ZarzissineCAS# 160568-14-9 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 160568-14-9 | SDF | Download SDF |
| PubChem ID | 6400641 | Appearance | Powder |
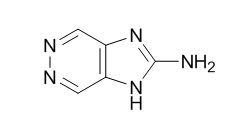
| Formula | C5H5N5 | M.Wt | 135.1 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 1H-imidazo[4,5-d]pyridazin-2-amine | ||
| SMILES | C1=C2C(=CN=N1)N=C(N2)N | ||
| Standard InChIKey | BUYBPEINWQQOFV-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C5H5N5/c6-5-9-3-1-7-8-2-4(3)10-5/h1-2H,(H3,6,9,10) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Zarzissine exhibits cytotoxicity against three human and murine tumor cell lines. |

Zarzissine Dilution Calculator

Zarzissine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 7.4019 mL | 37.0096 mL | 74.0192 mL | 148.0385 mL | 185.0481 mL |
| 5 mM | 1.4804 mL | 7.4019 mL | 14.8038 mL | 29.6077 mL | 37.0096 mL |
| 10 mM | 0.7402 mL | 3.701 mL | 7.4019 mL | 14.8038 mL | 18.5048 mL |
| 50 mM | 0.148 mL | 0.7402 mL | 1.4804 mL | 2.9608 mL | 3.701 mL |
| 100 mM | 0.074 mL | 0.3701 mL | 0.7402 mL | 1.4804 mL | 1.8505 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 5,7,3',4'-Tetrahydroxy-3-methoxy-8-geranylflavone
Catalog No.:BCN6847
CAS No.:1605304-56-0
- BW 723C86 hydrochloride
Catalog No.:BCC6915
CAS No.:160521-72-2
- 12S-hydroxyandrographolide
Catalog No.:BCN4700
CAS No.:869593-50-0
- Bisandrographolide A
Catalog No.:BCN4701
CAS No.:160498-00-0
- Antirhine
Catalog No.:BCN4003
CAS No.:16049-28-8
- THZ2
Catalog No.:BCC3986
CAS No.:1604810-84-5
- THZ1
Catalog No.:BCC4005
CAS No.:1604810-83-4
- 30-Oxopseudotaraxasterol
Catalog No.:BCN7135
CAS No.:160481-71-0
- 3',5,5',7-Tetrahydroxyflavanone
Catalog No.:BCN1710
CAS No.:160436-10-2
- X-NeuNAc
Catalog No.:BCC2063
CAS No.:160369-85-7
- Bisdehydroneotuberostemonine
Catalog No.:BCN7072
CAS No.:160333-27-7
- L-368,899 hydrochloride
Catalog No.:BCC7438
CAS No.:160312-62-9
- DMP 543
Catalog No.:BCC7331
CAS No.:160588-45-4
- Villosin
Catalog No.:BCN1711
CAS No.:160598-92-5
- 1,4,6-Trihydroxy-5-methoxy-7-prenylxanthone
Catalog No.:BCN1548
CAS No.:160623-47-2
- 2,3-Dihydro-3alpha-methoxynimbolide
Catalog No.:BCN7093
CAS No.:1607828-35-2
- Ardisicrenoside B
Catalog No.:BCN8078
CAS No.:160791-12-8
- Indirubin-3'-oxime
Catalog No.:BCC7185
CAS No.:160807-49-8
- AMG319
Catalog No.:BCC6510
CAS No.:1608125-21-8
- H-Thr(Bzl)-ol
Catalog No.:BCC2577
CAS No.:160841-03-2
- Ethyl 2-(3-cyano-4-isobutoxyphenyl)-4-methyl-5-thiazolecarboxylate
Catalog No.:BCC8967
CAS No.:160844-75-7
- Chrysin 8-C-glucoside
Catalog No.:BCN7982
CAS No.:160880-89-7
- Fmoc-Valinol
Catalog No.:BCC2694
CAS No.:160885-98-3
- Hosenkoside F
Catalog No.:BCN2520
CAS No.:160896-45-7
[Chemical constitunents of seeds of Oroxylum indicum].[Pubmed:23672042]
Zhongguo Zhong Yao Za Zhi. 2013 Jan;38(2):204-7.
OBJECTIVE: To study the chemical constituents in the seeds of Oroxylum indicum. METHOD: Twenty compounds were isolated and purified by silica gel, and Sephadex LH-20 column chromatography, and their structures were determined by spectroscopic analysis including NMR and MS. RESULT: Twenty compounds were isolated and identified as oroxin A (1), oroxin B (2), chrysin (3), baicalein (4), quercetin (5), apigenin (6), kaempferol (7), quercetin-3-O-ara-binopyranoside (8), lupeol C9), lup-20 (29)-ene-2alpha,3beta-diol (10), pinosylvin (11), dihydropinosylvin (12), cholest-5-ene-3, 7-diol (13), rengyol (14), isorengyol (15), Zarzissine (16), (E) -pinosylvin-3-O-beta-D-glucopyranoside (17), adenosine (18), sitosterol (19) and daucosterol (20). CONCLUSION: Compounds 11-13 and 15-18 were obtained from the genus Oroxylum for the first time, and except compound 18, the remaining 6 compounds were obtained from the family Bignoniaceae for the first time.
Zarzissine, a new cytotoxic guanidine alkaloid from the Mediterranean sponge Anchinoe paupertas.[Pubmed:7807130]
J Nat Prod. 1994 Oct;57(10):1455-7.
From a CH2Cl2 extract of the Mediterranean sponge Anchinoe paupertas were isolated and characterized Zarzissine [2], a new 4,5-guanidino-pyridazine compound and the known p-hydroxybenzaldehyde [1]. The structure of Zarzissine [2] was elucidated by spectroscopic methods, including the application of a number of 2D nmr techniques. Biological activities of compounds 1 and 2 were also determined, with Zarzissine exhibiting cytotoxicity against three human and murine tumor cell lines.


