WilforineCAS# 11088-09-8 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 11088-09-8 | SDF | Download SDF |
| PubChem ID | 73320 | Appearance | White-yellowish powder |
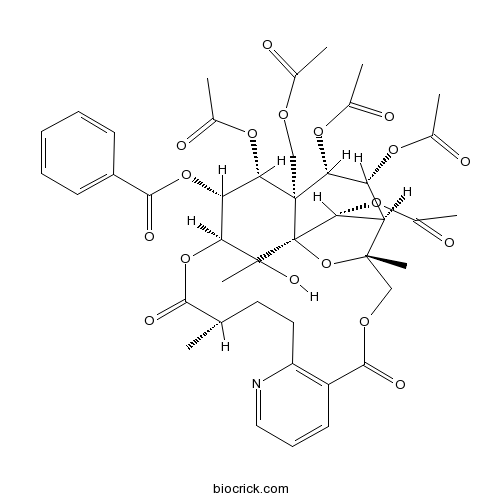
| Formula | C43H49NO18 | M.Wt | 867.9 |
| Type of Compound | Sesquiterpenoids | Storage | Desiccate at -20°C |
| Synonyms | 26-Deoxywilfordine | ||
| Solubility | Soluble in methan | ||
| SMILES | CC1CCC2=C(C=CC=N2)C(=O)OCC3(C4C(C(C5(C(C(C(C(C5(C4OC(=O)C)O3)(C)O)OC1=O)OC(=O)C6=CC=CC=C6)OC(=O)C)COC(=O)C)OC(=O)C)OC(=O)C)C | ||
| Standard InChIKey | ZOCKGJZEUVPPPI-FZILPIMGSA-N | ||
| Standard InChI | InChI=1S/C43H49NO18/c1-21-16-17-29-28(15-12-18-44-29)39(52)55-19-40(7)30-31(56-23(3)46)35(58-25(5)48)42(20-54-22(2)45)36(59-26(6)49)32(60-38(51)27-13-10-9-11-14-27)34(61-37(21)50)41(8,53)43(42,62-40)33(30)57-24(4)47/h9-15,18,21,30-36,53H,16-17,19-20H2,1-8H3/t21-,30+,31+,32-,33+,34-,35+,36-,40-,41?,42+,43-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Wilforine has anti-inflammatory effect, which might be mediated by down-regulation of the expression of inflammatory factors TNF-α, IL-6 and NO. It also has insecticidal activity by inhibiting the Na+-K+-ATPase in the central nervous system. |
| Targets | TNF-α | IL Receptor | NO | ATPase | Sodium channel | Potassium channel |
| In vitro | Establishment of Tripterygium wilfordii Hook. f. Hairy root culture and optimization of its culture conditions for the production of triptolide and wilforine.[Pubmed: 24651642]J Microbiol Biotechnol. 2014 Jun 28;24(6):823-34.
|
| Structure Identification | Biomed Chromatogr. 2015 Jul;29(7):1042-7.Application of a sensitive and specific LC-MS/MS method for determination of wilforine from Tripterygium wilfordii Hook. F. in rat plasma for a bioavailability study.[Pubmed: 25425175]
|

Wilforine Dilution Calculator

Wilforine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.1522 mL | 5.761 mL | 11.5221 mL | 23.0441 mL | 28.8052 mL |
| 5 mM | 0.2304 mL | 1.1522 mL | 2.3044 mL | 4.6088 mL | 5.761 mL |
| 10 mM | 0.1152 mL | 0.5761 mL | 1.1522 mL | 2.3044 mL | 2.8805 mL |
| 50 mM | 0.023 mL | 0.1152 mL | 0.2304 mL | 0.4609 mL | 0.5761 mL |
| 100 mM | 0.0115 mL | 0.0576 mL | 0.1152 mL | 0.2304 mL | 0.2881 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Wilforine is a sesquiterpene pyridine alkaloid; important bioactive compound in T. wilfordii plants, and is effective in treating idiopathic pulmonary fibrosis.
References:
[1]. Zhu C, et al. Establishment of Tripterygium wilfordii Hook. f. Hairy root culture and optimization of its culture conditions for the production of triptolide and wilforine. J Microbiol Biotechnol. 2014 Jun 28;24(6):823-34.
[2]. Miao GP, et al. Elicitation and in situ adsorption enhanced secondary metabolites production of Tripterygium wilfordii Hook. f. adventitious root fragment liquid cultures in shake flask and a modified bubble column bioreactor. Bioprocess Biosyst Eng. 2014
- Entrectinib
Catalog No.:BCC6410
CAS No.:1108743-60-7
- Sparfloxacin
Catalog No.:BCC4848
CAS No.:110871-86-8
- MCOPPB trihydrochloride
Catalog No.:BCC4161
CAS No.:1108147-88-1
- Cinobufotalin
Catalog No.:BCN2283
CAS No.:1108-68-5
- Garcinexanthone A
Catalog No.:BCN5993
CAS No.:1107620-67-6
- Asebotin
Catalog No.:BCN7233
CAS No.:11075-15-3
- beta-Escin
Catalog No.:BCC8172
CAS No.:11072-93-8
- Epimedin C
Catalog No.:BCN1040
CAS No.:110642-44-9
- Epimedin B
Catalog No.:BCN1039
CAS No.:110623-73-9
- Epimedin A
Catalog No.:BCN1038
CAS No.:110623-72-8
- human Insulin expressed in yeast
Catalog No.:BCC7689
CAS No.:11061-68-0
- Laminin (925-933)
Catalog No.:BCC1015
CAS No.:110590-60-8
- [Sar9,Met(O2)11]-Substance P
Catalog No.:BCC6960
CAS No.:110880-55-2
- Tunicamycin
Catalog No.:BCC7699
CAS No.:11089-65-9
- PF-04620110
Catalog No.:BCC2335
CAS No.:1109276-89-2
- Squalene
Catalog No.:BCN5995
CAS No.:111-02-4
- Decanedioic acid
Catalog No.:BCN5996
CAS No.:111-20-6
- Diethanolamine
Catalog No.:BCN1797
CAS No.:111-42-2
- Oleylethanolamide
Catalog No.:BCC7084
CAS No.:111-58-0
- 1-Heptylamine
Catalog No.:BCN1801
CAS No.:111-68-2
- Methyl Laurate
Catalog No.:BCC8211
CAS No.:111-82-0
- Deacetylsalannin
Catalog No.:BCN4733
CAS No.:1110-56-1
- (-)-Dihydroquercetin
Catalog No.:BCN3370
CAS No.:111003-33-9
- Efonidipine hydrochloride monoethanolate
Catalog No.:BCC7767
CAS No.:111011-76-8
Application of a sensitive and specific LC-MS/MS method for determination of wilforine from Tripterygium wilfordii Hook. F. in rat plasma for a bioavailability study.[Pubmed:25425175]
Biomed Chromatogr. 2015 Jul;29(7):1042-7.
A highly selective and specific LC-MS/MS method was developed and validated for the determination of Wilforine in rat plasma. The analyte was separated from plasma matrix by using methyl tertiary butyl ether liquid-liquid extraction with bulleyacinitine A as internal standard (IS). The analysis was carried out on a Sepax GP-Phenyl column using a mixture of methanol and 10 mmol/L ammonium formate buffer solution containing 0.1% formic acid (75:25, v/v) as the mobile phase pumped at a flow rate of 1.0 mL/min. The detection was operated using a triple-quadrupole mass spectrometer in multiple selected reaction monitoring with the parent-to-product quantifier transitions [M + H](+) m/z 867.6 -->206.0 for Wilforine and 664.1 -->584.1 for IS. The main advantage of this method was the high sensitivity (a lower limit of quantification of 0.02 ng/mL) and the small amount of sample (0.1 mL plasma per sample). The method was fully validated to be accurate and precise with a linear range of 0.02-100 ng/mL, and successfully applied to a bioavailability study of Wilforine in rats after intravenous and oral administration. The oral absolute bioavailability of Wilforine in rats was estimated to be 84%.
Establishment of Tripterygium wilfordii Hook. f. Hairy root culture and optimization of its culture conditions for the production of triptolide and wilforine.[Pubmed:24651642]
J Microbiol Biotechnol. 2014 Jun 28;24(6):823-34.
In order to solve the shortage of natural Tripterygium wilfordii Hook. f. plant resource for the production of the important secondary metabolites triptolide and Wilforine, hairy roots were induced from its root calli by Agrobacterium rhizogenes. Induced hairy roots not only could be maintained and grown well in hormone-free half-strength Murashige and Skoog medium but also could produce sufficient amounts of both triptolide and Wilforine. Although hairy roots produced approximately 15% less triptolide than adventitious roots and 10% less Wilforine than naturally grown roots, they could grow fast and could be a suitable system for producing both secondary metabolites compared with other tissues. Addition of 50 micrometer methyl jasmonate (MeJA) could slightly affect hairy root growth, but dramatically stimulated the production of both triptolide and Wilforine, whereas 50 micrometer salicylic acid had no apparent effect on hairy root growth with slightly stimulatory effects on the production of both secondary metabolites. Addition of precursor nicotinic acid, isoleucine, or aspartic acid at the concentration of 500 micrometer had varying effects on hairy root growth, but none of them had stimulatory effects on triptolide production, and only the former two had slightly beneficial effects on Wilforine production. The majority of triptolide produced was secreted into the medium, whereas most of the produced Wilforine was retained inside of hairy roots. Our studies provide a promising way to produce triptolide and Wilforine in T. wilfordii hairy root cultures combined with MeJA treatment.


