20-HydroxyecdysoneCAS# 5289-74-7 |
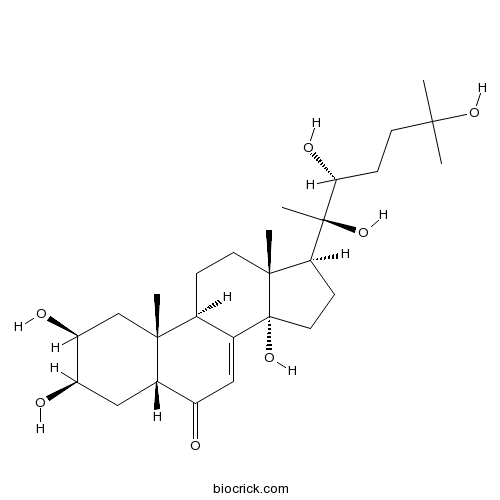
- Rhapontisterone B
Catalog No.:BCN2664
CAS No.:698975-64-3
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 5289-74-7 | SDF | Download SDF |
| PubChem ID | 5459840 | Appearance | White-pale yellow powder |
| Formula | C27H44O7 | M.Wt | 480.6 |
| Type of Compound | Steroids | Storage | Desiccate at -20°C |
| Synonyms | Commisterone; Crustecdysone; β-Ecdysterone; 20-Hydroxy α-ecdysone; Isoinokosterone; Polypodine A; Polypodine C; Viticosterone | ||
| Solubility | DMSO : 125 mg/mL (260.08 mM; Need ultrasonic) | ||
| Chemical Name | (2S,3R,5R,9R,10R,13R,14S,17S)-2,3,14-trihydroxy-10,13-dimethyl-17-[(2R,3R)-2,3,6-trihydroxy-6-methylheptan-2-yl]-2,3,4,5,9,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-6-one | ||
| SMILES | CC12CCC3C(=CC(=O)C4C3(CC(C(C4)O)O)C)C1(CCC2C(C)(C(CCC(C)(C)O)O)O)O | ||
| Standard InChIKey | NKDFYOWSKOHCCO-YPVLXUMRSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 20-Hydroxyecdysone (20E) is a naturally occurring ecdysteroid hormone which controls the ecdysis (moulting) and metamorphosis of arthropods. 20E could as ingredients in nutritional supplements for various sports, particularly bodybuilding; it induces autophagy and caspase activity, it slowly reduces food consumption and then indirectly induces a state of starvation resulting in the elevation of the mRNA levels of InR , IRS , PI3K110 , and PDK in the Bombyx fat body during molting and pupation, and it inhibits innate immunity in the fat body during Bombyx postembryonic development. |
| Targets | Calcium Channel | PI3K | mTOR | Akt | Autophagy | PDK | Caspase | InR | IRS |
| In vitro | 20-Hydroxyecdysone is required for, and negatively regulates, transcription of Drosophila pupal cuticle protein genes.[Pubmed: 1713868]Dev Biol. 1991 Aug;146(2):569-82.
|
| In vivo | 20-hydroxyecdysone mediates non-canonical regulation of mosquito vitellogenins through alternative splicing.[Pubmed: 24720618]Insect Mol Biol. 2014 Aug;23(4):407-16.
Vitellogenesis is one of the most well-studied physiological processes in mosquitoes. Expression of mosquito vitellogenin genes is classically described as being restricted to female adult reproduction.
E93 predominantly transduces 20-hydroxyecdysone signaling to induce autophagy and caspase activity in Drosophila fat body.[Pubmed: 24316411]Insect Biochem Mol Biol. 2014 Feb;45:30-9.During the larval-prepupal transition in Drosophila, a balancing crosstalk occurs between autophagy and caspase activity in the remodeling fat body: the inhibition of autophagy induces caspase activity and the inhibition of caspases induces autophagy.
Both autophagy and caspase activity are induced by a pulse of molting hormone (20-Hydroxyecdysone, 20E) via the 20E nuclear receptor complex, EcR-USP.
|
| Kinase Assay | Transcriptional regulation of the insulin signaling pathway genes by starvation and 20-hydroxyecdysone in the Bombyx fat body.[Pubmed: 20197069 ]The steroid hormone 20-hydroxyecdysone via nongenomic pathway activates Ca2+/calmodulin-dependent protein kinase II to regulate gene expression.[Pubmed: 25670853 ]J Biol Chem. 2015 Mar 27;290(13):8469-81.The steroid hormone 20-Hydroxyecdysone (20E) triggers calcium signaling pathway to regulate 20E response gene expression, but the mechanism underlying this process remains unclear.
J Insect Physiol. 2010 Oct;56(10):1436-44.Genetic studies in the fruitfly, Drosophila melanogaster, have uncovered a conserved insulin/insulin growth factor signaling (IIS) pathway that regulates nutrition-dependent growth rates of insects.
|

20-Hydroxyecdysone Dilution Calculator

20-Hydroxyecdysone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.0807 mL | 10.4037 mL | 20.8073 mL | 41.6146 mL | 52.0183 mL |
| 5 mM | 0.4161 mL | 2.0807 mL | 4.1615 mL | 8.3229 mL | 10.4037 mL |
| 10 mM | 0.2081 mL | 1.0404 mL | 2.0807 mL | 4.1615 mL | 5.2018 mL |
| 50 mM | 0.0416 mL | 0.2081 mL | 0.4161 mL | 0.8323 mL | 1.0404 mL |
| 100 mM | 0.0208 mL | 0.104 mL | 0.2081 mL | 0.4161 mL | 0.5202 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Olean-12-ene-3,11-diol
Catalog No.:BCN5686
CAS No.:5282-14-4
- Cearoin
Catalog No.:BCN7772
CAS No.:52811-37-7
- Dalbergiphenol
Catalog No.:BCN7451
CAS No.:52811-31-1
- L-Quisqualic acid
Catalog No.:BCC6568
CAS No.:52809-07-1
- Isobavachromene
Catalog No.:BCN3192
CAS No.:52801-22-6
- (±)-Galgravin
Catalog No.:BCN8283
CAS No.:528-63-2
- Cyanidin Chloride
Catalog No.:BCN1231
CAS No.:528-58-5
- Delphinidin chloride
Catalog No.:BCN3015
CAS No.:528-53-0
- Fisetin
Catalog No.:BCN5024
CAS No.:528-48-3
- Magnolol
Catalog No.:BCN5687
CAS No.:528-43-8
- H-Phe(3,4-DiCl)-OH
Catalog No.:BCC3178
CAS No.:52794-99-7
- H-D-Phe(3,4-DiCl)-OH
Catalog No.:BCC3179
CAS No.:52794-98-6
- Chamazulene
Catalog No.:BCC8145
CAS No.:529-05-5
- Ombuin
Catalog No.:BCN5691
CAS No.:529-40-8
- Myricetin
Catalog No.:BCN5692
CAS No.:529-44-2
- Gentisein
Catalog No.:BCN3356
CAS No.:529-49-7
- Azaleatin
Catalog No.:BCN8207
CAS No.:529-51-1
- Scutellarein
Catalog No.:BCN5380
CAS No.:529-53-3
- Prunin
Catalog No.:BCN5693
CAS No.:529-55-5
- Genistin
Catalog No.:BCN2396
CAS No.:529-59-9
- Euxanthone
Catalog No.:BCN5694
CAS No.:529-61-3
- 6,7-Dihydroxy-4-Methylcoumarin
Catalog No.:BCC8287
CAS No.:529-84-0
- Motilin (human, porcine)
Catalog No.:BCC5894
CAS No.:52906-92-0
- PRIMA-1MET
Catalog No.:BCC2414
CAS No.:5291-32-7
E93 predominantly transduces 20-hydroxyecdysone signaling to induce autophagy and caspase activity in Drosophila fat body.[Pubmed:24316411]
Insect Biochem Mol Biol. 2014 Feb;45:30-9.
During the larval-prepupal transition in Drosophila, a balancing crosstalk occurs between autophagy and caspase activity in the remodeling fat body: the inhibition of autophagy induces caspase activity and the inhibition of caspases induces autophagy. Both autophagy and caspase activity are induced by a pulse of molting hormone (20-Hydroxyecdysone, 20E) via the 20E nuclear receptor complex, EcR-USP. We here demonstrate that E93, a 20E primary-response gene encoding an HTH transcription factor, predominantly transduces 20E signaling to induce autophagy and caspase activity in the remodeling fat body. RNAi knockdown or mutation of E93 blocks autophagy and caspase activity, E93 overexpression induces them both, while E93 overexpression has a better rescuing effect on the inhibition of autophagy than caspase activity caused by EcR(DN) overexpression. At the transcriptional level, E93 not only greatly impacts the 20E-triggered transcriptional cascade, but also upregulates essential autophagy and apoptosis genes. Meanwhile, at the phosphorylational level, E93 blocks the PI3K-TORC1 signaling to initiate autophagy. Taken together, we conclude that autophagy and caspase activity are induced by 20E and predominantly transduced by E93 in the remodeling fat body of Drosophila.
The steroid hormone 20-hydroxyecdysone via nongenomic pathway activates Ca2+/calmodulin-dependent protein kinase II to regulate gene expression.[Pubmed:25670853]
J Biol Chem. 2015 Mar 27;290(13):8469-81.
The steroid hormone 20-Hydroxyecdysone (20E) triggers calcium signaling pathway to regulate 20E response gene expression, but the mechanism underlying this process remains unclear. We propose that the 20E-induced phosphorylation of Ca(2+)/calmodulin-dependent protein kinase II (CaMKII) serves an important function in 20E response gene transcription in the lepidopteran insect Helicoverpa armigera. CaMKII showed increased expression and phosphorylation during metamorphosis. 20E elevated CaMKII phosphorylation. However, the G protein-coupled receptor (GPCR) and ryanodine receptor inhibitor suramin, the phospholipase C inhibitor U73122, and the inositol 1,4,5-triphosphate receptor inhibitor xestospongin C suppressed 20E-induced CaMKII phosphorylation. Two ecdysone-responsible GPCRs and Galphaq protein were involved in 20E-induced CaMKII phosphorylation by RNA interference analysis. 20E regulated CaMKII threonine phosphorylation at amino acid 290, thereby inducing CaMKII nuclear translocation. CaMKII knockdown by dsCaMKII injection into the larvae prevented the occurrence of larval-pupal transition and suppressed 20E response gene expression. CaMKII phosphorylation and nuclear translocation maintained USP1 lysine acetylation at amino acid 303 by inducing histone deacetylase 3 phosphorylation and nuclear export. The lysine acetylation of USP1 was necessary for the interaction of USP1 with EcRB1 and their binding to the ecdysone response element. Results suggest that 20E (via GPCR activation and calcium signaling) activates CaMKII phosphorylation and nuclear translocation, which regulate USP1 lysine acetylation to form an EcRB1-USP1 complex for 20E response gene transcription.
20-hydroxyecdysone mediates non-canonical regulation of mosquito vitellogenins through alternative splicing.[Pubmed:24720618]
Insect Mol Biol. 2014 Aug;23(4):407-16.
Vitellogenesis is one of the most well-studied physiological processes in mosquitoes. Expression of mosquito vitellogenin genes is classically described as being restricted to female adult reproduction. We report premature vitellogenin transcript expression in three vector mosquitoes: Culex tarsalis, Aedes aegypti and Anopheles gambiae. Vitellogenins expressed during non-reproductive stages are alternatively spliced to retain their first intron and encode premature termination codons. We show that intron retention results in transcript degradation by translation-dependent nonsense-mediated mRNA decay. This is probably an example of regulated unproductive splicing and translation (RUST), a mechanism known to regulate gene expression in numerous organisms but which has never been described in mosquitoes. We demonstrate that the hormone 20-Hydroxyecdysone (20E) is responsible for regulating post-transcriptional splicing of vitellogenin. After exposure of previtellogenic fat bodies to 20E, vitellogenin expression switches from a non-productive intron-retaining transcript to a spliced protein-coding transcript. This effect is independent of factors classically known to influence transcription, such as juvenile hormone-mediated competence and amino acid signalling through the target of rapamycin pathway. Non-canonical regulation of vitellogenesis through RUST is a novel role for the multifunctional hormone 20E, and may have important implications for general patterns of gene regulation in mosquitoes.
Balancing crosstalk between 20-hydroxyecdysone-induced autophagy and caspase activity in the fat body during Drosophila larval-prepupal transition.[Pubmed:24036278]
Insect Biochem Mol Biol. 2013 Nov;43(11):1068-78.
In the fruitfly, Drosophila melanogaster, autophagy and caspase activity function in parallel in the salivary gland during metamorphosis and in a common regulatory hierarchy during oogenesis. Both autophagy and caspase activity progressively increase in the remodeling fat body, and they are induced by a pulse of the molting hormone (20-Hydroxyecdysone, 20E) during the larval-prepupal transition. Inhibition of autophagy and/or caspase activity in the remodeling fat body results in 25-40% pupal lethality, depending on the genotypes. Interestingly, a balancing crosstalk occurs between autophagy and caspase activity in this tissue: the inhibition of autophagy induces caspase activity and the inhibition of caspases induces autophagy. The Drosophila remodeling fat body provides an in vivo model for understanding the molecular mechanism of the balancing crosstalk between autophagy and caspase activity, which oppose with each other and are induced by the common stimulus 20E, and blockage of either path reinforces the other path.
Transcriptional regulation of the insulin signaling pathway genes by starvation and 20-hydroxyecdysone in the Bombyx fat body.[Pubmed:20197069]
J Insect Physiol. 2010 Oct;56(10):1436-44.
Genetic studies in the fruitfly, Drosophila melanogaster, have uncovered a conserved insulin/insulin growth factor signaling (IIS) pathway that regulates nutrition-dependent growth rates of insects. From the silkworm, Bombyx mori, we have identified and characterized several key genes involved in the IIS pathway, including InR, IRS, PI3K110, PI3K60, PTEN, PDK, and Akt. Tissue distribution analysis showed that most of these genes were highly expressed in the fat body implying that the IIS pathway is functionally important within insect adipose tissue. Developmental profile studies revealed that the expression levels of InR, IRS, PI3K110, and PDK were elevated in the fat body during molting and pupation, periods when animals ceased feeding and hemolymph levels of 20-Hydroxyecdysone (20E) were high. Starvation rapidly up-regulated the mRNA levels of these same genes in the fat body, while 20E slowly induced their transcription. We conclude that 20E slowly reduces food consumption and then indirectly induces a state of starvation resulting in the elevation of the mRNA levels of InR, IRS, PI3K110, and PDK in the Bombyx fat body during molting and pupation.
20-Hydroxyecdysone is required for, and negatively regulates, transcription of Drosophila pupal cuticle protein genes.[Pubmed:1713868]
Dev Biol. 1991 Aug;146(2):569-82.
Transcripts of ecdysone-dependent genes (EDGs) accumulate in isolated imaginal discs with 8 hr after exposure to a pulse of the steroid hormone 20-Hydroxyecdysone (20-HE; 1 microgram/ml for 6 hr) but not in discs cultured in the continuous presence or absence of the hormone. Sequence analyses show that two of the EDGs are members of gene families encoding insect cuticle proteins. We conclude that a third EDG encodes a cuticle protein because the conceptual glycine-rich protein contains sequence motifs similar to those found in insect egg shell proteins and vertebrate cytokeratins and because expression of this gene is limited to tissues that deposit the pupal cuticle. Nuclear run-on assays show that the hormone-dependent expression of each of these EDGs is due to transcriptional regulation. Readdition of hormone to imaginal discs actively synthesizing the EDG messages causes rapid repression of EDG transcription. Thus, 20-HE acts as both a positive and a negative regulator of EDG transcription. Sequences in the promoter regions of two of the EDGs are similar to an ecdysone response element and may play a role in negative regulation.


