ValaciclovirProdrug of aciclovir for herpes virus treatment CAS# 124832-26-4 |
- Daptomycin
Catalog No.:BCC1057
CAS No.:103060-53-3
- Nelarabine
Catalog No.:BCC1072
CAS No.:121032-29-9
- Gemcitabine HCl
Catalog No.:BCC1076
CAS No.:122111-03-9
- Clofarabine
Catalog No.:BCC1078
CAS No.:123318-82-1
- Ifosfamide
Catalog No.:BCC1164
CAS No.:3778-73-2
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 124832-26-4 | SDF | Download SDF |
| PubChem ID | 60773 | Appearance | Powder |
| Formula | C13H20N6O4 | M.Wt | 324.34 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | Valaciclovir | ||
| Solubility | 25℃: DMSO | ||
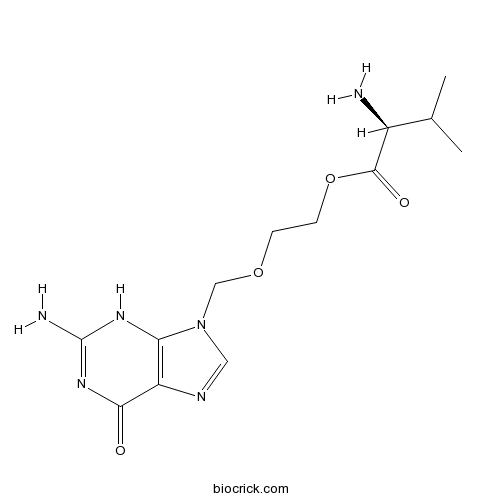
| Chemical Name | 2-[(2-amino-6-oxo-3H-purin-9-yl)methoxy]ethyl (2S)-2-amino-3-methylbutanoate | ||
| SMILES | CC(C)C(C(=O)OCCOCN1C=NC2=C1NC(=NC2=O)N)N | ||
| Standard InChIKey | HDOVUKNUBWVHOX-QMMMGPOBSA-N | ||
| Standard InChI | InChI=1S/C13H20N6O4/c1-7(2)8(14)12(21)23-4-3-22-6-19-5-16-9-10(19)17-13(15)18-11(9)20/h5,7-8H,3-4,6,14H2,1-2H3,(H3,15,17,18,20)/t8-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Valacyclovir is an antiviral drug used in the management of herpes simplex, herpes zoster, and herpes B.
IC50 Value: 2.9 microg/ml (for HSV-1 W)[4].
Target: HSV infection
in vitro: VACV uptake was concentration dependent and saturable with a Michaelis-Menten constant and maximum velocity of 1.64 +/- 0.06 mM and 23.34 +/- 0.36 nmol/mg protein/5 min, respectively. A very similar Km value was obtained in hPEPT1/CHO cells and in rat and rabbit tissues and Caco-2 cells, suggesting that hPEPT1 dominates the intestinal transport properties of VACV in vitro [5].
in vivo: For treatment of a first episode of genital herpes, a large comparative trial has shown that valacyclovir (1 g twice a day) is as effective as acyclovir (200 mg five times a day) when given for 10 days. For treating recurrences, two trials show that valacyclovir is as effective as acyclovir (200 mg five times a day) with a treatment period of 5 days. A daily dose of 1 g of valacyclovir is as effective as 2 g daily. Valacyclovir can be administered once a day[1]. The concentrations of acyclovir in serum and CSF were measured at steady state after 6 days of oral treatment with 1,000 mg of valacyclovir three times a day [2]. EC50 values of PE and AC in 3T3 cells were 0.02 and 0.01 ug/ml, while values in BHK cells were 0.2 and 0.03 ug/ml. Treatment of infected immunosuppressed mice and FA and VA (b.i.d., 5.5 days) reduced the proportion with erythema from 100% to 24% and 38%, and eliminated ear paralysis, ear lesions (vesicles, etc) and death. Virus was absent from ear and brainstem by day 6, but reappeared after discontinuation in mice treated with VA [3].
Clinical trial: Evaluation of Valaciclovir in Patients with Chickenpox. Phage3 References: | |||||

Valaciclovir Dilution Calculator

Valaciclovir Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.0832 mL | 15.4159 mL | 30.8318 mL | 61.6637 mL | 77.0796 mL |
| 5 mM | 0.6166 mL | 3.0832 mL | 6.1664 mL | 12.3327 mL | 15.4159 mL |
| 10 mM | 0.3083 mL | 1.5416 mL | 3.0832 mL | 6.1664 mL | 7.708 mL |
| 50 mM | 0.0617 mL | 0.3083 mL | 0.6166 mL | 1.2333 mL | 1.5416 mL |
| 100 mM | 0.0308 mL | 0.1542 mL | 0.3083 mL | 0.6166 mL | 0.7708 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
IC50 Value: 2.9 microg/ml (for HSV-1 W)[4]. Valacyclovir, the metabolic precursor of , is now approved for treatment and prevention of genital infection with herpes simplex viruses [1]. in vitro: VACV uptake was concentration dependent and saturable with a Michaelis-Menten constant and maximum velocity of 1.64 +/- 0.06 mM and 23.34 +/- 0.36 nmol/mg protein/5 min, respectively. A very similar Km value was obtained in hPEPT1/CHO cells and in rat and rabbit tissues and Caco-2 cells, suggesting that hPEPT1 dominates the intestinal transport properties of VACV in vitro [5]. in vivo: For treatment of a first episode of genital herpes, a large comparative trial has shown that valacyclovir (1 g twice a day) is as effective as acyclovir (200 mg five times a day) when given for 10 days. For treating recurrences, two trials show that valacyclovir is as effective as acyclovir (200 mg five times a day) with a treatment period of 5 days. A daily dose of 1 g of valacyclovir is as effective as 2 g daily. Valacyclovir can be administered once a day[1]. The concentrations of acyclovir in serum and CSF were measured at steady state after 6 days of oral treatment with 1,000 mg of valacyclovir three times a day [2]. EC50 values of PE and AC in 3T3 cells were 0.02 and 0.01 ug/ml, while values in BHK cells were 0.2 and 0.03 ug/ml. Treatment of infected immunosuppressed mice and FA and VA (b.i.d., 5.5 days) reduced the proportion with erythema from 100% to 24% and 38%, and eliminated ear paralysis, ear lesions (vesicles, etc) and death. Virus was absent from ear and brainstem by day 6, but reappeared after discontinuation in mice treated with VA [3]. Clinical trial: Evaluation of Valaciclovir in Patients with Chickenpox. Phage3
- USP7-USP47 inhibitor
Catalog No.:BCC4113
CAS No.:1247825-37-1
- P 22077
Catalog No.:BCC3616
CAS No.:1247819-59-5
- Losartan Potassium (DuP 753)
Catalog No.:BCC1080
CAS No.:124750-99-8
- Quinovic acid 3-O-(6-deoxy-beta-D-glucopyranoside) 28-O-beta-D-glucopyranosyl ester
Catalog No.:BCN1595
CAS No.:124727-10-2
- Meprednisone
Catalog No.:BCC4893
CAS No.:1247-42-3
- Methyl-Dodovisate A
Catalog No.:BCN4719
CAS No.:1246937-33-6
- 5'-Prenylaliarin
Catalog No.:BCN4829
CAS No.:1246926-09-9
- 5,7,4'-Trihydroxy-3,6-dimethoxy-3',5'-diprenylflavone
Catalog No.:BCN1596
CAS No.:1246926-08-8
- VS-5584 (SB2343)
Catalog No.:BCC2047
CAS No.:1246560-33-7
- MPI-0479605
Catalog No.:BCC5347
CAS No.:1246529-32-7
- SR1078
Catalog No.:BCC1963
CAS No.:1246525-60-9
- (-)-Dihydroguaiaretic acid
Catalog No.:BCN8002
CAS No.:124649-78-1
- Valacyclovir hydrochloride
Catalog No.:BCC4051
CAS No.:124832-27-5
- Nadifloxacin
Catalog No.:BCC4804
CAS No.:124858-35-1
- 5-Dehydroxyparatocarpin K
Catalog No.:BCN4017
CAS No.:124858-37-3
- Isoaltholactone
Catalog No.:BCN4826
CAS No.:124868-11-7
- Benzoyl leuco methylene blue
Catalog No.:BCC8862
CAS No.:1249-97-4
- (R)-(+)-Tolterodine
Catalog No.:BCC4054
CAS No.:124937-51-5
- Tolterodine tartrate
Catalog No.:BCC4586
CAS No.:124937-52-6
- Tectoroside
Catalog No.:BCN6672
CAS No.:124960-89-0
- Raddeanoside R8
Catalog No.:BCN6555
CAS No.:124961-61-1
- Ajugacumbin A
Catalog No.:BCN3657
CAS No.:124961-66-6
- Ajugamarin L2
Catalog No.:BCN3662
CAS No.:124961-67-7
- Prednisone 21-acetate
Catalog No.:BCC9128
CAS No.:125-10-0
Prophylactic Efficacy Against Herpes Zoster and Costs Difference Between Acyclovir and Valaciclovir in Hematological Patients.[Pubmed:27566094]
In Vivo. 2016 09-10;30(5):701-5.
BACKGROUND: Immunocompromised hematological patients are at increased risk of herpes zoster (HZ). We examined the efficacy of acyclovir and Valaciclovir in preventing HZ. We also created a simulation to reduce prophylactic medicine costs. PATIENTS AND METHODS: We retrospectively evaluated 573 hematological patients who received chemotherapy, and assessed the difference in the costs between the acyclovir (Zovirax(R)) and Valaciclovir (Valtrex(R)) groups. RESULTS: Forty-four out of the 573 patients (7.7%) developed HZ. Out of them, there were 37 patients (84.1%) who received corticosteroids. Moreover, in total, there were 67 patients receiving acyclovir prophylaxis and 42 patients receiving Valaciclovir prophylaxis, out of which one from each group occurred with HZ. The total 5-year cost of acyclovir and Valaciclovir was yen2,869,917 and yen4,809,952, respectively. Therefore, by changing from Valaciclovir to acyclovir, medical costs could be reduced by 28.3%. Additionally, switching to generic inexpensive acyclovir would possibly reduce them to 15.0%. CONCLUSION: Chemotherapy, including corticosteroids, is associated with a high incidence of HZ. Additionally, there was no prophylactic difference between acyclovir and Valaciclovir. We expect that use of generic acyclovir could reduce prophylaxis costs by 85.0%.
Efficacy of interferon in recurrent valaciclovir-refractory erythema multiforme in a patient not infected with hepatitis C virus.[Pubmed:27334053]
Clin Exp Dermatol. 2016 Aug;41(6):648-50.
Recurrent erythema multiforme (EM) is rare and is most typically related to infections with herpes simplex virus. Prophylactic administration of Valaciclovir is the first-line treatment, but there is no agreement about second-line treatment in cases of ineffectiveness. We present a 31-year-old man who was not infected with hepatitis C virus (HCV), and had a history of severe and recurrent EM, unresponsive to Valaciclovir, colchicine and hydroxychloroquine. The patient noticed that an intermittent flu-like illness seemed to have abrogated an EM flare. Because of this observation, the next EM flares were treated with short courses of interferon, which gave rapid and complete efficacy. Efficacy of interferon in EM has only been reported in two previous patients, in whom the drug was administered to treat HCV infection. Efficiency was attributed to treatment of the underlying HCV infection, which was thought to be the origin of EM in both cases. This is the first case, to our knowledge, reporting a dramatic response to interferon-alfa in a patient who was not infected with HCV.
Valaciclovir to prevent Cytomegalovirus mediated adverse modulation of the immune system in ANCA-associated vasculitis (CANVAS): study protocol for a randomised controlled trial.[Pubmed:27450392]
Trials. 2016 Jul 22;17(1):338.
BACKGROUND: The ANCA-associated vasculitides (AAV) are systemic autoimmune inflammatory disorders characterised by necrotising inflammation affecting small to medium-sized blood vessels. Despite improvements in survival, infection and cardiovascular disease remain leading causes of morbidity and mortality. Considerable evidence suggests that CD4 + CD28null T-cell expansions, predominantly seen in Cytomegalovirus (CMV) seropositive individuals, are associated with systemic dysregulation of immune function leading to a heightened risk of infection and cardiovascular disease. In patients with AAV, CD4 + CD28null expansions are driven by CMV and are associated with an increased risk of infection and mortality. The aim of this study is to explore in detail the ways in which CMV modulates the immune system and to determine whether treatment with Valaciclovir blocks subclinical CMV reactivation in CMV seropositive AAV patients and ameliorates the CMV-induced adverse effects on the immune system. METHODS/DESIGN: CANVAS is a single-centre prospective open-label randomised controlled proof-of-concept trial of 50 adult CMV seropositive patients with stable AAV. Participants will be randomly allocated to receive Valaciclovir orally (2 g QDS or reduced according to renal function) or no additional treatment for 6 months with an additional 6-month follow-up period. The primary outcome is the proportion of patients with CMV reactivation, as assessed by measurable viral load on quantitative blood and urine CMV polymerase chain reaction. The secondary outcomes are safety, change in the proportion of CD4+ CMV-specific T-cell population (defined as CD4 + CD28null cells) and change in soluble markers of inflammation from baseline to 6 months. Further tertiary and exploratory outcomes include persistence of the effect of Valaciclovir on the proportion of CD4 + CD28null cells at 6 months post completion of treatment, change in the immune phenotype of CD4+ T cells and change in blood pressure and arterial stiffness parameters from baseline to 6 months. DISCUSSION: The results of this study will enable larger studies to be conducted to determine whether by controlling subclinical CMV reactivation, we can improve clinical endpoints such as infection and cardiovascular disease. The potential impact of this study is not limited to AAV, as CD4 + CD28null cells have been linked to adverse outcomes in other inflammatory conditions and in the context of an ageing immune system. TRIAL REGISTRATION: ClinicalTrials.gov Identifier NCT01633476 (registered 29 June 2012).


