DAPK Substrate PeptideSynthetic peptide substrate for DAPK CAS# 386769-53-5 |
- Limonin
Catalog No.:BCN6057
CAS No.:1180-71-8
- Fosamprenavir Calcium Salt
Catalog No.:BCC1581
CAS No.:226700-81-8
- HIV-1 integrase inhibitor
Catalog No.:BCC1618
CAS No.:544467-07-4
- BMS-626529
Catalog No.:BCC1427
CAS No.:701213-36-7
- HIV-1 integrase inhibitor 2
Catalog No.:BCC1619
CAS No.:957890-42-5
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 386769-53-5 | SDF | Download SDF |
| PubChem ID | 90479796 | Appearance | Powder |
| Formula | C70H115N25O17 | M.Wt | 1578.82 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 1 mg/ml in water | ||
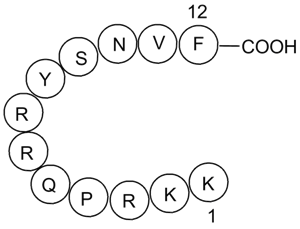
| Sequence | KKRPQRRYSNVF | ||
| Chemical Name | (2S)-2-[[(2S)-2-[[(2S)-4-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-5-amino-2-[[(2S)-1-[(2S)-2-[[(2S)-6-amino-2-[[(2S)-2,6-diaminohexanoyl]amino]hexanoyl]amino]-5-(diaminomethylideneamino)pentanoyl]pyrrolidine-2-carbonyl]amino]-5-oxopentanoyl]amino]-5-(diaminomethylideneamino)pentanoyl]amino]-5-(diaminomethylideneamino)pentanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-3-hydroxypropanoyl]amino]-4-oxobutanoyl]amino]-3-methylbutanoyl]amino]-3-phenylpropanoic acid | ||
| SMILES | CC(C)C(C(=O)NC(CC1=CC=CC=C1)C(=O)O)NC(=O)C(CC(=O)N)NC(=O)C(CO)NC(=O)C(CC2=CC=C(C=C2)O)NC(=O)C(CCCN=C(N)N)NC(=O)C(CCCN=C(N)N)NC(=O)C(CCC(=O)N)NC(=O)C3CCCN3C(=O)C(CCCN=C(N)N)NC(=O)C(CCCCN)NC(=O)C(CCCCN)N | ||
| Standard InChIKey | DTEBSHTZDDUPBW-RHBFSANCSA-N | ||
| Standard InChI | InChI=1S/C70H115N25O17/c1-38(2)55(65(109)92-50(67(111)112)35-39-14-4-3-5-15-39)94-62(106)49(36-54(75)99)91-63(107)51(37-96)93-61(105)48(34-40-22-24-41(97)25-23-40)90-59(103)45(19-11-31-83-69(78)79)86-57(101)44(18-10-30-82-68(76)77)87-60(104)46(26-27-53(74)98)88-64(108)52-21-13-33-95(52)66(110)47(20-12-32-84-70(80)81)89-58(102)43(17-7-9-29-72)85-56(100)42(73)16-6-8-28-71/h3-5,14-15,22-25,38,42-52,55,96-97H,6-13,16-21,26-37,71-73H2,1-2H3,(H2,74,98)(H2,75,99)(H,85,100)(H,86,101)(H,87,104)(H,88,108)(H,89,102)(H,90,103)(H,91,107)(H,92,109)(H,93,105)(H,94,106)(H,111,112)(H4,76,77,82)(H4,78,79,83)(H4,80,81,84)/t42-,43-,44-,45-,46-,47-,48-,49-,50-,51-,52-,55-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Synthetic peptide substrate for death associated protein kinase (DAPK) (Km = 9 μM). |

DAPK Substrate Peptide Dilution Calculator

DAPK Substrate Peptide Molarity Calculator

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Synthetic peptide substrate for death associated protein kinase (DAPK) (Km = 9 μM).
- Desvenlafaxine Succinate
Catalog No.:BCC5048
CAS No.:386750-22-7
- Neodiosmin
Catalog No.:BCN8337
CAS No.:38665-01-9
- Triptonide
Catalog No.:BCN5924
CAS No.:38647-11-9
- Tripdiolide
Catalog No.:BCN5985
CAS No.:38647-10-8
- Benzoylpaeoniflorin
Catalog No.:BCN6293
CAS No.:38642-49-8
- Lasiodonin
Catalog No.:BCN7156
CAS No.:38602-52-7
- Chloroprocaine HCl
Catalog No.:BCC5556
CAS No.:3858-89-7
- Prostaglandin F2α
Catalog No.:BCC7889
CAS No.:38562-01-5
- Oxotremorine M
Catalog No.:BCC6920
CAS No.:3854-04-4
- Daphnicyclidin H
Catalog No.:BCN7080
CAS No.:385384-29-2
- Daphnicyclidin F
Catalog No.:BCN6400
CAS No.:385384-26-9
- Daphnicyclidin D
Catalog No.:BCN7081
CAS No.:385384-24-7
- Groenlandicine
Catalog No.:BCN8189
CAS No.:38691-95-1
- Preskimmianine
Catalog No.:BCN6667
CAS No.:38695-41-9
- Seneciphylline N-oxide
Catalog No.:BCN5439
CAS No.:38710-26-8
- 3-Epibetulinic acid
Catalog No.:BCN8531
CAS No.:38736-77-5
- Triptolide
Catalog No.:BCN5984
CAS No.:38748-32-2
- Worenine
Catalog No.:BCN2557
CAS No.:38763-29-0
- Swazine
Catalog No.:BCN2143
CAS No.:38763-74-5
- Cucurbitacin D
Catalog No.:BCN2355
CAS No.:3877-86-9
- Tetrahydroisocucurbitacin I
Catalog No.:BCN7874
CAS No.:3877-89-2
- Muristerone A
Catalog No.:BCC2397
CAS No.:38778-30-2
- Tectoruside
Catalog No.:BCN8262
CAS No.:38784-73-5
- Tandutinib (MLN518)
Catalog No.:BCC4499
CAS No.:387867-13-2
DAPK-HSF1 interaction as a positive-feedback mechanism stimulating TNF-induced apoptosis in colorectal cancer cells.[Pubmed:25380824]
J Cell Sci. 2014 Dec 15;127(Pt 24):5273-87.
Death-associated protein kinase (DAPK) is a serine-threonine kinase with tumor suppressor function. Previously, we demonstrated that tumor necrosis factor (TNF) induced DAPK-mediated apoptosis in colorectal cancer. However, the protein-protein interaction network associated with TNF-DAPK signaling still remains unclear. We identified HSF1 as a new DAPK phosphorylation target in response to low concentrations of TNF and verified a physical interaction between DAPK and HSF1 both in vitro and in vivo. We show that HSF1 binds to the DAPK promoter. Transient overexpression of HSF1 protein led to an increase in DAPK mRNA level and consequently to an increase in the amount of apoptosis. By contrast, treatment with a DAPK-specific inhibitor as well as DAPK knockdown abolished the phosphorylation of HSF1 at Ser230 (pHSF1(Ser230)). Furthermore, translational studies demonstrated a positive correlation between DAPK and pHSF1(Ser230) protein expression in human colorectal carcinoma tissues. Taken together, our data define a novel link between DAPK and HSF1 and highlight a positive-feedback loop in DAPK regulation under mild inflammatory stress conditions in colorectal tumors. For the first time, we show that under TNF the pro-survival HSF1 protein can be redirected to a pro-apoptotic program.
KLHL39 suppresses colon cancer metastasis by blocking KLHL20-mediated PML and DAPK ubiquitination.[Pubmed:25619834]
Oncogene. 2015 Oct 1;34(40):5141-51.
Cullin 3 (Cul3)-family ubiquitin ligases use the BTB-domain-containing proteins for the recruitment of substrates, but the regulation of this family of ubiquitin ligases has not been completely understood. KLHL20 is a BTB-family protein and targets tumor suppressor promyelocytic leukemia protein (PML) and death-associated protein kinase (DAPK) to its kelch-repeat domain for ubiquitination and degradation. Here, we show that another BTB-kelch protein KLHL39 is recruited to the substrate-binding domain of KLHL20 but is not a substrate of Cul3-KLHL20 complex. Interestingly, KLHL39 does not bind Cul3 because of the absence of certain conserved residues in the BTB domain. Instead, KLHL39 blocks KLHL20-mediated ubiquitination of PML and DAPK by disrupting the binding of these substrates to KLHL20 as well as the binding of KLHL20 to Cul3. Through the two mechanisms, KLHL39 increases the stability of PML and DAPK. In human colon cancers, downregulations of KLHL39, PML and DAPK are associated with metastatic progression. Furthermore, preclinical data indicate that KLHL39 promotes colon cancer migration, invasion and survival in vitro and metastasis in vivo through a PML- and DAPK-dependent mechanism. Our study identifies KLHL39 as a negative regulator of Cul3-KLHL20 ubiquitin ligase and reveals a role of KLHL39-mediated PML and DAPK stabilization in colon cancer metastasis.
A PEF/Y substrate recognition and signature motif plays a critical role in DAPK-related kinase activity.[Pubmed:24440081]
Chem Biol. 2014 Feb 20;21(2):264-73.
Knowledge about protein kinase substrate preferences is biased toward residues immediately adjacent to the site of phosphorylation. By a combined structural, biochemical, and cellular approach, we have discovered an unexpected substrate recognition element with the consensus sequence PEF/Y in the tumor suppressor death-associated protein kinase 1. This motif can be effectively blocked by a specific pseudosubstrate-type interaction with an autoregulatory domain of this kinase. In this arrangement, the central PEF/Y glutamate interacts with a conserved arginine distant to the phosphorylation site in sequence and structure. We also demonstrate that the element is crucial for kinase activity regulation and substrate recognition. The PEF/Y motif distinguishes close death-associated protein kinase relatives from canonical calcium/calmodulin-dependent protein kinases. Insight into this signature and mode of action offers new opportunities to identify specific small molecule inhibitors in PEF/Y-containing protein kinases.
DAPK and cytoskeleton-associated functions.[Pubmed:24166137]
Apoptosis. 2014 Feb;19(2):329-38.
Death-associated protein kinase (DAPK) undergoes activation in response to various death stimuli, and they have been associated with an increase in DAPK catalytic activity. One of the most prominent features of DAPK-induced cell death is the effect on the cytoskeleton, including loss of matrix attachment, and membrane blebbing. One known cytoskeletal-associated substrate of DAPK is the myosin-II light chain, phosphorylated by DAPK on Ser(19), thus stabilizing actin stress fibres. Moreover, paxillin, a component of focal adhesions, was found to be localized in close proximity to the tips of the DAPK-positive filaments, indicating that stress fibres containing DAPK extend to focal contacts. Forced expression of DAPK in multiple cell types results in morphological changes such as cell rounding, membrane blebbing, shrinking and detachment. During directed migration, DAPK functions as a potent inhibitor of cell polarization, as evidenced by its perturbation of the formation of static protrusion at the leading edge. Furthermore, DAPK inhibits random migration by suppressing directional persistence. One of the studies considered DAPK as an anoikis inducer. Others showed that DAP-kinase inhibits the activities of cell surface integrins by converting them into an inactive conformation. Biochemical experiments have established the DAPK binding to Syntaxin1 and its subsequent phosphorylation at Ser(188) in a Ca(2+) dependent manner. This phosphorylation event has been shown to decrease the binding of Syntaxin to MUNC18-1, a protein critically involved in synaptic vesicle docking. Here, we have investigated the structural interactions that modulate DAPK phosphorylation with Syntaxin and its functional role in binding to the MUNC18-1 to regulate vesicle docking. This review will summarize our current knowledge of the role of DAPK on cytoskeleton reorganization and report the mechanisms that regulate these changes.
The DAP-kinase interactome.[Pubmed:24220855]
Apoptosis. 2014 Feb;19(2):316-28.
DAP-kinase (DAPK) is a Ca(2+)/calmodulin regulated Ser/Thr kinase that activates a diverse range of cellular activities. It is subject to multiple layers of regulation involving both intramolecular signaling, and interactions with additional proteins, including other kinases and phosphatases. Its protein stability is modulated by at least three distinct ubiquitin-dependent systems. Like many kinases, DAPK participates in several signaling cascades, by phosphorylating additional kinases such as ZIP-kinase and protein kinase D (PKD), or Pin1, a phospho-directed peptidyl-prolyl isomerase that regulates the function of many phosphorylated proteins. Other substrate targets have more direct cellular effects; for example, phosphorylation of the myosin II regulatory chain and tropomyosin mediate some of DAPK's cytoskeletal functions, including membrane blebbing during cell death and cell motility. DAPK induces distinct death pathways of apoptosis, autophagy and programmed necrosis. Among the substrates implicated in these processes, phosphorylation of PKD, Beclin 1, and the NMDA receptor has been reported. Interestingly, not all cellular effects are mediated by DAPK's catalytic activity. For example, by virtue of protein-protein interactions alone, DAPK activates pyruvate kinase isoform M2, the microtubule affinity regulating kinases and inflammasome protein NLRP3, to promote glycolysis, influence microtubule dynamics, and enhance interleukin-1beta production, respectively. In addition, a number of other substrates and interacting proteins have been identified, the physiological significance of which has not yet been established. All of these substrates, effectors and regulators together comprise the DAPK interactome. By presenting the components of the interactome network, this review will clarify both the mechanisms by which DAPK function is regulated, and by which it mediates its various cellular effects.
A protein kinase associated with apoptosis and tumor suppression: structure, activity, and discovery of peptide substrates.[Pubmed:11483604]
J Biol Chem. 2001 Oct 19;276(42):38956-65.
Death-associated protein kinase (DAPK) has been implicated in apoptosis and tumor suppression, depending on cellular conditions, and associated with mechanisms of disease. However, DAPK has not been characterized as an enzyme due to the lack of protein or peptide substrates. Therefore, we determined the structure of DAPK catalytic domain, used a homology model of docked peptide substrate, and synthesized positional scanning substrate libraries in order to discover peptide substrates with K(m) values in the desired 10 microm range and to obtain knowledge about the preferences of DAPK for phosphorylation site sequences. Mutagenesis of DAPK catalytic domain at amino acids conserved among protein kinases or unique to DAPK provided a link between structure and activity. An enzyme assay for DAPK was developed and used to measure activity in adult brain and monitor protein purification based on the physical and chemical properties of the open reading frame of the DAPK cDNA. The results allow insight into substrate preferences and regulation of DAPK, provide a foundation for proteomic investigations and inhibitor discovery, and demonstrate the utility of the experimental approach, which can be extended potentially to kinase open reading frames identified by genome sequencing projects or functional genetics screens and lacking a known substrate.


