11-Keto-beta-boswellic acidCAS# 17019-92-0 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 17019-92-0 | SDF | Download SDF |
| PubChem ID | 71749672 | Appearance | White powder |
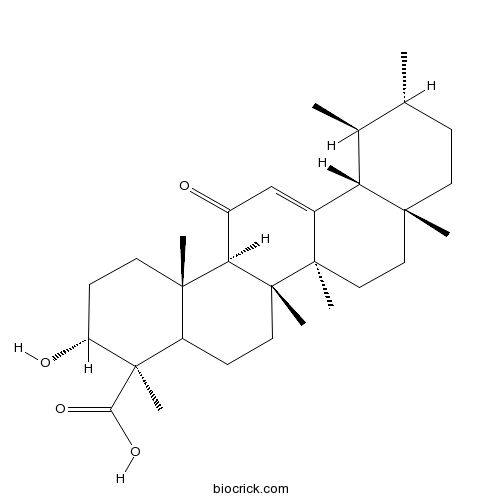
| Formula | C30H46O4 | M.Wt | 470.68 |
| Type of Compound | Triterpenoids | Storage | Desiccate at -20°C |
| Synonyms | KBA; 11-Oxo-β-boswellic acid | ||
| Solubility | Soluble in acetone, chloroform, diethyl ether, ethanol and methanol; insoluble in water | ||
| Chemical Name | (3R,4R,6aR,6bS,8aR,11R,12S,12aR,14aR,14bS)-3-hydroxy-4,6a,6b,8a,11,12,14b-heptamethyl-14-oxo-1,2,3,4a,5,6,7,8,9,10,11,12,12a,14a-tetradecahydropicene-4-carboxylic acid | ||
| SMILES | CC1CCC2(CCC3(C(=CC(=O)C4C3(CCC5C4(CCC(C5(C)C(=O)O)O)C)C)C2C1C)C)C | ||
| Standard InChIKey | YIMHGPSYDOGBPI-YEIMTCQQSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 11-Keto-beta-boswellic acid, a novel Nrf2 activator, and a selective 5-lipoxygenase (5-LOX) inhibitor; it exerts dose dependent cardioprotective effect manifested by dose-dependent reduction in serum lactate dehydrogenase; and it can increase the Nrf2 and HO-1 expression, which provides protection against oxygen and glucose deprivation (OGD)-induced oxidative insult. 11-Keto-beta-boswellic acid possesses significant anti-inflammatory, and anti-tumoral activities. |
| Targets | NO | PI3K | Akt | HSP (e.g. HSP90) | NF-kB | Bcl-2/Bax | Caspase | PARP | Nrf2 | HO-1 | PARP | LOX | COX | TNF-α | 5-LOX |
| In vitro | Biotransformation of 11-keto-β-boswellic acid by Cunninghamella blakesleana.[Pubmed: 23962801]Phytochemistry. 2013 Dec;96:330-6.11-Keto-beta-boswellic acid (KBA), as one of the active constituents in the gum resin of Boswellia serrata, possesses significant biological activities including anti-inflammatory activity. However, its extensive metabolism and low polarity has limited the systemic availability of 11-Keto-beta-boswellic acid. The present research was aimed to obtain and explore the various possible derivatives of 11-Keto-beta-boswellic acid through biotransformation by Cunninghamella blakesleana AS 3.970.
11-Keto-boswellic acid derived amides and monodesmosidic saponins induce apoptosis in breast and cervical cancers cells.[Pubmed: 26073487 ]Eur J Med Chem. 2015 Jul 15;100:98-105.Beta-boswellic acids are considered the main bioactive components of frankincense. Their potential to act as cytotoxic agents, as well as that of their derivatives remained unexploited so far.
|
| In vivo | Posttreatment with 11-Keto-β-Boswellic Acid Ameliorates Cerebral Ischemia-Reperfusion Injury: Nrf2/HO-1 Pathway as a Potential Mechanism.[Pubmed: 25452227]Mol Neurobiol. 2015 Dec;52(3):1430-1439.Oxidative stress is well known to play a pivotal role in cerebral ischemia-reperfusion injury. The nuclear factor erythroid-2-related factor 2 (Nrf2)/heme oxygenase-1 (HO-1) pathway has been considered a potential target for neuroprotection in stroke. 11-Keto-beta-boswellic acid(KBA) is a triterpenoid compound from extracts of Boswellia serrata. The aim of the present study was to determine whether KBA, a novel Nrf2 activator, can protect against cerebral ischemic injury.
|
| Kinase Assay | A novel cyano derivative of 11-keto-β-boswellic acid causes apoptotic death by disrupting PI3K/AKT/Hsp-90 cascade, mitochondrial integrity, and other cell survival signaling events in HL-60 cells.[Pubmed: 21751262]Mol Carcinog. 2012 Sep;51(9):679-95.Intervention of apoptosis is a promising strategy for discovery of novel anti-cancer therapeutics.
In this study, we examined the ability of a novel cyano derivative of 11-Keto-beta-boswellic acid , that is, butyl 2-cyano-3,11-dioxours-1,12-dien-24-oate (BCDD) to induce apoptosis in cancer cells.
|
| Cell Research | A propionyloxy derivative of 11-keto-β-boswellic acid induces apoptosis in HL-60 cells mediated through topoisomerase I & II inhibition.[Pubmed: 21056033 ]Chem Biol Interact. 2011 Jan 15;189(1-2):60-71.Boswellic acids have invariably been reported for their antiproliferative potential in various cell systems. In the present study the growth inhibitory effect of propionyloxy derivative of 11-Keto-beta-boswellic acid (PKBA; a semisynthetic analogue of 11-keto-β-boswellic acid) on HL-60 promyelocytic leukemia cells is being reported for the first time.
|
| Animal Research | The selective 5-LOX inhibitor 11-keto-β-boswellic acid protects against myocardial ischemia reperfusion injury in rats: involvement of redox and inflammatory cascades.[Pubmed: 23771412]Naunyn Schmiedebergs Arch Pharmacol. 2013 Sep;386(9):823-33.Myocardial ischemia induces 5-lipoxygenase (LOX) translocation and leukotriene production in the heart. Leukotrienes increase inflammatory responses aggravating, thereby, ischemia-reperfusion (I/R) injury. This study aimed to investigate whether the selective 5-LOX inhibitor 11-Keto-beta-boswellic acid (11-keto BA), in three different dose levels, exert a protective effect on myocardial I/R injury in an in vivo rat heart model.
|

11-Keto-beta-boswellic acid Dilution Calculator

11-Keto-beta-boswellic acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.1246 mL | 10.6229 mL | 21.2459 mL | 42.4917 mL | 53.1146 mL |
| 5 mM | 0.4249 mL | 2.1246 mL | 4.2492 mL | 8.4983 mL | 10.6229 mL |
| 10 mM | 0.2125 mL | 1.0623 mL | 2.1246 mL | 4.2492 mL | 5.3115 mL |
| 50 mM | 0.0425 mL | 0.2125 mL | 0.4249 mL | 0.8498 mL | 1.0623 mL |
| 100 mM | 0.0212 mL | 0.1062 mL | 0.2125 mL | 0.4249 mL | 0.5311 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Nepicastat (SYN-117) HCl
Catalog No.:BCC2286
CAS No.:170151-24-3
- Curcolone
Catalog No.:BCN3559
CAS No.:17015-43-9
- Alvimopan dihydrate
Catalog No.:BCC1348
CAS No.:170098-38-1
- Pseudobufarenogin
Catalog No.:BCN8234
CAS No.:17008-69-4
- Bufarenogin
Catalog No.:BCN2297
CAS No.:17008-65-0
- Reserpine hydrochloride
Catalog No.:BCC4279
CAS No.:16994-56-2
- LY 333531 hydrochloride
Catalog No.:BCC7969
CAS No.:169939-93-9
- Iso-cuparenal
Catalog No.:BCN7350
CAS No.:16982-01-7
- Mesuol
Catalog No.:BCN6583
CAS No.:16981-20-7
- Dibutyryl-cAMP, sodium salt
Catalog No.:BCC8079
CAS No.:16980-89-5
- Angelol K
Catalog No.:BCN8142
CAS No.:169736-93-0
- Ro 60-0175 fumarate
Catalog No.:BCC7196
CAS No.:169675-09-6
- Bauerenol acetate
Catalog No.:BCN1106
CAS No.:17020-04-1
- 24-Methylenecycloartane-3beta,26-diol
Catalog No.:BCN1530
CAS No.:17020-27-8
- CD 2314
Catalog No.:BCC6071
CAS No.:170355-37-0
- CD 2665
Catalog No.:BCC7778
CAS No.:170355-78-9
- Enzastaurin (LY317615)
Catalog No.:BCC1100
CAS No.:170364-57-5
- 4-(6-Methyl-4-oxohept-5-en-2-yl)cyclohex-2-en-1-one
Catalog No.:BCN7528
CAS No.:170380-68-4
- 1,4-Epidioxybisabola-2,10-dien-9-one
Catalog No.:BCN7532
CAS No.:170380-69-5
- Isohyperectine
Catalog No.:BCN3405
CAS No.:170384-75-5
- SC 236
Catalog No.:BCC7809
CAS No.:170569-86-5
- Oteromycin
Catalog No.:BCN1849
CAS No.:170591-45-4
- YC 1
Catalog No.:BCC7912
CAS No.:170632-47-0
- Fmoc-D-Abu-OH
Catalog No.:BCC3203
CAS No.:170642-27-0
The selective 5-LOX inhibitor 11-keto-beta-boswellic acid protects against myocardial ischemia reperfusion injury in rats: involvement of redox and inflammatory cascades.[Pubmed:23771412]
Naunyn Schmiedebergs Arch Pharmacol. 2013 Sep;386(9):823-33.
Myocardial ischemia induces 5-lipoxygenase (LOX) translocation and leukotriene production in the heart. Leukotrienes increase inflammatory responses aggravating, thereby, ischemia-reperfusion (I/R) injury. This study aimed to investigate whether the selective 5-LOX inhibitor 11-Keto-beta-boswellic acid (11-keto BA), in three different dose levels, exert a protective effect on myocardial I/R injury in an in vivo rat heart model. Sixty male Wister rats were used in this study and divided into five equal groups (n=12): GP1, sham-operated receiving normal saline; Gp 2, rats were subjected to 45 min left anterior descending coronary artery ligation followed by 4 h reperfusion to serve as I/R group. Gps 3-5 received 11-keto BA in doses 250, 500, 1,000 mg/kg, respectively, via an oral gavage for 7 days then were exposed to I/R. I/R injury induced a significant elevation in myeloperoxidase activity and gene expression of intracellular adhesion molecules, cyclooxygenase-2, 5-lipooxygenasae, nuclear factor kappa-beta, tumor necrosis factor alpha, nuclear factor (erythroid-derived 2)-like 2, and hemeoxygenease-1 consequently with reduction in glutathione peroxidase in heart tissues. Furthermore, immunohistochemical examination of the heart tissues showed positive immuostaining for both 3-nitrotyrosine and caspase-3 with DNA-ladder formation in all diseased rats. 11-keto BA in three dose levels exerted dose dependent cardioprotective effect manifested by dose-dependent reduction in serum lactate dehydrogenase and infract size through mechanisms related to enhancement of antioxidant capacity and prevention of inflammatory cascades.
A novel cyano derivative of 11-keto-beta-boswellic acid causes apoptotic death by disrupting PI3K/AKT/Hsp-90 cascade, mitochondrial integrity, and other cell survival signaling events in HL-60 cells.[Pubmed:21751262]
Mol Carcinog. 2012 Sep;51(9):679-95.
Intervention of apoptosis is a promising strategy for discovery of novel anti-cancer therapeutics. In this study, we examined the ability of a novel cyano derivative of 11-Keto-beta-boswellic acid, that is, butyl 2-cyano-3,11-dioxours-1,12-dien-24-oate (BCDD) to induce apoptosis in cancer cells. BCDD inhibited cell proliferation with 48 h IC(50) of 0.67 microM in HL-60, 1 microM in Molt4, and 1.5 microM in THP1 cells. The mechanism of cell death was investigated in HL-60 cells where it caused apoptosis by acting against several potential apoptosis suppressive targets. It inhibited phosphatidylinositol-3-kinase (PI3K)/AKT activity, NF-kappaB, Hsp-90, and survivin which may enhance the sensitivity of cells to apoptosis. Also, BCDD decreased the activity of Bid and Bax in cytosol, caused DeltaPsi(mt) loss, releasing pro-apoptotic cytochrome c, SMAC/DIABLO leading to caspase-9-mediated down stream activation of caspase-3, ICAD, and PARP1 cleavage. Translocation of apoptotis-inducing factor (AIF) from mitochondria to the nucleus indicated some caspases-independent apoptosis. Though it upregulated DR-5 and caspase-8, the caspase inhibitor yet had no effect on apoptosis as against 75% inhibition by caspase-9 inhibitor. Attempts were made to examine any acclaimed role of AIF in the activation of caspase-8 using siRNA where it had no effect on caspase-8 activity while the Bax-siRNA inhibited caspase-3 activation suggesting predominance of intrinsic signaling. Our studies thus demonstrated that BCDD exerts multi-focal action in cancer cells while it required 10-fold higher the concentration to produce cytotoxicity in normal human PBMC and gingival cell line, and therefore, may find usefulness in the management of human leukemia.
Biotransformation of 11-keto-beta-boswellic acid by Cunninghamella blakesleana.[Pubmed:23962801]
Phytochemistry. 2013 Dec;96:330-6.
11-Keto-beta-boswellic acid (KBA), as one of the active constituents in the gum resin of Boswellia serrata, possesses significant biological activities including anti-inflammatory activity. However, its extensive metabolism and low polarity has limited the systemic availability of KBA. The present research was aimed to obtain and explore the various possible derivatives of KBA through biotransformation by Cunninghamella blakesleana AS 3.970. A total of ten transformed compounds were isolated and purified, and their chemical structures were characterized as 7beta-hydroxy-11-Keto-beta-boswellic acid; 7beta, 15alpha-dihydroxy-11-Keto-beta-boswellic acid ; 7beta, 16beta-dihydroxy-11-Keto-beta-boswellic acid; 7beta, 16alpha-dihydroxy-11-Keto-beta-boswellic acid; 7beta, 22beta-dihydroxy-11-Keto-beta-boswellic acid; 7beta, 21beta-dihydroxy-11-Keto-beta-boswellic acid; 7beta, 20beta-dihydroxy-11-Keto-beta-boswellic acid; 7beta, 30-dihydroxy-11-Keto-beta-boswellic acid; 3alpha, 7beta-dihydroxy-11-oxours-12-ene-24, 30-dioic acid and 3alpha, 7beta-dihydroxy-30-(2-hydroxypropanoyloxy)-11-oxours-12-en-24-oic acid by various spectroscopic methods. The biotransformation processes include hydroxylation, oxidation and esterification. Primary structure-activity relationships (SAR) of inhibitory effects on NO production in RAW 264.7 macrophage cells are discussed.
A propionyloxy derivative of 11-keto-beta-boswellic acid induces apoptosis in HL-60 cells mediated through topoisomerase I & II inhibition.[Pubmed:21056033]
Chem Biol Interact. 2011 Jan 15;189(1-2):60-71.
Boswellic acids have invariably been reported for their antiproliferative potential in various cell systems. In the present study the growth inhibitory effect of propionyloxy derivative of 11-Keto-beta-boswellic acid (PKBA; a semisynthetic analogue of 11-Keto-beta-boswellic acid) on HL-60 promyelocytic leukemia cells is being reported for the first time. In the preliminary studies, in vitro cytotoxicity of PKBA was investigated against eight human cancer cell lines viz., IMR-32, SF-295 (both neuroblastoma), PC-3 (prostate), Colo-205 (colon), MCF-7 (breast), OVCAR-5 (ovary), HL-60, Molt-4 (both leukemia) and their respective IC(50) values were found to be 5.95, 7.11, 15.2, 14.5, 15, 15.9, 8.7 & 9.5mug/ml, respectively. For determining the mechanism of cell death in HL-60 cells, PKBA was subjected to different mechanistic studies. DNA relaxation assay of PKBA revealed inhibition of both topoisomerases I & II. The fragmentation analysis of DNA revealed typical ladders indicating the cytotoxic effect to be mediated by induction of apoptosis. The morphologic studies of PKBA showed the presence of true apoptotic bodies. Apoptosis was confirmed further by flow-cytometric detection of sub-G(1) peaks and enhanced annexin-V-FITC binding of the cells. The activation of apoptotic cascade by PKBA in HL-60 cells was found to be associated with the loss of mitochondrial membrane potential, release of cytochrome c, activation of initiator and executioner caspases and cleavage of poly ADP ribose polymerase (PARP). In vivo studies of PKBA revealed anti-tumoral activity against both ascitic and solid murine tumor models. These studies thus demonstrate PKBA to induce apoptosis in HL-60 cells due to the inhibition of topoisomerases I and II.
11-Keto-boswellic acid derived amides and monodesmosidic saponins induce apoptosis in breast and cervical cancers cells.[Pubmed:26073487]
Eur J Med Chem. 2015 Jul 15;100:98-105.
Beta-boswellic acids are considered the main bioactive components of frankincense. Their potential to act as cytotoxic agents, as well as that of their derivatives remained unexploited so far. In this study we were able to prepare derivatives of 11-Keto-beta-boswellic acid (KBA) that showed lower IC50 values as determined by a sulphorhodamine B (SRB) assay using several different human tumour cell lines. Monodesmosidic saponins of KBA are as cytotoxic as 3-acetyl-KBA. The presence of a free hydroxyl group at position C-3 seems to lower cytotoxicity while the presence of an amide function at C-24 improves cytotoxicity. The most active compound of this series gave IC50 values as low as 4.5 muM. Cell death proceeded mainly via apoptosis.
Posttreatment with 11-Keto-beta-Boswellic Acid Ameliorates Cerebral Ischemia-Reperfusion Injury: Nrf2/HO-1 Pathway as a Potential Mechanism.[Pubmed:25452227]
Mol Neurobiol. 2015 Dec;52(3):1430-1439.
Oxidative stress is well known to play a pivotal role in cerebral ischemia-reperfusion injury. The nuclear factor erythroid-2-related factor 2 (Nrf2)/heme oxygenase-1 (HO-1) pathway has been considered a potential target for neuroprotection in stroke. 11-Keto-beta-boswellic acid (KBA) is a triterpenoid compound from extracts of Boswellia serrata. The aim of the present study was to determine whether KBA, a novel Nrf2 activator, can protect against cerebral ischemic injury. Middle cerebral artery occlusion (MCAO) was operated on male Sprague-Dawley rats. KBA (25 mg/kg) applied 1 h after reperfusion significantly reduced infarct volumes and apoptotic cells as well as increased neurologic scores at 48 h after reperfusion. Meanwhile, posttreatment with KBA significantly decreased malondialdehyde (MDA) levels, restored the superoxide dismutase (SOD) activity, and increased the protein Nrf2 and HO-1 expression in brain tissues. In primary cultured astrocytes, KBA increased the Nrf2 and HO-1 expression, which provided protection against oxygen and glucose deprivation (OGD)-induced oxidative insult. But knockdown of Nrf2 or HO-1 attenuated the protective effect of KBA. In conclusion, these findings provide evidence that the neuroprotection of KBA against oxidative stress-induced ischemic injury involves the Nrf2/HO-1 pathway.


