(2-Amino-1-hydroxyethyl)phosphonic acidCAS# 115511-00-7 |
Quality Control & MSDS
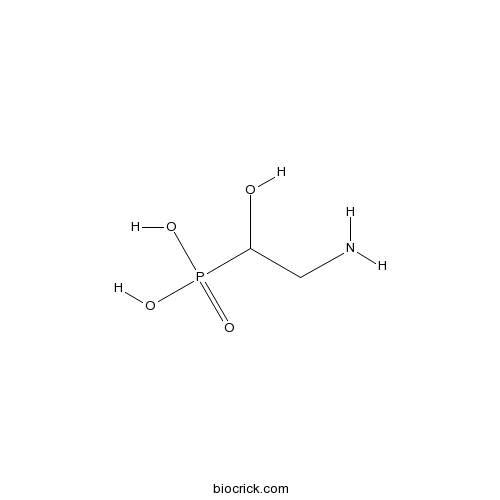
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 115511-00-7 | SDF | Download SDF |
| PubChem ID | 440756 | Appearance | Cryst. |
| Formula | C2H8NO4P | M.Wt | 141.06 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Synonyms | 1-Hydroxy-2-Aminoethylphosphonic Acid | ||
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (2-amino-1-hydroxyethyl)phosphonic acid | ||
| SMILES | C(C(O)P(=O)(O)O)N | ||
| Standard InChIKey | RTTXIBKRJFIBBG-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C2H8NO4P/c3-1-2(4)8(5,6)7/h2,4H,1,3H2,(H2,5,6,7) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Structure Identification | Acs Catalysis, 2017:3521-3531.Mechanism of Organophosphonate Catabolism by Diiron Oxygenase PhnZ: A Third Iron-Mediated O-O Activation Scenario in Nature.[Reference: WebLink]Diiron oxygenase PhnZ catalyzes the catabolism of organophosphonate (Pn) (R)-2-amino-1-hydroxyethylphosphonic((2-Amino-1-hydroxyethyl)phosphonic acid) to glycine and inorganic phosphate (Pi).
|

(2-Amino-1-hydroxyethyl)phosphonic acid Dilution Calculator

(2-Amino-1-hydroxyethyl)phosphonic acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 7.0892 mL | 35.4459 mL | 70.8918 mL | 141.7836 mL | 177.2295 mL |
| 5 mM | 1.4178 mL | 7.0892 mL | 14.1784 mL | 28.3567 mL | 35.4459 mL |
| 10 mM | 0.7089 mL | 3.5446 mL | 7.0892 mL | 14.1784 mL | 17.723 mL |
| 50 mM | 0.1418 mL | 0.7089 mL | 1.4178 mL | 2.8357 mL | 3.5446 mL |
| 100 mM | 0.0709 mL | 0.3545 mL | 0.7089 mL | 1.4178 mL | 1.7723 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- H-Lys(Z)-OH
Catalog No.:BCC2986
CAS No.:1155-64-2
- Z-Glu-OH
Catalog No.:BCC2781
CAS No.:1155-62-0
- 5,6,7,7a-Tetrahydrothieno[3,2-c]pyridine-2(4H)-one hydrochloride
Catalog No.:BCC8721
CAS No.:115473-15-9
- Glaucin B
Catalog No.:BCN6035
CAS No.:115458-73-6
- Sootepin D
Catalog No.:BCN6034
CAS No.:1154518-97-4
- Risedronate Sodium
Catalog No.:BCC2501
CAS No.:115436-72-1
- Niloticin
Catalog No.:BCN6033
CAS No.:115404-57-4
- Molidustat (BAY85-3934)
Catalog No.:BCC6412
CAS No.:1154028-82-6
- EIPA
Catalog No.:BCC7672
CAS No.:1154-25-2
- Kanshone B
Catalog No.:BCN7700
CAS No.:115370-61-1
- 2,2-Dimethyl-6-phenylpyrano[3,4-b]pyran-8-one
Catalog No.:BCN7280
CAS No.:1153624-36-2
- Kanshone A
Catalog No.:BCN7279
CAS No.:115356-18-8
- Fmoc-Cys(Trt)-Opfp
Catalog No.:BCC3480
CAS No.:115520-21-3
- BI 224436
Catalog No.:BCC5531
CAS No.:1155419-89-8
- Marbofloxacin
Catalog No.:BCC2510
CAS No.:115550-35-1
- Marbofloxacin hydrochloride
Catalog No.:BCC4250
CAS No.:115551-26-3
- 2'-Hydroxygenistein
Catalog No.:BCN6036
CAS No.:1156-78-1
- 4-Androstenediol
Catalog No.:BCC8692
CAS No.:1156-92-9
- Cinnamyl coumarate
Catalog No.:BCN7739
CAS No.:115610-30-5
- Cinnamyl isoferulate
Catalog No.:BCN7718
CAS No.:115610-31-6
- Cinnamyl caffeate
Catalog No.:BCN7721
CAS No.:115610-32-7
- H-DL-Pro-NH2
Catalog No.:BCC3027
CAS No.:115630-49-4
- B2
Catalog No.:BCC7505
CAS No.:115687-05-3
- Daidzein dimethyl ether
Catalog No.:BCN6761
CAS No.:1157-39-7


