AnonamineCAS# 111566-66-6 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 111566-66-6 | SDF | Download SDF |
| PubChem ID | 6441178 | Appearance | Powder |
| Formula | C19H27NO7 | M.Wt | 381.42 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
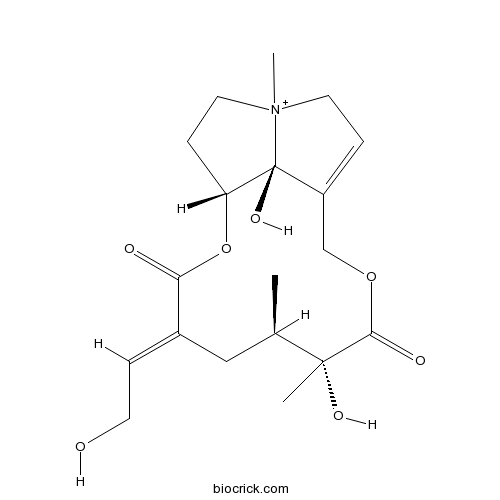
| SMILES | CC1CC(=CCO)C(=O)OC2CC[N+]3(C2(C(=CC3)COC(=O)C1(C)O)O)C | ||
| Standard InChIKey | SFTHPGHXDNRVHD-FLRXXRQUSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Anonamine is a natural product from Senecio scandens. |
| In vitro | Macrocyclic pyrrolizidine alkaloids from Senecio anonymus. Separation of a complex alkaloid extract using droplet counter-current chromatography.[Pubmed: 3210016]J Nat Prod. 1988 Jul-Aug;51(4):690-702.
|
| Structure Identification | J Nat Prod. 1991 Sep-Oct;54(5):1425-6.Acetylanonamine, a new secopyrrolizidine alkaloid from Senecio anonymus.[Pubmed: 1800641]A new secopyrrolizidine alkaloid, acetylAnonamine, was isolated from Senecio anonymus.
|

Anonamine Dilution Calculator

Anonamine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.6218 mL | 13.1089 mL | 26.2178 mL | 52.4356 mL | 65.5445 mL |
| 5 mM | 0.5244 mL | 2.6218 mL | 5.2436 mL | 10.4871 mL | 13.1089 mL |
| 10 mM | 0.2622 mL | 1.3109 mL | 2.6218 mL | 5.2436 mL | 6.5545 mL |
| 50 mM | 0.0524 mL | 0.2622 mL | 0.5244 mL | 1.0487 mL | 1.3109 mL |
| 100 mM | 0.0262 mL | 0.1311 mL | 0.2622 mL | 0.5244 mL | 0.6554 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 3',5-Dihydroxy-4',5',6,7-tetramethoxyflavone
Catalog No.:BCN1620
CAS No.:111537-41-8
- Fmoc-D-Phg-OH
Catalog No.:BCC3316
CAS No.:111524-95-9
- Nyasicol
Catalog No.:BCN5999
CAS No.:111518-95-7
- Nyasicoside
Catalog No.:BCN5998
CAS No.:111518-94-6
- Metformin HCl
Catalog No.:BCC4799
CAS No.:1115-70-4
- L-Cysteinesulfinic acid
Catalog No.:BCC6571
CAS No.:1115-65-7
- H-Ala-OEt.HCl
Catalog No.:BCC2687
CAS No.:1115-59-9
- Ac-DL-Met-OH
Catalog No.:BCC2999
CAS No.:1115-47-5
- 25-Anhydroalisol F
Catalog No.:BCN3361
CAS No.:1114895-01-0
- Amlodipine Besylate
Catalog No.:BCC4397
CAS No.:111470-99-6
- Naltrindole hydrochloride
Catalog No.:BCC6773
CAS No.:111469-81-9
- Axinysone B
Catalog No.:BCN7713
CAS No.:1114491-60-9
- Cyanidin-3-O-arabinoside chloride
Catalog No.:BCN3023
CAS No.:111613-04-8
- GSK1838705A
Catalog No.:BCC3787
CAS No.:1116235-97-2
- Elastase Inhibitor
Catalog No.:BCC1225
CAS No.:111682-13-4
- Remacemide hydrochloride
Catalog No.:BCC7129
CAS No.:111686-79-4
- Demethoxyfumitremorgin C
Catalog No.:BCN7240
CAS No.:111768-16-2
- H-Glu-Oet.HCl
Catalog No.:BCC2685
CAS No.:1118-89-4
- (S)-(-)-HA-966
Catalog No.:BCC6589
CAS No.:111821-58-0
- 10-O-Methylprotosappanin B
Catalog No.:BCN6599
CAS No.:111830-77-4
- Hancinone C
Catalog No.:BCN4751
CAS No.:111843-10-8
- UCPH 101
Catalog No.:BCC7692
CAS No.:1118460-77-7
- BIM 23042
Catalog No.:BCC5998
CAS No.:111857-96-6
- 2,4-Dihydroxyphenylacetyl-L-asparagine
Catalog No.:BCC6585
CAS No.:111872-98-1
Acetylanonamine, a new secopyrrolizidine alkaloid from Senecio anonymus.[Pubmed:1800641]
J Nat Prod. 1991 Sep-Oct;54(5):1425-6.
A new secopyrrolizidine alkaloid, acetylAnonamine, was isolated from Senecio anonymus. The structure was established by high resolution nmr (1H-1H COSY and 1H-13C HETCOR), ms, comparison of these spectral data with those of Anonamine, and synthesis of 1 from 2.
Macrocyclic pyrrolizidine alkaloids from Senecio anonymus. Separation of a complex alkaloid extract using droplet counter-current chromatography.[Pubmed:3210016]
J Nat Prod. 1988 Jul-Aug;51(4):690-702.
Ten 12-membered macrocyclic pyrrolizidine alkaloids, all of them esters of the necines, retronecine or otonecine, have been isolated from Senecio anonymus. The separation, carried out by droplet counter-current chromatography, afforded senecionine [1], integerrimine [2], retrorsine [3], senkirkine [5], neosenkirkine [6], otosenine [10], hydroxysenkirkine [7], and a new alkaloid given the trivial name Anonamine [9]. Traces of usaramine [4] and another new alkaloid, hydroxyneosenkirkine [8], were detected by 1H nmr. In addition, the previously unreported 3a beta-hydroxy-4-ethoxy-2,6-perhydroindoledione [11] was isolated. X-ray structures were obtained for neosenkirkine [6], hydroxysenkirkine [7], Anonamine [9], and [11]. 1H-13C heteronuclear shift correlated nmr (HETCOR) provided unambiguous chemical shift assignments for 13C-nmr data. Antitumor activity was assayed using the A204-rhabdomyosarcoma cell line in soft agarose.


