDAr downregulation and hypofunction, and favor the stimulation of CBr as an effective experimental therapeutic strategy.
URB597 improves cognitive impairment induced by chronic cerebral hypoperfusion by inhibiting mTOR-dependent autophagy.[Pubmed:28042028]
Neuroscience. 2017 Mar 6;344:293-304.
Chronic cerebral hypoperfusion (CCH) is associated with various ischemic cerebrovascular diseases that are characterized by cognitive impairment. The role of autophagy in cognitive dysfunction under conditions of CCH is poorly understood. To address this issue, the present study investigated the effect of the fatty acid amide hydrolase (FAAH) inhibitor URB597 on autophagy and cognition in a CCH model as well as the underlying mechanisms. Cognitive function was evaluated with the Morris water maze and by assessing long-term potentiation (LTP). The expression of autophagy-related proteins and mammalian target of rapamycin (mTOR) signaling pathway components was evaluated by immunofluorescence and western blot analyses, and ultrastructural changes were examined by transmission electron microscopy (EM). URB597 improved cognitive impairment by inhibiting CCH-induced autophagy, which was associated with mTOR signaling. Moreover, the ultrastructural deterioration resulting from CCH was improved by chronic treatment with URB597. These findings indicate that URB597 modulates autophagy in an mTOR-dependent manner, and mitigates neuronal damage and cognitive deterioration caused by CCH.
URB597 reduces biochemical, behavioral and morphological alterations in two neurotoxic models in rats.[Pubmed:28157650]
Biomed Pharmacother. 2017 Apr;88:745-753.
BACKGROUND: URB597 is a compound largely linked to the inhibition of fatty acid amide hydrolase (FAAH), an enzyme responsible for the metabolic degradation of the endocannabinoid anandamide (AEA). Despite this pharmacological property accounts for its modulatory profile demonstrated in some neurotoxic paradigms, the possible protective properties of this agent have been poorly investigated, and deserve exploration in different neurotoxic models. In this study, we explored the effects of URB597 on oxidative damage to lipids and other major endpoints of toxicity in two neurotoxic models in vivo in rats (the first one produced by the mitochondrial neurotoxin 3-nitropropionic acid [3-NP], and the other generated by the striatal injection of the pro-oxidant toxin 6-hydroxidopamine [6-OHDA]) in order to provide further supporting evidence of its modulatory profile. METHODS: Male Wistar adult rats were treated for 5 or 7 consecutive days with URB597 (0.3mg/kg, i.p.) and simultaneously exposed to three injections of 3-NP (30mg/kg, i.p.) or a single intrastriatal infusion of 6-OHDA (0.02mg/2mul), respectively. Twenty four hours after all treatments were administered, lipid peroxidation was measured in the striatum of 3-NP-treated rats, and in the midbrain of 6-OHDA-treated rats. Motor skills and histological assessment in the striatum were also evaluated in 3-NP-treated rats 6 and 7days after the last drug administration, respectively; whereas apomorphine-induced circling behavior and tyrosine hydroxylase immunolocalization in the striatum and substantia nigra were investigated 21 and 22days after the last drug infusion, respectively. RESULTS: URB597 prevented the oxidative damage to lipids induced by 3-NP in the striatum, and this effect could account for the attenuation of motor deficits in this model. Attenuation of motor disturbances induced by URB597 in both models was associated with the morphological preservation of the striatum in the 3-NP model and the partial preservation of tyrosine hydroxylase in the 6-OHDA model in the SNpc and striatum. CONCLUSION: The modulatory actions exerted by URB597 in both toxic models support its potential against toxic conditions implying motor and neurochemical alterations linked to energy depletion, excitotoxicity and oxidative stress. Although most of these effects could be attributable to its action on FAAH and further AEA accumulation, in light of our present findings other properties are suggested.
Actions of the FAAH inhibitor URB597 in neuropathic and inflammatory chronic pain models.[Pubmed:16331291]
Br J Pharmacol. 2006 Feb;147(3):281-8.
While cannabinoid receptor agonists have analgesic activity in chronic pain states, they produce a spectrum of central CB(1) receptor-mediated motor and psychotropic side effects. The actions of endocannabinoids, such as anandamide are terminated by removal from the extracellular space, then subsequent enzymatic degradation by fatty-acid amide hydrolase (FAAH). In the present study, we compared the effect of a selective FAAH inhibitor, URB597, to that of a pan-cannabinoid receptor agonist HU210 in rat models of chronic inflammatory and neuropathic pain. Systemic administration of URB597 (0.3 mg kg(-1)) and HU210 (0.03 mg kg(-1)) both reduced the mechanical allodynia and thermal hyperalgesia in the CFA model of inflammatory pain. In contrast, HU210, but not URB597, reduced mechanical allodynia in the partial sciatic nerve-ligation model of neuropathic pain. HU210, but not URB597, produced a reduction in motor performance in unoperated rats. The effects of URB597 in the CFA model were dose dependent and were reduced by coadministration with the cannabinoid CB1 antagonist AM251 (1 mg kg(-1)), or the CB2 and SR144528 (1 mg kg(-1)). Coadministration with AM251 plus SR144528 completely reversed the effects of URB597. These findings suggest that the FAAH inhibitor URB597 produces cannabinoid CB1 and CB2 receptor-mediated analgesia in inflammatory pain states, without causing the undesirable side effects associated with cannabinoid receptor activation.
Pharmacological profile of the selective FAAH inhibitor KDS-4103 (URB597).[Pubmed:16834756]
CNS Drug Rev. 2006 Spring;12(1):21-38.
In the present article, we review the pharmacological properties of KDS-4103 (URB597), a highly potent and selective inhibitor of the enzyme fatty-acid amide hydrolase (FAAH), which catalyzes the intracellular hydrolysis of the endocannabinoid anandamide. In vitro, KDS-4103 inhibits FAAH activity with median inhibitory concentrations (IC(50)) of 5 nM in rat brain membranes and 3 nM in human liver microsomes. In vivo, KDS-4103 inhibits rat brain FAAH activity after intraperitoneal (i.p.) administration with a median inhibitory dose (ID(50)) of 0.15 mg/kg. The compound does not significantly interact with other cannabinoid-related targets, including cannabinoid receptors and anandamide transport, or with a broad panel of receptors, ion channels, transporters and enzymes. By i.p. administration to rats and mice KDS-4103 elicits significant, anxiolytic-like, antidepressant-like and analgesic effects, which are prevented by treatment with CB1 receptor antagonists. By contrast, at doses that significantly inhibit FAAH activity and substantially raise brain anandamide levels, KDS-4103 does not evoke classical cannabinoid-like effects (e.g., catalepsy, hypothermia, hyperphagia), does not cause place preference, and does not produce generalization to the discriminative effects of the active ingredient of cannabis, Delta9-tetrahydrocannabinol (Delta9-THC). These findings suggest that KDS-4103 acts by enhancing the tonic actions of anandamide on a subset of CB(1) receptors, which may normally be engaged in controlling emotions and pain. KDS-4103 is orally available in rats and cynomolgus monkeys. Sub-chronic repeated dose studies (1,500 mg/kg, per os) in these two species have not demonstrated systemic toxicity. Likewise, no toxicity was noted in bacterial cytotoxicity tests in vitro and in the Ames test. Furthermore, no deficits were observed in rats on the rotarod test after acute i.p. treatment with KDS-4103 at doses up to 5 mg/kg or in a functional observation battery after oral doses up to 1,500 mg/kg. The results suggest that KDS-4103 will offer a novel approach with a favorable therapeutic window for the treatment of anxiety, depression and pain.
An endocannabinoid mechanism for stress-induced analgesia.[Pubmed:15973410]
Nature. 2005 Jun 23;435(7045):1108-12.
Acute stress suppresses pain by activating brain pathways that engage opioid or non-opioid mechanisms. Here we show that an opioid-independent form of this phenomenon, termed stress-induced analgesia, is mediated by the release of endogenous marijuana-like (cannabinoid) compounds in the brain. Blockade of cannabinoid CB(1) receptors in the periaqueductal grey matter of the midbrain prevents non-opioid stress-induced analgesia. In this region, stress elicits the rapid formation of two endogenous cannabinoids, the lipids 2-arachidonoylglycerol (2-AG) and anandamide. A newly developed inhibitor of the 2-AG-deactivating enzyme, monoacylglycerol lipase, selectively increases 2-AG concentrations and, when injected into the periaqueductal grey matter, enhances stress-induced analgesia in a CB1-dependent manner. Inhibitors of the anandamide-deactivating enzyme fatty-acid amide hydrolase, which selectively elevate anandamide concentrations, exert similar effects. Our results indicate that the coordinated release of 2-AG and anandamide in the periaqueductal grey matter might mediate opioid-independent stress-induced analgesia. These studies also identify monoacylglycerol lipase as a previously unrecognized therapeutic target.


