BYK 204165PARP-1 inhibitor,potent and selective CAS# 1104546-89-5 |
- MK-4827
Catalog No.:BCC1761
CAS No.:1038915-60-4
- BMN-673 8R,9S
Catalog No.:BCC1422
CAS No.:1207456-00-5
- XAV-939
Catalog No.:BCC1120
CAS No.:284028-89-3
- PJ34
Catalog No.:BCC1865
CAS No.:344458-19-1
- ABT-888 (Veliparib)
Catalog No.:BCC1267
CAS No.:912444-00-9
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1104546-89-5 | SDF | Download SDF |
| PubChem ID | 25113764 | Appearance | Powder |
| Formula | C15H12N2O2 | M.Wt | 252.27 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 125 mg/mL (495.50 mM; Need ultrasonic) | ||
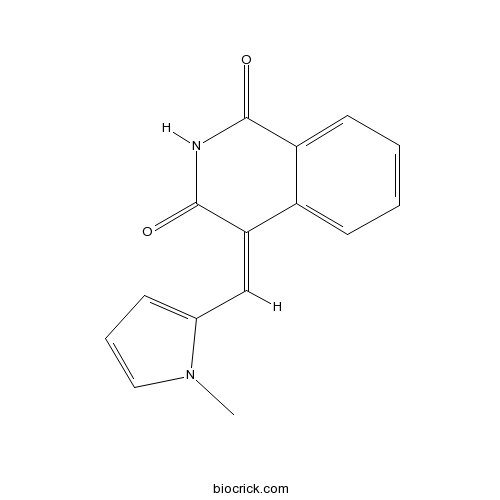
| Chemical Name | (4Z)-4-[(1-methylpyrrol-2-yl)methylidene]isoquinoline-1,3-dione | ||
| SMILES | CN1C=CC=C1C=C2C3=CC=CC=C3C(=O)NC2=O | ||
| Standard InChIKey | BTYSIDSTHDDAJW-LCYFTJDESA-N | ||
| Standard InChI | InChI=1S/C15H12N2O2/c1-17-8-4-5-10(17)9-13-11-6-2-3-7-12(11)14(18)16-15(13)19/h2-9H,1H3,(H,16,18,19)/b13-9- | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Potent and selective poly(ADP-ribose) polymerase (PARP)-1 inhibitor (pIC50 values are 5.38 and 7.35 for PARP-2 and PARP-1 respectively). |

BYK 204165 Dilution Calculator

BYK 204165 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.964 mL | 19.82 mL | 39.6401 mL | 79.2801 mL | 99.1002 mL |
| 5 mM | 0.7928 mL | 3.964 mL | 7.928 mL | 15.856 mL | 19.82 mL |
| 10 mM | 0.3964 mL | 1.982 mL | 3.964 mL | 7.928 mL | 9.91 mL |
| 50 mM | 0.0793 mL | 0.3964 mL | 0.7928 mL | 1.5856 mL | 1.982 mL |
| 100 mM | 0.0396 mL | 0.1982 mL | 0.3964 mL | 0.7928 mL | 0.991 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
BYK 204165 is a potent and selective poly(ADP-ribose) polymerase (PARP)-1 inhibitor with pIC50 values of 5.38 and 7.35 for PARP-2 and PARP-1 respectively [1].
PARP1 is a nuclear enzyme activated by strand breaks in DNA and involved in the regulation of various cell processes such as DNA repair, apoptosis, organ dysfunction or necrosis [1].
BYK 204165 (PARP inhibitor XIV) is a cell-permeable isoquinolinedione compound that potently and selectively inhibits PARP1. BYK 204165’s potency is similar or higher than most current standard PARP-1 inhibitors (just slightly lower than PJ34), and it is 100-fold selective for PARP-1 over PARP-2 [1].
BYK 204165 was not tested in vivo because of BYK its poor water solubility.
References:
[1]. Eltze T, Boer R, Wagner T, et al. Imidazoquinolinone, imidazopyridine, and isoquinolindione derivatives as novel and potent inhibitors of the poly(ADP-ribose) polymerase (PARP): a comparison with standard PARP inhibitors. Mol Pharmacol, 2008, 74(6): 1587-98.
- ML-7 hydrochloride
Catalog No.:BCC1770
CAS No.:110448-33-4
- Dolastatin 10
Catalog No.:BCC4056
CAS No.:110417-88-4
- Gelsemiol
Catalog No.:BCN5992
CAS No.:110414-77-2
- Higenamine HCl
Catalog No.:BCN2831
CAS No.:11041-94-4
- Meclizine hydrochloride
Catalog No.:BCC9017
CAS No.:1104-22-9
- 2,3-Dihydroheveaflavone
Catalog No.:BCN4019
CAS No.:110382-42-8
- 3-O-Methyltagitinin F
Catalog No.:BCN5991
CAS No.:110382-37-1
- Ch 55
Catalog No.:BCC7241
CAS No.:110368-33-7
- S 32826
Catalog No.:BCC7678
CAS No.:1103672-43-0
- Bavisant dihydrochloride hydrate
Catalog No.:BCC1404
CAS No.:1103522-80-0
- CGS 19755
Catalog No.:BCC6986
CAS No.:110347-85-8
- FD-838
Catalog No.:BCN6396
CAS No.:110341-78-1
- Crotastriatine
Catalog No.:BCN2101
CAS No.:11051-94-8
- 6,11-Di-O-acetylalbrassitriol
Catalog No.:BCN7273
CAS No.:110538-20-0
- Scutebarbatine F
Catalog No.:BCN5377
CAS No.:910099-78-4
- Albrassitriol
Catalog No.:BCN7274
CAS No.:110557-39-6
- Salermide
Catalog No.:BCC7867
CAS No.:1105698-15-4
- Laminin (925-933)
Catalog No.:BCC1015
CAS No.:110590-60-8
- human Insulin expressed in yeast
Catalog No.:BCC7689
CAS No.:11061-68-0
- Epimedin A
Catalog No.:BCN1038
CAS No.:110623-72-8
- Epimedin B
Catalog No.:BCN1039
CAS No.:110623-73-9
- Epimedin C
Catalog No.:BCN1040
CAS No.:110642-44-9
- beta-Escin
Catalog No.:BCC8172
CAS No.:11072-93-8
- Asebotin
Catalog No.:BCN7233
CAS No.:11075-15-3
Ciclesonide ( Byk Gulden).[Pubmed:12054077]
Curr Opin Investig Drugs. 2002 Jan;3(1):78-83.
Ciclesonide, a non-halogenated inhaled corticosteroid with anti-inflammatory activity, is under development by Byk Gulden, Aventis and Teijin as a potential treatment for asthma [213439]. It was also being developed by Byk Gulden for chronic obstructive pulmonary disease (COPD), but no development had been reported for this indication since 1999; however, Teijin was carrying out clinical trials in this indication at the end of 2000. During 2000, Byk Gulden was carrying out phase III trials in the US and Europe and in March 2001, results were expected in the third quarter of 2001 [312399], [383726], [423659]. Two inhalant formulations (multidose powder and propellant filled) and a nasal formulation of ciclesonide are being developed by Byk Gulden for the treatment of asthma and seasonal allergic rhinitis, respectively [337147]. The compound is formulated for once-daily dosing and demonstrated good efficacy without corticosteroid-associated systemic side effects [409257]. In January 2001, Byk Gulden expected launch of a CFC-free multidose inhaler formulation in 2003 [395596]; in March 1999, launch of a nasal formulation was expected in 2004 and a multidose powder inhaler in 2005 [337147]. By September 2001, the compound was in phase III trials in the US for asthma, with a potential US launch anticipated by Aventis in 2004 [423465]. In November 2001, Aventis expected to submit an NDA to the FDA in 2003 [428057]. Teijin, which has a development and licensing agreement with Byk Gulden for the treatment of asthma and COPD in Japan, commenced phase I trials of ciclesonide in Japan in spring 1999, had completed these during 2000, and began phase II trials by September of that year [383726]. An NDA is expected to befiled in Japan in 2003. In October 2000 and April 2001, Merrill Lynch predicted peak sales of Euro400 million in 2007, with sales of Euro5 million in 2002, rising to Euro150 million in 2004 [395562], [420574]. Deutsche Bank predicted in August 2001, that sales of the product would reach Euro70 million in 2004, rising to Euro150 million in 2005 [420814].
Roflumilast: APTA 2217, B9302-107, BY 217, BYK 20869.[Pubmed:15139782]
Drugs R D. 2004;5(3):176-81.
Roflumilast [APTA 2217, B9302-107, BY 217, BYK 20869] is a selective phosphodiesterase IV inhibitor. It is being developed by Altana Pharma (formerly Byk Gulden), a subsidiary of Altana Group, as an orally administered therapy for asthma, chronic obstructive pulmonary disease (COPD), allergic rhinitis and psoriasis. The drug is awaiting regulatory approval in Europe for the treatment of asthma and COPD. Byk Gulden has stated that roflumilast relieves asthma symptoms through both an anti-inflammatory effect and a muscle relaxant effect. Roflumilast has potential as first-line long-term therapy in mild-to-moderate COPD and as additive long-term therapy in moderate-to-severe COPD. Altana has stated that roflumilast is to be marketed under the brand name Daxas. Altana Group and Pharmacia Corporation (now Pfizer) signed an agreement on 22 April 2002 to collaborate on the development and commercialisation of roflumilast for the treatment of respiratory disorders, including asthma and COPD. The companies will jointly develop the drug for the US, Europe and other markets. Pharmacia will co-ordinate development in the US and Altana will co-ordinate development in Europe. After approval of the drug, Pharmacia and Altana will jointly launch and promote roflumilast in the US, Europe and elsewhere. Altana will receive an upfront payment and additional milestone payments. Altana additionally has the option to co-promote Pharmacia products in the US and elsewhere. On 16 April 2003, Pharmacia Corporation was acquired by, and merged into, Pfizer. In November 2002, Altana and Tanabe Seiyaku signed an agreement to collaborate on the development and commercialisation of roflumilast for the treatment of respiratory diseases, including asthma and COPD. Tanabe Seiyaku and Altana will develop roflumilast for asthma and COPD in Japan, and will jointly launch and co-promote roflumilast in Japan following regulatory approval. Roflumilast has been in multinational phase III clinical studies in Europe for the treatment of asthma and COPD. In September 2003, Altana announced the completion of a phase III trial in COPD in more than 1400 patients; the trial showed positive results. In the US, roflumilast is in phase III trials for the treatment of asthma and phase II trials for the treatment of COPD. Phase I clinical trials of roflumilast were begun in Japan by Tanabe Seiyaku in the fourth quarter of 2003. Altana has stated that roflumilast has shown significant superiority over placebo in the treatment of asthma in phase II trials. The efficacy of the drug appears to be comparable to low-dose inhaled corticosteroids in the treatment of asthma and at least equal to inhaled corticosteroids in the treatment of COPD. Altana Group presented data from phase II trials in 516 patients with COPD at an analyst meeting [August 2001, Bad Homburg, Germany] that showed that roflumilast 500 microg/day significantly improved FEV(1) at 24 weeks compared with placebo. In March 2004, Altana Pharma presented pharmacokinetic data from a phase I trial of roflumilast at the 60th Annual Meeting of the American Academy of Allergy, Asthma and Immunology (AAAAI-2004) [San Francisco, CA, USA]. This open-label, randomised, two-period crossover study investigated the pharmacokinetics of oral roflumilast and its active metabolite, roflumilast N-oxide, among 12 healthy male subjects. Participants received single doses of oral roflumilast 500 microg and intravenous (i.v) roflumilast 150 microg as a 15-min short-term infusion. In November 2002, the combined global market for asthma and COPD products was estimated to be worth >11 billion US dollars. In Japan, products in this market segment reached sales of approximately 1.5 billion US dollars in 2001. Roflumilast has patent protection in Europe and Japan until 2014 and in the US until 2015. The Financial Times in April 2002 claimed that roflumilast is an 'important' product for Altana, due to be listed on the New York Stock Exchange later in the same month. The Altana chairman confirmed that the company had been in talks with Pfizer, Bristol-Myers Squibb and Novartis with regard to future development and commercialisation of roflumilast. In September 2002, Dow Jones Newswires stated that Altana is to file for European approval of roflumilast 1 year later than initially was expected; however, this has not changed the company's outlook for the product, which was said to remain at at 1 billion Euros. In August 2001, the Financial Times reported that roflumilast, for the indication of smoker's cough alone, has the potential to reach sales of more than 500 million US dollars a year. A future co-marketing deal for roflumilast in the US was said to be "a key step towards expanding Altana's presence in the US".
Clinical evaluation of the Byk LIA-mat CA125 II assay: discussion of a reference value.[Pubmed:9068593]
Clin Chem. 1997 Mar;43(3):491-7.
The Byk LIA-mat CA125 II assay was compared with the Centocor IRMA CA125 II. Serum samples studied (n = 1012) were obtained from 652 apparently healthy females, 61 pregnant women, and 299 patients with benign and malignant gynecological tumors. The CA125 II assay value at the 95th percentile of the total healthy group was 29 kU/L for the LIA-mat and 32 kU/L for the Centocor assay. For the LIA-mat assay the 95th percentile was 31 kU/L (Centocor 36 kU/L) for the group < 45 years and 21 kU/L (Centocor 25 kU/L) for women > 55 years of age. By using ROC curves we found the optimal pretreatment Byk LIA-mat CA125 II value differentiating between benign and malignant ovarian tumors to be 95 kU/L. Pretreatment CA125 values > 1000 kU/L were detected in serum samples of patients with advanced epithelial ovarian cancer.
Imidazoquinolinone, imidazopyridine, and isoquinolindione derivatives as novel and potent inhibitors of the poly(ADP-ribose) polymerase (PARP): a comparison with standard PARP inhibitors.[Pubmed:18809672]
Mol Pharmacol. 2008 Dec;74(6):1587-98.
We have identified three novel structures for inhibitors of the poly(ADP-ribose) polymerase (PARP), a nuclear enzyme activated by strand breaks in DNA and implicated in DNA repair, apoptosis, organ dysfunction or necrosis. 2-[4-(5-Methyl-1H-imidazol-4-yl)-piperidin-1-yl]-4,5-dihydro-imidazo[4,5,1-i,j]qu inolin-6-one (BYK49187), 2-(4-pyridin-2-yl-phenyl)-4,5-dihydro-imidazo[4,5,1-i,j]quinolin-6-one (BYK236864), 6-chloro-8-hydroxy-2,3-dimethyl-imidazo-[1,2-alpha]-pyridine (BYK20370), and 4-(1-methyl-1H-pyrrol-2-ylmethylene)-4H-isoquinolin-1,3-dione (BYK204165) inhibited cell-free recombinant human PARP-1 with pIC(50) values of 8.36, 7.81, 6.40, and 7.35 (pK(i) 7.97, 7.43, 5.90, and 7.05), and murine PARP-2 with pIC(50) values of 7.50, 7.55, 5.71, and 5.38, respectively. BYK49187, BYK236864, and BYK20370 displayed no selectivity for PARP-1/2, whereas BYK204165 displayed 100-fold selectivity for PARP-1. The IC(50) values for inhibition of poly(ADP-ribose) synthesis in human lung epithelial A549 and cervical carcinoma C4I cells as well in rat cardiac myoblast H9c2 cells after PARP activation by H(2)O(2) were highly significantly correlated with those at cell-free PARP-1 (r(2) = 0.89-0.96, P < 0.001) but less with those at PARP-2 (r(2) = 0.78-0.84, P < 0.01). The infarct size caused by coronary artery occlusion and reperfusion in the anesthetized rat was reduced by 22% (P < 0.05) by treatment with BYK49187 (3 mg/kg i.v. bolus and 3 mg/kg/h i.v. during 2-h reperfusion), whereas the weaker PARP inhibitors, BYK236864 and BYK20370, were not cardioprotective. In conclusion, the imidazoquinolinone BYK49187 is a potent inhibitor of human PARP-1 activity in cell-free and cellular assays in vitro and reduces myocardial infarct size in vivo. The isoquinolindione BYK204165 was found to be 100-fold more selective for PARP-1. Thus, both compounds might be novel and valuable tools for investigating PARP-1-mediated effects.


