BMS 299897γ--secretase inhibitor, potent and orally active CAS# 290315-45-6 |
- BMS-708163 (Avagacestat)
Catalog No.:BCC2104
CAS No.:1146699-66-2
- Compound W
Catalog No.:BCC2341
CAS No.:173550-33-9
- DAPT (GSI-IX)
Catalog No.:BCC3618
CAS No.:208255-80-5
- Semagacestat (LY450139)
Catalog No.:BCC3610
CAS No.:425386-60-3
- MK-0752
Catalog No.:BCC2090
CAS No.:471905-41-6
- Begacestat
Catalog No.:BCC2346
CAS No.:769169-27-9
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 290315-45-6 | SDF | Download SDF |
| PubChem ID | 11249248 | Appearance | Powder |
| Formula | C24H21ClF3NO4S | M.Wt | 511.94 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : ≥ 30 mg/mL (58.60 mM) *"≥" means soluble, but saturation unknown. | ||
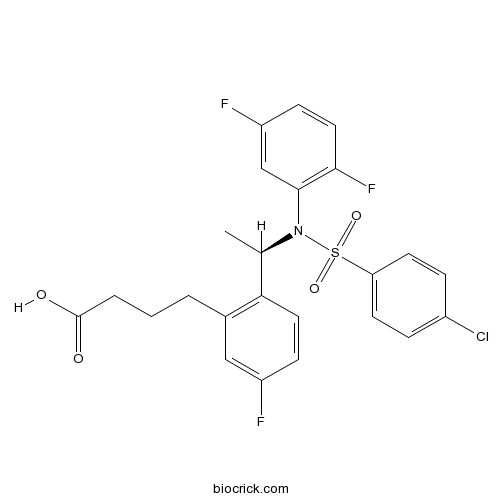
| Chemical Name | 4-[2-[(1R)-1-(N-(4-chlorophenyl)sulfonyl-2,5-difluoroanilino)ethyl]-5-fluorophenyl]butanoic acid | ||
| SMILES | CC(C1=C(C=C(C=C1)F)CCCC(=O)O)N(C2=C(C=CC(=C2)F)F)S(=O)(=O)C3=CC=C(C=C3)Cl | ||
| Standard InChIKey | IZAOBRWCUGOKNH-OAHLLOKOSA-N | ||
| Standard InChI | InChI=1S/C24H21ClF3NO4S/c1-15(21-11-7-18(26)13-16(21)3-2-4-24(30)31)29(23-14-19(27)8-12-22(23)28)34(32,33)20-9-5-17(25)6-10-20/h5-15H,2-4H2,1H3,(H,30,31)/t15-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Orally active, potent γ-secretase inhibitor (IC50 = 12 nM). Inhibits Aβ40 and Aβ42 formation in vitro (IC50 values are 7.4 and 7.9 nM respectively) and reduces Aβ in the brain, plasma and cerebrospinal fluid in vivo. Exhibits no Notch toxicity. Brain penetrant. |

BMS 299897 Dilution Calculator

BMS 299897 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.9534 mL | 9.7668 mL | 19.5335 mL | 39.0671 mL | 48.8338 mL |
| 5 mM | 0.3907 mL | 1.9534 mL | 3.9067 mL | 7.8134 mL | 9.7668 mL |
| 10 mM | 0.1953 mL | 0.9767 mL | 1.9534 mL | 3.9067 mL | 4.8834 mL |
| 50 mM | 0.0391 mL | 0.1953 mL | 0.3907 mL | 0.7813 mL | 0.9767 mL |
| 100 mM | 0.0195 mL | 0.0977 mL | 0.1953 mL | 0.3907 mL | 0.4883 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
BMS-299897 is a selective inhibitor of γ-secretase with IC50 value of 12 nM [1].
γ-secretase is an intergral membrane protein whose well-known substrate is amyloid precursor protein and involves in the process of cleaving single-pass transmembrane proteins at residues with the transmembrane domain. It has been revealed thatγ-secretase cleave APP in any of multiple sites and generate a 39 to 42 amino acids long peptide, among which Aβ40 is the most common isoform and Aβ42 is the most susceptible to conformational changes leading to amyloid fibrillogenesis. Many studies have shown that reduction of brain Aβ synthesis by gamma-secretase inhibitors is a promising approach for the Alzheimer's disease treatment [1] [2].
BMS 299897 is a selective inhibitorγ-secretase. When tested HEKwt culture with BMS-299897, the expression of Aβ(1-40) in culture supernatant had a robust rise throughγ-secretase inhibition [3].
In male Swiss mice model with intracerebroventricular (i.c.v.) injection of Aβ(25-35) which induced Alzheimer's disease pathomimetic toxicity, administration of BMS-299897 markedly attenuated Aβ(1-42)-whose accumulation marginally contributed to the toxicity or long-term memory deficits-seeding and toxicity induced by Aβ(25-35) through inhibiting γ-secretase [4]. When tested with Tg2576 mice, oral administration of BMS-299897 markedly reduced Abeta peptide concentrations by inhibitingγ-secretase [5] [2].
References:
[1]. Anderson, J.J., et al., Reductions in beta-amyloid concentrations in vivo by the gamma-secretase inhibitors BMS-289948 and BMS-299897. Biochem Pharmacol, 2005. 69(4): p. 689-98.
[2]. Barten, D.M., et al., Dynamics of {beta}-amyloid reductions in brain, cerebrospinal fluid, and plasma of {beta}-amyloid precursor protein transgenic mice treated with a {gamma}-secretase inhibitor. J Pharmacol Exp Ther, 2005. 312(2): p. 635-43.
[3]. Burton, C.R., et al., The amyloid-beta rise and gamma-secretase inhibitor potency depend on the level of substrate expression. J Biol Chem, 2008. 283(34): p. 22992-3003.
[4]. Meunier, J., et al., The gamma-secretase inhibitor 2-[(1R)-1-[(4-chlorophenyl)sulfonyl](2,5-difluorophenyl) amino]ethyl-5-fluorobenzenebutanoic acid (BMS-299897) alleviates Abeta1-42 seeding and short-term memory deficits in the Abeta25-35 mouse model of Alzheimer's disease. Eur J Pharmacol, 2013. 698(1-3): p. 193-9.
[5]. Goldstein, M.E., et al., Ex vivo occupancy of gamma-secretase inhibitors correlates with brain beta-amyloid peptide reduction in Tg2576 mice. J Pharmacol Exp Ther, 2007. 323(1): p. 102-8.
- Glucosamine sulfate
Catalog No.:BCN5981
CAS No.:29031-19-4
- Daun02
Catalog No.:BCC1518
CAS No.:290304-24-4
- 3-Epiglochidiol
Catalog No.:BCN5193
CAS No.:29028-10-2
- Fmoc-Gly-OH
Catalog No.:BCC3498
CAS No.:29022-11-5
- Benzoyl-DL-valine
Catalog No.:BCC8864
CAS No.:2901-80-6
- Methyl reserpate
Catalog No.:BCN3489
CAS No.:2901-66-8
- Boc-Phg-OH
Catalog No.:BCC3311
CAS No.:2900-27-8
- TRAM-34
Catalog No.:BCC1122
CAS No.:289905-88-0
- 3-(Benzylthio)-Propionic acid
Catalog No.:BCC2839
CAS No.:2899-66-3
- H-Methioninol
Catalog No.:BCC2721
CAS No.:2899-37-8
- H-Tryptophanol
Catalog No.:BCC2701
CAS No.:2899-29-8
- H-Trp-Oet.HCl
Catalog No.:BCC2673
CAS No.:2899-28-7
- Bis(2-carboxyethyl)isocyanurate
Catalog No.:BCC8880
CAS No.:2904-40-7
- Matricin
Catalog No.:BCC8209
CAS No.:29041-35-8
- Hexanorcucurbitacin D
Catalog No.:BCN7875
CAS No.:29065-05-2
- Pachymic acid
Catalog No.:BCN6347
CAS No.:29070-92-6
- 5-Hydroxy-3',4',7-trimethoxyflavone
Catalog No.:BCN5194
CAS No.:29080-58-8
- Eupteleasaponin I
Catalog No.:BCN7839
CAS No.:290809-29-9
- Avosentan
Catalog No.:BCC1387
CAS No.:290815-26-8
- Cimiracemoside D
Catalog No.:BCN2789
CAS No.:290821-39-5
- Glipizide
Catalog No.:BCC3785
CAS No.:29094-61-9
- Pinoresinol dimethyl ether
Catalog No.:BCN6767
CAS No.:29106-36-3
- Procyanidin B2
Catalog No.:BCN6315
CAS No.:29106-49-8
- Guanfacine
Catalog No.:BCC5180
CAS No.:29110-47-2
Studies on the pharmacokinetics and metabolism of a gamma-secretase inhibitor BMS-299897, and exploratory investigation of CYP enzyme induction.[Pubmed:19480557]
Xenobiotica. 2009 Jul;39(7):544-55.
BMS-299897 is a gamma-secretase inhibitor that was effective in reducing amyloid beta-peptide (A beta) in transgenic mice and guinea pigs. Therefore, pharmacokinetic and drug metabolism studies were conducted in animals to support its clinical development. The compound appeared to have low to intermediate total body clearance and was orally bioavailable (24-100%). The oral absorption of BMS-299897 from solid dosage forms appeared to be dissolution rate-limited. BMS-299897 was distributed into extravascular space (V(ss) >or= 1.3 l kg(-1)), including brain (brain-to-plasma ratio = 0.13-0.50). BMS-299897 appeared to be a P-glycoprotein (P-gp) substrate as the brain-to-plasma ratio was two-fold higher in the mdr1a knockout mouse as compared with the wild-type. Apparent autoinduction by BMS-299897 was observed in murine and rat efficacy and toxicity studies. In vitro, BMS-299897 was a weaker inducer of cytochrome P450 3A4 (CYP3A4) and a weaker transactivator of human pregnane X receptor (hPXR) as compared with rifampicin. Induction of human UGT1A and UGT2B was evaluated in primary human hepatocytes, but the results were inconclusive. A low potential for autoinduction in humans was predicted at a clinical dose of 250 mg and the prediction was consistent with the findings from a clinical multiple-dose study with BMS-299897 in probable Alzheimer's patients.
The gamma-secretase inhibitor 2-[(1R)-1-[(4-chlorophenyl)sulfonyl](2,5-difluorophenyl) amino]ethyl-5-fluorobenzenebutanoic acid (BMS-299897) alleviates Abeta1-42 seeding and short-term memory deficits in the Abeta25-35 mouse model of Alzheimer's disease.[Pubmed:23123349]
Eur J Pharmacol. 2013 Jan 5;698(1-3):193-9.
Alzheimer's disease pathomimetic toxicity could be induced in mice within one week after the intracerebroventricular (i.c.v.) injection of an aggregated preparation of the highly toxic and endogenous amyloid-beta fragment Abeta(25-35). It was recently reported that Abeta(25-35) also provokes a modification of APP processing with accumulation of endogenous Abeta(1-42). We here analyzed whether a gamma-secretase inhibitor, BMS-299897, attenuated this Abeta(25-35)-induced Abeta(1-42) seeding and toxicity. The compound was administered at 0.1-1 nmol/mouse, concomittantly with Abeta(25-35) (9 nmol) in male Swiss mice. After one week, the contents in Abeta(1-42) and Abeta(1-40), and the levels in lipid peroxidation were analyzed in the mouse hippocampus. Mice were submitted to spontaneous alternation, passive avoidance and object recognition to analyze their short- and long-term memory abilities. Abeta(25-35) increased Abeta(1-42) content (+240%) but failed to affect Abeta(1-40). BMS-299897 blocked the increase in Abeta(1-42) content and decreased Abeta(1-40) levels significantly. The compound did not affect Abeta(25-35)-induced increase in hippocampal lipid peroxidation. Behaviorally, BMS-299897 blocked the Abeta(25-35)-induced deficits in spontaneous alternation or novel object recognition, using a 1h intertrial time interval. BMS-299896 failed to affect the passive avoidance impairments or novel object recognition, using a 24h intertrial time interval. These results confirmed that Abeta(25-35) injection provoked an accumulation in endogenous Abeta(1-42), an effect blocked by gamma-secretase inhibition. This Abeta(1-42) accumulation marginally contributed to the toxicity or long-term memory deficits. However, since the seeded Abeta(1-42) affected short-term memory, the rapid Abeta(25-35) injection Alzheimer's disease model could be used to screen the activity of new secretase inhibitors.
In vitro and in vivo metabolism of a gamma-secretase inhibitor BMS-299897 and generation of active metabolites in milligram quantities with a microbial bioreactor.[Pubmed:17168689]
Curr Drug Metab. 2006 Dec;7(8):883-96.
BMS-299897 is a gamma-secretase inhibitor that has the potential for treatment of Alzheimer's disease. The metabolism of [(14)C]BMS-299897 was investigated in human liver microsomes, in rat, dog, monkey and human hepatocytes and in bile duct cannulated rats. Seven metabolites (M1-M7) were identified from in vitro and in vivo studies. LC-MS/MS analysis showed that M1 and M2 were regioisomeric acylglucuronide conjugates of BMS-299897. Metabolites M3, M4 and M6 were identified as monohydroxylated metabolites of BMS-299897 and M5 was identified as the dehydrogenated product of monooxygenated BMS-299897. In vivo, 52% of the radioactive dose was excreted in bile within 0-6 h from bile duct cannulated rats following a single oral dose of 15 mg/kg of [(14)C]BMS-299897. Glucuronide conjugates, M1 and M2 accounted for 80% of the total radioactivity in rat bile. In addition to M1 and M2, M7 was observed in rat bile which was identified as a glucuronide conjugate of an oxidative metabolite M5. For structure elucidation and pharmacological activity testing of the metabolites, ten microbial cultures were screened for their ability to metabolize BMS-299897 to form these metabolites. Among them, the fungus Cunninghamella elegans produced two major oxidative metabolites M3 and M4 that had the same HPLC retention time and mass spectral properties as those found in in vitro incubations. NMR analysis indicated that M3 and M4 were stereoisomers, with the hydroxyl group on the benzylic position. However, M3 and M4 were unstable and converted to their corresponding lactones readily. Based on x-ray analysis of the synthetically prepared lactone of M3, the stereochemistry of benzylic hydroxyl group was assigned as the R configuration. Both the hydroxy metabolites (M3 and M4) and the lactone of M3 showed gamma-secretase inhibition with IC(50) values similar to that of the parent compound. This study demonstrates the usefulness of microbial systems as bioreactors to generate metabolites of BMS-299897 in large quantities for structure elucidation and activity testing. This study also demonstrates the biotransformation profile of BMS-299897 is qualitatively similar across the species including rat, dog, monkey and human which provides a basis to support rat, dog and monkey as preclinical models for toxicological testing.
Liquid-liquid extraction of strongly protein bound BMS-299897 from human plasma and cerebrospinal fluid, followed by high-performance liquid chromatography/tandem mass spectrometry.[Pubmed:17204392]
J Pharm Biomed Anal. 2007 Apr 11;43(5):1728-36.
BMS-299897 is a gamma-secretase inhibitor that is being developed for the treatment of Alzheimer's disease. Liquid-liquid extraction (LLE), chromatographic/tandem mass spectrometry (LC/MS/MS) methods have been developed and validated for the quantitation of BMS-299897 in human plasma and cerebrospinal fluid (CSF). Both methods utilized (13)C6-BMS-299897, the stable label isotope analog, as the internal standard. For the human plasma extraction method, two incubation steps were required after the addition of 5 mM ammonium acetate and the internal standard in acetonitrile to release the analyte bound to proteins prior to LLE with toluene. For the human CSF extraction method, after the addition of 0.5 N HCl and the internal standard, CSF samples were extracted with toluene and no incubation was required. The organic layers obtained from both extraction methods were removed and evaporated to dryness. The residues were reconstituted and injected into the LC/MS/MS system. Chromatographic separation was achieved isocratically on a MetaChem C18 Hypersil BDS column (2.0 mm x 50 mm, 3 microm). The mobile phase contained 10 mM ammonium acetate pH 5 and acetonitrile. Detection was by negative ion electrospray tandem mass spectrometry. The standard curves ranged from 1 to 1000 ng/ml for human plasma and 0.25-100 ng/ml for human CSF. Both standard curves were fitted to a 1/x weighted quadratic regression model. For both methods, the intra-assay precision was within 8.2% CV, the inter-assay precision was within 5.4% CV, and assay accuracy was within +/-7.4% of the nominal values. The validation and sample analysis results demonstrated that both methods had acceptable precision and accuracy across the calibration ranges.
Ex vivo occupancy of gamma-secretase inhibitors correlates with brain beta-amyloid peptide reduction in Tg2576 mice.[Pubmed:17640949]
J Pharmacol Exp Ther. 2007 Oct;323(1):102-8.
Reduction of brain beta-amyloid peptide (Abeta) synthesis by gamma-secretase inhibitors is a promising approach for the treatment of Alzheimer's disease. However, measurement of central pharmacodynamic effects in the Alzheimer's disease patient will be a challenge. Determination of drug occupancy may facilitate the analysis of efficacy of gamma-secretase inhibitors in a clinical setting. In this study, the relationship of gamma-secretase site occupancy and brain Abeta40 reduction by gamma-secretase inhibitors was examined in Tg2576 mice. [3H](2R,3S)-2-Isobutyl-N1-((S)-1-methyl-2-oxo-5-phenyl-2,3-dihydro-1H-benzo[e][1, 4]diazepin-3-yl)-3-propylsuccinamide (IN973) was used as a gamma-secretase radioligand, since it has been shown to bind to gamma-secretase in rat, rhesus, and human brains with high affinity and specificity. We extended these findings by showing that [3H]IN973 bound to gamma-secretase in Tg2576 brains with an affinity, specificity, and regional localization very similar to the other species. To quantify gamma-secretase occupancy by gamma-secretase inhibitors, an ex vivo binding assay was developed using [3H]IN973 and frozen brain sections from drug-treated mice. Gamma-secretase occupancy and brain Abeta40 reduction were found to be highly correlated in animals dosed with either 2-[(1R)-1-[[4-chlorophenyl)-sulfonyl](2,5-difluorophenyl)amino] ethyl]-5-fluoro-benzenepropanoic acid (BMS-299897) or (S)-2-((S)-2-(3,5-difluorophenyl)-2-hydroxyacetamido)-N-((S,Z)-3-methyl-4-oxo-4,5 -dihydro-3H-benzo[d][1,2]diazepin-5-yl)propanamide (BMS-433796) over a wide range of doses and times postdose, with the exception of the earliest times postdose. This lag in Abeta40 response to gamma-secretase inhibition is probably related to the delayed clearance of previously produced Abeta40. The excellent correlation between brain Abeta40 and gamma-secretase occupancy suggests that a positron emission tomography ligand for gamma-secretase could be a valuable biomarker to determine whether gamma-secretase inhibitors bind to their target in humans.
Reductions in beta-amyloid concentrations in vivo by the gamma-secretase inhibitors BMS-289948 and BMS-299897.[Pubmed:15670587]
Biochem Pharmacol. 2005 Feb 15;69(4):689-98.
A primary pathological feature of Alzheimer's disease is beta-amyloid (Abeta)-containing plaques in brain and cerebral vasculature. Reductions in the formation of Abeta peptides by gamma-secretase inhibitors may be a viable therapy for reducing Abeta in Alzheimer's disease. Here we report on the effects of two orally active gamma-secretase inhibitors. BMS-289948 (4-chloro-N-(2,5-difluorophenyl)-N-((1R)-{4-fluoro-2-[3-(1H-imidazol-1-yl)propyl] phenyl}ethyl)benzenesulfonamide hydrochloride) and BMS-299897 (4-[2-((1R)-1-{[(4-chlorophenyl)sulfonyl]-2,5-difluoroanilino}ethyl)-5-fluorophen yl]butanoic acid) markedly reduced both brain and plasma Abeta(1-40) in APP-YAC mice with ED(50) values of 86 and 22 mg/kg per os (po), respectively, for BMS-289948, and 30 and 16 mg/kg po, respectively, for BMS-299897. Both compounds also dose-dependently increased brain concentrations of APP carboxy-terminal fragments, consistent with inhibition of gamma-secretase. BMS-289948 and BMS-299897 (100 mg/kg po) reduced brain and plasma Abeta(1-40) rapidly (within 20min) and maximally within 3 h. BMS-299897 also dose-dependently reduced cortical, cerebrospinal fluid (CSF), and plasma Abeta in guinea pigs with ED(50) values of 30 mg/kg intraperitoneally, without affecting CSF levels of alpha-sAPP. The reductions in cortical Abeta correlated significantly with the reductions in both plasma (r(2) = 0.77) and CSF (r(2) = 0.61) Abeta. The decreases in Abeta were apparent at 3 and 6 h post-administration of BMS-299897, but not at 12h. These results demonstrate that BMS-289948 and BMS-299897 are orally bioavailable, functional gamma-secretase inhibitors with the ability to markedly reduce Abeta peptide concentrations in APP-YAC transgenic mice and in guinea pigs. These compounds may be useful pharmacologically for examining the effects of reductions in beta-amyloid peptides in both animal models and in Alzheimer's disease.
Dynamics of {beta}-amyloid reductions in brain, cerebrospinal fluid, and plasma of {beta}-amyloid precursor protein transgenic mice treated with a {gamma}-secretase inhibitor.[Pubmed:15452193]
J Pharmacol Exp Ther. 2005 Feb;312(2):635-43.
gamma-Secretase inhibitors are one promising approach to the development of a therapeutic for Alzheimer's disease (AD). gamma-Secretase inhibitors reduce brain beta-amyloid peptide (Abeta), which is believed to be a major contributor in the etiology of AD. Transgenic mice overexpressing the human beta-amyloid precursor protein (APP) are valuable models to examine the dynamics of Abeta changes with gamma-secretase inhibitors in plaque-free and plaque-bearing animals. BMS-299897 2-[(1R)-1-[[(4-chlorophenyl)sulfony](2,5-difluorophenyl)amino]ethyl]-5-fluorobenz enepropanoic acid, a gamma-secretase inhibitor, showed dose- and time dependent reductions of Abeta in brain, cerebrospinal fluid (CSF), and plasma in young transgenic mice, with a significant correlation between brain and CSF Abeta levels. Because CSF and brain interstitial fluid are distinct compartments in composition and location, this correlation could not be assumed. In contrast, aged transgenic mice with large accumulations of Abeta in plaques showed reductions in CSF Abeta in the absence of measurable changes in plaque Abeta in the brain after up to 2 weeks of treatment. Hence, CSF Abeta levels were a valuable measure of gamma-secretase activity in the central nervous system in either the presence or absence of plaques. Transgenic mice were also used to examine potential side effects due to Notch inhibition. BMS-299897 was 15-fold more effective at preventing the cleavage of APP than of Notch in vitro. No changes in the maturation of CD8(+) thymocytes or of intestinal goblet cells were observed in mice treated with BMS-299897, showing that it is possible for gamma-secretase inhibitors to reduce brain Abeta without causing Notch-mediated toxicity.


