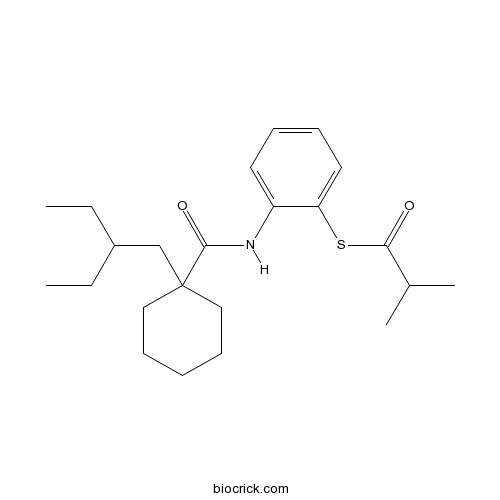
Dalcetrapib (JTT-705, RO4607381)rhCETP inhibitor CAS# 211513-37-0 |
- Evacetrapib (LY2484595)
Catalog No.:BCC2329
CAS No.:1186486-62-3
- Torcetrapib
Catalog No.:BCC2330
CAS No.:262352-17-0
- Anacetrapib (MK-0859)
Catalog No.:BCC2327
CAS No.:875446-37-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 211513-37-0 | SDF | Download SDF |
| PubChem ID | 6918540 | Appearance | Powder |
| Formula | C23H35NO2S | M.Wt | 389.59 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | JTT-705; RO4607381 | ||
| Solubility | DMSO : ≥ 50 mg/mL (128.34 mM) H2O : < 0.1 mg/mL (insoluble) *"≥" means soluble, but saturation unknown. | ||
| Chemical Name | S-[2-[[1-(2-ethylbutyl)cyclohexanecarbonyl]amino]phenyl] 2-methylpropanethioate | ||
| SMILES | CCC(CC)CC1(CCCCC1)C(=O)NC2=CC=CC=C2SC(=O)C(C)C | ||
| Standard InChIKey | YZQLWPMZQVHJED-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C23H35NO2S/c1-5-18(6-2)16-23(14-10-7-11-15-23)22(26)24-19-12-8-9-13-20(19)27-21(25)17(3)4/h8-9,12-13,17-18H,5-7,10-11,14-16H2,1-4H3,(H,24,26) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Dalcetrapib is an inhibitor of rhCETP with IC50 value of 0.2 μM. | |||||
| Targets | rhCETP | |||||
| IC50 | 0.2 μM | |||||

Dalcetrapib (JTT-705, RO4607381) Dilution Calculator

Dalcetrapib (JTT-705, RO4607381) Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.5668 mL | 12.834 mL | 25.668 mL | 51.336 mL | 64.17 mL |
| 5 mM | 0.5134 mL | 2.5668 mL | 5.1336 mL | 10.2672 mL | 12.834 mL |
| 10 mM | 0.2567 mL | 1.2834 mL | 2.5668 mL | 5.1336 mL | 6.417 mL |
| 50 mM | 0.0513 mL | 0.2567 mL | 0.5134 mL | 1.0267 mL | 1.2834 mL |
| 100 mM | 0.0257 mL | 0.1283 mL | 0.2567 mL | 0.5134 mL | 0.6417 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
IC50: 6 μM for CETP inhibition in human plasma
Cholesteryl ester transfer protein (CETP) is a plasma protein that transfers neutral lipids among the lipoproteins. Its most important action is the exchange of cholesteryl esters in high-density lipoprotein (HDL) for triglycerides in very low-density lipoprotein. Thus, CETP is a potentially atherogenic protein, and its atherogenicity has been supported by many studies. Dalcetrapib is a novel inhibitors of CETP.
In vitro: Dalcetrapib achieved 50% inhibition of CETP activity in human plasma at a concentration of 6 μM. The mechanism of action was considered to involve the formation of a disulfide bond between the thiol form of Dalcetrapib and the cysteine residue at position 13 (Cys13) of CETP. [1].
In vivo: Dalcetrapib achieved 95% inhibition of CETP activity in male Japanese white rabbits at an oral dose of 30 mg/kg. It increased the plasma HDL cholesterol level by 27% and 54%, respectively, when given at oral doses of 30 or 100 mg/kg once a day for 3 days to male Japanese white rabbits [1].
Clinical trials: In a phase II study, Dalcetrapib showed no evidence of a pathological effect related to the arterial wall over 24 months. Moreover, this trial suggests possible beneficial vascular effects of dalcetrapib, including the reduction in total vessel enlargement over 24 months, but long-term safety and clinical outcomes efficacy of dalcetrapib need to be analysed [2].
References:
[1] Shinkai H, Maeda K, Yamasaki T, Okamoto H, Uchida I. bis(2-(Acylamino)phenyl) disulfides, 2-(acylamino)benzenethiols, and S-(2-(acylamino)phenyl) alkanethioates as novel inhibitors of cholesteryl ester transfer protein. J Med Chem. 2000;43(19):3566-72.
[2] Fayad ZA, Mani V, Woodward M, Kallend D, Abt M, Burgess T, Fuster V, Ballantyne CM, Stein EA, Tardif JC, Rudd JH, Farkouh ME, Tawakol A. Safety and efficacy of dalcetrapib on atherosclerotic disease using novel non-invasive multimodality imaging (dal-PLAQUE): a randomised clinical trial. Lancet. 2011;378(9802):1547-59.
- Dendrobine
Catalog No.:BCN5923
CAS No.:2115-91-5
- Astrocasine
Catalog No.:BCN2150
CAS No.:2114-92-3
- R18
Catalog No.:BCC2383
CAS No.:211364-78-2
- m-Chlorophenylbiguanide hydrochloride
Catalog No.:BCC6650
CAS No.:2113-05-5
- 9,17-Octadecadiene-12,14-diyne-1,11,16-triol
Catalog No.:BCN1497
CAS No.:211238-60-7
- Rubranol
Catalog No.:BCN4917
CAS No.:211126-61-3
- Sobetirome
Catalog No.:BCC1957
CAS No.:211110-63-3
- Marsformoxide B
Catalog No.:BCN6687
CAS No.:2111-46-8
- SB 265610
Catalog No.:BCC5936
CAS No.:211096-49-0
- Mahanimbine
Catalog No.:BCN3174
CAS No.:21104-28-9
- BMY 7378
Catalog No.:BCC5063
CAS No.:21102-95-4
- CART (62-76) (rat, human)
Catalog No.:BCC6008
CAS No.:210978-19-1
- WHI-P154
Catalog No.:BCC2202
CAS No.:211555-04-3
- WHI-P97
Catalog No.:BCC2056
CAS No.:211555-05-4
- WHI-P180
Catalog No.:BCC3928
CAS No.:211555-08-7
- Nudicaucin B
Catalog No.:BCN7843
CAS No.:211557-36-7
- Picroside IV
Catalog No.:BCN6533
CAS No.:211567-04-3
- 2',4',5'-Trimethoxy-2'',2''-dimethylpyrano[5'',6'':6,7]isoflavone
Catalog No.:BCN1496
CAS No.:211799-56-3
- Nudicaucin A
Catalog No.:BCN7842
CAS No.:211815-97-3
- Flumorph
Catalog No.:BCC5467
CAS No.:211867-47-9
- Gliclazide
Catalog No.:BCC5002
CAS No.:21187-98-4
- BIBR 953 (Dabigatran, Pradaxa)
Catalog No.:BCC2139
CAS No.:211914-51-1
- BIBR-1048
Catalog No.:BCC3738
CAS No.:211915-06-9
- 1,3,7-Trihydroxy-2-methoxyxanthone
Catalog No.:BCN7549
CAS No.:211948-69-5
Safety and tolerability of dalcetrapib (RO4607381/JTT-705): results from a 48-week trial.[Pubmed:20097702]
Eur Heart J. 2010 Feb;31(4):480-8.
AIMS: Co-primary objectives were to evaluate dalcetrapib (JTT-705/RO4607381), which targets cholesteryl ester transfer protein (CETP), effects on high-density lipoprotein cholesterol (HDL-C) in participants with coronary heart disease or risk equivalents and to evaluate potential changes in mesenteric lymph nodes. METHODS AND RESULTS: Double-blind trial with participants randomized (2:1) to dalcetrapib 900 mg/day (higher than 600 mg phase III dose) or placebo, both with atorvastatin, for 24 weeks (n = 135; one without post-baseline efficacy data was excluded from intent-to-treat population); a subset continued for 24-week extension (n = 77). Lipid changes and safety parameters were assessed. Mesenteric lymph nodes were evaluated by magnetic resonance imaging. Dalcetrapib increased HDL-C (33.4%, Week 24; 33.8%, Week 48), decreased CETP activity (-53.5%, Week 24; -56.5%, Week 48), and increased apolipoprotein A-I (11.4%, Week 24; 16.4%, Week 48). Dalcetrapib showed no clinically relevant differences vs. placebo in adverse events, laboratory parameters including aldosterone, electrocardiograms, and vital signs including blood pressure (BP). Dalcetrapib had no measurable, clinically relevant effect on lymph node size. CONCLUSION: Dalcetrapib 900 mg administered for up to 48 weeks showed no clinically relevant changes in lymph nodes, BP, or other safety parameters. Dalcetrapib effectively increased HDL-C over 48 weeks of treatment.
Dalcetrapib: JTT 705; JTT-705; R 1658; R1658; RG1658; RO 4607381; RO4607381.[Pubmed:20509713]
Drugs R D. 2010;10(1):33-6.
Roche and Japan Tobacco are in a licensing agreement to develop and commercialize dalcetrapib, a cholesteryl ester transfer protein (CETP) inhibitor to slow or prevent atherosclerosis. This drug is currently in phase III development. This review discusses the development history and scientific profile of this new compound.


