(R)-(+)-CorypalmineCAS# 13063-54-2 |
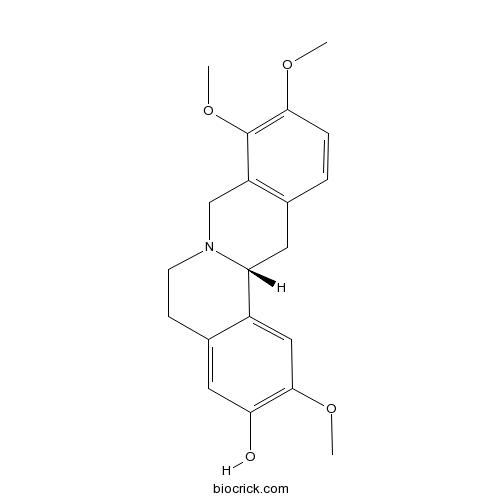
- Corypalmine
Catalog No.:BCN4111
CAS No.:6018-40-2
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 13063-54-2 | SDF | Download SDF |
| PubChem ID | 12304090 | Appearance | Powder |
| Formula | C20H23NO4 | M.Wt | 341.40 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (13aR)-2,9,10-trimethoxy-6,8,13,13a-tetrahydro-5H-isoquinolino[2,1-b]isoquinolin-3-ol | ||
| SMILES | COC1=C(C2=C(CC3C4=CC(=C(C=C4CCN3C2)O)OC)C=C1)OC | ||
| Standard InChIKey | BMCZTYDZHNTKPR-MRXNPFEDSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | (-)-Corypalmine has inhibition activity on spore germination of plant pathogenic and saprophytic fungi. |
| Targets | Antifection |
| In vitro | Efficacy of alkaloid (-)-corypalmine against spore germination of some fungi.[Pubmed: 12094740]Folia Microbiol (Praha). 2002;47(3):287-90.Inhibition activity of the alkaloid (-)-corypalmine on spore germination of plant pathogenic and saprophytic fungi (Alternaria solani, A brassicicola, A. brassicae, A. melongenae, Curvularia pallescens, C. lunata, C. maculans, Curvularisa sp., Colletotrichum sp., Helminthosporium speciferum, H. frumentacei, H. pennisetti, Heterosporium sp., Penicillum sp., Ustilago cynodontis) was determined. Spore germination of all the tested fungi was inhibited, Heterosporium sp. and Ustilago cynodontis being the most sensitive (complete inhibition of spore germination was observed at the very low concentration of 200 ppm). Curvularia palliscens, C. maculans and Curvularia sp. were less sensitive; complete inhibition of spore germination occurred at 400 ppm. |

(R)-(+)-Corypalmine Dilution Calculator

(R)-(+)-Corypalmine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.9291 mL | 14.6456 mL | 29.2912 mL | 58.5823 mL | 73.2279 mL |
| 5 mM | 0.5858 mL | 2.9291 mL | 5.8582 mL | 11.7165 mL | 14.6456 mL |
| 10 mM | 0.2929 mL | 1.4646 mL | 2.9291 mL | 5.8582 mL | 7.3228 mL |
| 50 mM | 0.0586 mL | 0.2929 mL | 0.5858 mL | 1.1716 mL | 1.4646 mL |
| 100 mM | 0.0293 mL | 0.1465 mL | 0.2929 mL | 0.5858 mL | 0.7323 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Nitidine chloride
Catalog No.:BCN4957
CAS No.:13063-04-2
- Yangambin
Catalog No.:BCN6706
CAS No.:13060-14-5
- alpha-Terthienylmethanol
Catalog No.:BCN6161
CAS No.:13059-93-3
- Mogroside III
Catalog No.:BCN3167
CAS No.:130567-83-8
- Liriope muscari baily Saponins
Catalog No.:BCN2817
CAS No.:130551-41-6
- Cannabisin A
Catalog No.:BCC8138
CAS No.:130508-46-2
- SKF 96365 hydrochloride
Catalog No.:BCC6953
CAS No.:130495-35-1
- TC-F 2
Catalog No.:BCC6147
CAS No.:1304778-15-1
- L-655,708
Catalog No.:BCC7023
CAS No.:130477-52-0
- ent-11alpha-Hydroxyabieta-8(14),13(15)-dien-16,12alpha-olide
Catalog No.:BCN7330
CAS No.:130466-20-5
- A 68930 hydrochloride
Catalog No.:BCC7104
CAS No.:130465-39-3
- Batimastat sodium salt
Catalog No.:BCC2075
CAS No.:130464-84-5
- N-(2-Hydroxyethyl)-3-(4-nitrophenyl)propylamine
Catalog No.:BCC9053
CAS No.:130634-09-2
- Bindarit
Catalog No.:BCC4965
CAS No.:130641-38-2
- PD123319
Catalog No.:BCC5010
CAS No.:130663-39-7
- Ozanimod (RPC1063)
Catalog No.:BCC6533
CAS No.:1306760-87-1
- RP 001 hydrochloride
Catalog No.:BCC7905
CAS No.:1306761-53-4
- Dorzolamide HCl
Catalog No.:BCC2311
CAS No.:130693-82-2
- (R)-(+)-Propranolol hydrochloride
Catalog No.:BCC6810
CAS No.:13071-11-9
- SDZ WAG 994
Catalog No.:BCC7374
CAS No.:130714-47-5
- FR 122047 hydrochloride
Catalog No.:BCC7092
CAS No.:130717-51-0
- Decloxizine dihydrochloride
Catalog No.:BCC5549
CAS No.:13073-96-6
- m-CPP hydrochloride
Catalog No.:BCC5680
CAS No.:13078-15-4
- 6-O-benzoylgomisin O
Catalog No.:BCN3092
CAS No.:130783-32-3
Efficacy of alkaloid (-)-corypalmine against spore germination of some fungi.[Pubmed:12094740]
Folia Microbiol (Praha). 2002;47(3):287-90.
Inhibition activity of the alkaloid (-)-corypalmine on spore germination of plant pathogenic and saprophytic fungi (Alternaria solani, A brassicicola, A. brassicae, A. melongenae, Curvularia pallescens, C. lunata, C. maculans, Curvularisa sp., Colletotrichum sp., Helminthosporium speciferum, H. frumentacei, H. pennisetti, Heterosporium sp., Penicillum sp., Ustilago cynodontis) was determined. Spore germination of all the tested fungi was inhibited, Heterosporium sp. and Ustilago cynodontis being the most sensitive (complete inhibition of spore germination was observed at the very low concentration of 200 ppm). Curvularia palliscens, C. maculans and Curvularia sp. were less sensitive; complete inhibition of spore germination occurred at 400 ppm.


