GDC-mTOR inhibitorMTOR inhibitor CAS# 1207358-59-5 |
- mGlu2 agonist
Catalog No.:BCC1745
CAS No.:1311385-32-6
- LY341495
Catalog No.:BCC1724
CAS No.:201943-63-7
- MPEP Hydrochloride
Catalog No.:BCC1777
CAS No.:219911-35-0
- CPPHA
Catalog No.:BCC1501
CAS No.:693288-97-0
- Dipraglurant
Catalog No.:BCC1531
CAS No.:872363-17-2
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1207358-59-5 | SDF | Download SDF |
| PubChem ID | 59239114 | Appearance | Powder |
| Formula | C25H30N8O2 | M.Wt | 474.56 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 50 mg/mL (105.36 mM; Need ultrasonic) | ||
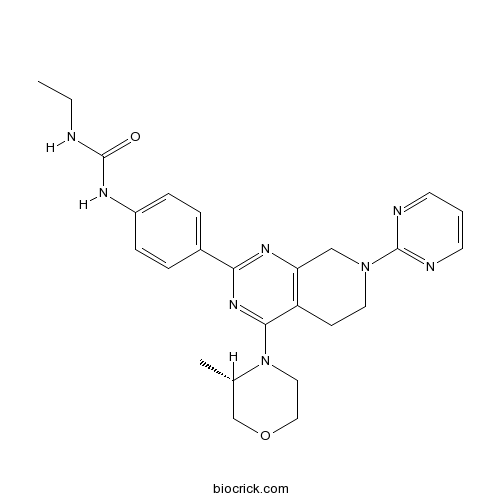
| Chemical Name | 1-ethyl-3-[4-[4-[(3S)-3-methylmorpholin-4-yl]-7-pyrimidin-2-yl-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-2-yl]phenyl]urea | ||
| SMILES | CCNC(=O)NC1=CC=C(C=C1)C2=NC3=C(CCN(C3)C4=NC=CC=N4)C(=N2)N5CCOCC5C | ||
| Standard InChIKey | WQBAZXIQANTUOY-KRWDZBQOSA-N | ||
| Standard InChI | InChI=1S/C25H30N8O2/c1-3-26-25(34)29-19-7-5-18(6-8-19)22-30-21-15-32(24-27-10-4-11-28-24)12-9-20(21)23(31-22)33-13-14-35-16-17(33)2/h4-8,10-11,17H,3,9,12-16H2,1-2H3,(H2,26,29,34)/t17-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | mTOR-IN-1 is a remarkably selective mTOR inhibitor with a Ki of 1.5 nM. mTOR-IN-1 suppresses mTORC1 and mTORC2 in cellular and in vivo pharmacokinetic (PK)/pharmacodynamic (PD) experiments.In Vitro:mTOR-IN-1 (Compound 12i) inhibits mTOR with a Ki of 1.5 nM, 500-fold selectivity over closely related PI3 kinases. mTOR-IN-1 inhibits NCI-PC3 and MCF7neo/Her2 cells proliferation with IC50s of 150 nM and 57 nM, respectively[2].In Vivo:mTOR-IN-1 (Compound 8h) has high free plasma clearance in both mice (1818 mL/min/kg) and rats (1538 mL/min/kg in rat) [1]. mTOR-IN-1 (Compounds 12i) is selected for this study due to its potency, selectivity, and favorable mouse PK profile. Plasma levels of mTOR-IN-1 6 h following oral administration in PC3 tumor-bearing mice along with the fold decreases of phosphorylated mTORC1 and -2 substrates relative to time-matched vehicle controls. mTOR-IN-1 has moderate terminal elimination half-life (t1/2=1.7 h for mouse(1 mg/kg, iv)). mTOR-IN-1 achieves tumor stasis at the highest 200 mg/kg/day dose examined, which appears to also be approaching the limit of tolerability for this molecule[2]. References: | |||||

GDC-mTOR inhibitor Dilution Calculator

GDC-mTOR inhibitor Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.1072 mL | 10.5361 mL | 21.0722 mL | 42.1443 mL | 52.6804 mL |
| 5 mM | 0.4214 mL | 2.1072 mL | 4.2144 mL | 8.4289 mL | 10.5361 mL |
| 10 mM | 0.2107 mL | 1.0536 mL | 2.1072 mL | 4.2144 mL | 5.268 mL |
| 50 mM | 0.0421 mL | 0.2107 mL | 0.4214 mL | 0.8429 mL | 1.0536 mL |
| 100 mM | 0.0211 mL | 0.1054 mL | 0.2107 mL | 0.4214 mL | 0.5268 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Description: IC50 Value: 1.5nM (Ki) GDC-mTOR inhibitor is a potent and selective ATP-competitive inhibitor of mTOR. It's derivative drug, GDC-0349, demonstrates pathway modulation and dose-dependent efficacy in mouse xenograft cancer models. Abstract: Selective inhibitors of mammalian target of rapamycin (mTOR) kinase based upon saturated heterocycles fused to a pyrimidine core were designed and synthesized. Each series produced compounds with Ki < 10 nM for the mTOR kinase and >500-fold selectivity over closely related PI3 kinases. This potency translated into strong pathway inhibition, as measured by phosphorylation of mTOR substrate proteins and antiproliferative activity in cell lines with a constitutively active PI3K pathway. Two compounds exhibiting suitable mouse PK were profiled in in vivo tumor models and were shown to suppress mTORC1 and mTORC2 signaling for over 12 h when dosed orally. Both compounds were additionally shown to suppress tumor growth in vivo in a PC3 prostate cancer model over a 14 day study.
- BI-847325
Catalog No.:BCC6511
CAS No.:1207293-36-4
- NVS-CRF38
Catalog No.:BCC8059
CAS No.:1207258-55-6
- 12alpha-Hydroxyevodol
Catalog No.:BCN6102
CAS No.:120722-04-5
- Sarcandrolide D
Catalog No.:BCN6621
CAS No.:1207185-03-2
- Scutebata G
Catalog No.:BCN6101
CAS No.:1207181-63-2
- Scutebata F
Catalog No.:BCN6100
CAS No.:1207181-62-1
- Scutebata E
Catalog No.:BCN6099
CAS No.:1207181-61-0
- Scutebata C
Catalog No.:BCN6098
CAS No.:1207181-59-6
- Scutebata B
Catalog No.:BCN6097
CAS No.:1207181-58-5
- Scutebata A
Catalog No.:BCN6096
CAS No.:1207181-57-4
- Psidial A
Catalog No.:BCN6095
CAS No.:1207181-35-8
- WAY 208466 dihydrochloride
Catalog No.:BCC7807
CAS No.:1207064-61-6
- GDC-0349
Catalog No.:BCC1094
CAS No.:1207360-89-1
- BMN-673 8R,9S
Catalog No.:BCC1422
CAS No.:1207456-00-5
- BMN 673
Catalog No.:BCC2205
CAS No.:1207456-01-6
- 5-OMe-UDP trisodium salt
Catalog No.:BCC6153
CAS No.:1207530-98-0
- LDV FITC
Catalog No.:BCC6229
CAS No.:1207610-07-8
- 3,2'-Epilarixinol
Catalog No.:BCN6496
CAS No.:1207671-28-0
- Huperzine A
Catalog No.:BCN1058
CAS No.:120786-18-7
- Gynosaponin I
Catalog No.:BCN4078
CAS No.:1207861-69-5
- Quassidine B
Catalog No.:BCN7022
CAS No.:1207862-37-0
- CaMKII-IN-1
Catalog No.:BCC5530
CAS No.:1208123-85-6
- VU 0365114
Catalog No.:BCC6164
CAS No.:1208222-39-2
- Ketone Ester
Catalog No.:BCC1677
CAS No.:1208313-97-6
A Cascade of Redox Reactions Generates Complexity in the Biosynthesis of the Protein Phosphatase-2 Inhibitor Rubratoxin A.[Pubmed:28370936]
Angew Chem Int Ed Engl. 2017 Apr 18;56(17):4782-4786.
Redox modifications are key complexity-generating steps in the biosynthesis of natural products. The unique structure of rubratoxin A (1), many of which arise through redox modifications, make it a nanomolar inhibitor of protein phosphatase 2A (PP2A). We identified the biosynthetic pathway of 1 and completely mapped the enzymatic sequence of redox reactions starting from the nonadride 5. Six redox enzymes are involved, including four alpha-ketoglutarate- and iron(II)-dependent dioxygenases that hydroxylate four sp(3) carbons; one flavin-dependent dehydrogenase that is involved in formation of the unsaturated lactone; and the ferric-reductase-like enzyme RbtH, which regioselectively reduces one of the maleic anhydride moieties in rubratoxin B to the gamma-hydroxybutenolide that is critical for PP2A inhibition. RbtH is proposed to perform sequential single-electron reductions of the maleic anhydride using electrons derived from NADH and transferred through a ferredoxin and ferredoxin reductase pair.
Phase 2 studies of oral hypoxia-inducible factor prolyl hydroxylase inhibitor FG-4592 for treatment of anemia in China.[Pubmed:28371815]
Nephrol Dial Transplant. 2017 Aug 1;32(8):1373-1386.
Background: FG-4592 (roxadustat) is an oral hypoxia-inducible factor (HIF) prolyl hydroxylase inhibitor (HIF-PHI) promoting coordinated erythropoiesis through the transcription factor HIF. Two Phase 2 studies were conducted in China to explore the safety and efficacy of FG-4592 (USAN name: roxadustat, CDAN name: ), a HIF-PHI, in patients with anemia of chronic kidney disease (CKD), both patients who were dialysis-dependent (DD) and patients who were not dialysis-dependent (NDD). Methods: In the NDD study, 91 participants were randomized to low (1.1-1.75 mg/kg) or high (1.50-2.25 mg/kg) FG-4592 starting doses or to placebo. In the DD study, 87 were enrolled to low (1.1-1.8 mg/kg), medium (1.5-2.3 mg/kg) and high (1.7-2.3 mg/kg) starting FG-4592 doses or to continuation of epoetin alfa. In both studies, only oral iron supplementation was allowed. Results: In the NDD study, hemoglobin (Hb) increase >/=1 g/dL from baseline was achieved in 80.0% of subjects in the low-dose cohort and 87.1% in the high-dose cohort, versus 23.3% in the placebo arm (P < 0.0001, both). In the DD study, 59.1%, 88.9% (P = 0.008) and 100% (P = 0.0003) of the low-, medium- and high-dose subjects maintained their Hb levels after 5- and 6-weeks versus 50% of the epoetin alfa-treated subjects. In both studies, significant reductions in cholesterol were noted in FG-4592-treated subjects, with stability or increases in serum iron, total iron-binding capacity (TIBC) and transferrin (without intravenous iron administration). In the NDD study, hepcidin levels were significantly reduced across all FG-4592-treated arms as compared with no change in the placebo arm. In the DD study, hepcidin levels were also reduced in a statistically significant dose-dependent manner in the highest dose group as compared with the epoetin alfa-treated group. Adverse events were similar for FG-4592-treated and control subjects. Conclusions: FG-4592 may prove an effective alternative for managing anemia of CKD. It is currently being investigated in a pivotal global Phase 3 program.
Bosutinib, an SRC inhibitor, induces caspase-independent cell death associated with permeabilization of lysosomal membranes in melanoma cells.[Pubmed:28371355]
Vet Comp Oncol. 2018 Mar;16(1):69-76.
BACKGROUND: SRC kinase (SRC proto-oncogene, non-receptor tyrosine kinase) is a promising target for the treatment of solid cancers including human melanoma. Bosutinib (Bosu), a SRC inhibitor, has already been applied to the treatment of human chronic myelogenous leukemia and also has been assessed its safety in dogs. AIM: The aim of this study was to clarify a novel anti-tumour mechanism of Bosu in canine and human melanoma cells. MATERIALS AND METHODS: The canine and human melanoma cells were treated with Bosu and its effects were evaluated by the cell viability, the protein expression levels such as caspase-3 and LC3, Annexin V/Propidium iodide staining, and confocal immunostaining. RESULTS: Bosu induced the massive caspase-independent cell death, and blocked autophagy flux, which resulted from lysosomal dysfunction. Lysosomal dysfunction caused by Bosu was due to lysosomal membrane permeabilization (LMP), which resulted in the release of lysosomal hydrolases including cathepsin B. CONCLUSION: Our data suggest that Bosu induces the cell death through induction of LMP in melanoma cells and is a promising therapeutic agent for treatment of melanoma in both dogs and humans.
Development of a Hypoxic Radiosensitizer-Prodrug Liposome Delivery DNA Repair Inhibitor Dbait Combination with Radiotherapy for Glioma Therapy.[Pubmed:28371526]
Adv Healthc Mater. 2017 Jun;6(12).
Gliomas are highly radioresistant tumors, mainly due to hypoxia in the core region of the gliomas and efficient DNA double-strand break repair. However, the design of a radiosensitizer incorporating the two above mechanisms is difficult and has rarely been reported. Thus, this study develops a hypoxic radiosensitizer-prodrug liposome (MLP) to deliver the DNA repair inhibitor Dbait (MLP/Dbait) to achieve the simultaneous entry of radiosensitizers with two different mechanisms into the glioma. MLP/Dbait effectively sensitizes glioma cells to X-ray radiotherapy (RT). Histological and microscopic examinations of dissected brain tissue confirm that MLP effectively delivers Dbait into the glioma. Furthermore, the combination of MLP/Dbait with RT significantly inhibits growth of the glioma, as assessed by in vivo bioluminescence imaging. These findings suggest that MLP is a promising candidate as a Dbait delivery system to enhance the effect of RT on glioma, owing to the synergistic effects of the two different radiosensitizers.


