(-)-HeraclenolCAS# 139079-42-8 |
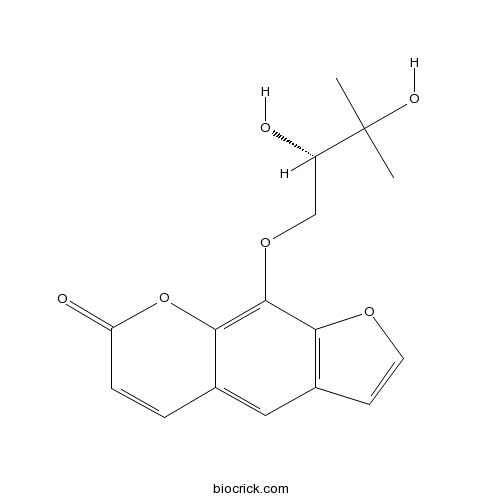
- Heraclenol
Catalog No.:BCN5234
CAS No.:31575-93-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 139079-42-8 | SDF | Download SDF |
| PubChem ID | 40429858 | Appearance | Powder |
| Formula | C16H16O6 | M.Wt | 304.29 |
| Type of Compound | Coumarins | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 9-[(2S)-2,3-dihydroxy-3-methylbutoxy]furo[3,2-g]chromen-7-one | ||
| SMILES | CC(C)(C(COC1=C2C(=CC3=C1OC=C3)C=CC(=O)O2)O)O | ||
| Standard InChIKey | FOINLJRVEBYARJ-NSHDSACASA-N | ||
| Standard InChI | InChI=1S/C16H16O6/c1-16(2,19)11(17)8-21-15-13-10(5-6-20-13)7-9-3-4-12(18)22-14(9)15/h3-7,11,17,19H,8H2,1-2H3/t11-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. (-)-Heraclenol shows in vitro anti-plasmodial activity against chloroquine-sensitive and chloroquine-resistant strains of Plasmodium falciparum. 2. (-)-Heraclenol has moderate antibacterial and antifungal activities. |
| Targets | Antifection |

(-)-Heraclenol Dilution Calculator

(-)-Heraclenol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.2863 mL | 16.4317 mL | 32.8634 mL | 65.7268 mL | 82.1585 mL |
| 5 mM | 0.6573 mL | 3.2863 mL | 6.5727 mL | 13.1454 mL | 16.4317 mL |
| 10 mM | 0.3286 mL | 1.6432 mL | 3.2863 mL | 6.5727 mL | 8.2158 mL |
| 50 mM | 0.0657 mL | 0.3286 mL | 0.6573 mL | 1.3145 mL | 1.6432 mL |
| 100 mM | 0.0329 mL | 0.1643 mL | 0.3286 mL | 0.6573 mL | 0.8216 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- PF-4981517
Catalog No.:BCC2270
CAS No.:1390637-82-7
- L-689,560
Catalog No.:BCC6774
CAS No.:139051-78-8
- Anemarsaponin B
Catalog No.:BCN6289
CAS No.:139051-27-7
- 2-PMDQ
Catalog No.:BCC6726
CAS No.:139047-55-5
- JMV 449
Catalog No.:BCC5863
CAS No.:139026-66-7
- Oplodiol
Catalog No.:BCN6204
CAS No.:13902-62-0
- Catalpin
Catalog No.:BCN6205
CAS No.:1390-72-3
- Carmine
Catalog No.:BCN2223
CAS No.:1390-65-4
- 3,4-Dihydroxybenzaldehyde
Catalog No.:BCN6214
CAS No.:139-85-5
- Ziprasidone hydrochloride monohydrate
Catalog No.:BCC2072
CAS No.:138982-67-9
- Capsazepine
Catalog No.:BCC1451
CAS No.:138977-28-3
- Coronarin D ethyl ether
Catalog No.:BCN6203
CAS No.:138965-89-6
- Tristin
Catalog No.:BCN4709
CAS No.:139101-67-0
- PF 4800567 hydrochloride
Catalog No.:BCC7904
CAS No.:1391052-28-0
- Vigabatrin Hydrochloride
Catalog No.:BCC5198
CAS No.:1391054-02-6
- Zanamivir
Catalog No.:BCC4946
CAS No.:139110-80-8
- BM-1074
Catalog No.:BCC2235
CAS No.:1391108-10-3
- Isomurralonginol acetate
Catalog No.:BCN4708
CAS No.:139115-59-6
- Tripterifordin
Catalog No.:BCN6206
CAS No.:139122-81-9
- Uralenol
Catalog No.:BCN7994
CAS No.:139163-15-8
- H-Tle-OH.HCl
Catalog No.:BCC2660
CAS No.:139163-43-2
- CEP-37440
Catalog No.:BCC5145
CAS No.:1391712-60-9
- ZM 241385
Catalog No.:BCC6902
CAS No.:139180-30-6
- Picfeltarraenin X
Catalog No.:BCN2859
CAS No.:1391826-61-1
Evaluation of furanocoumarins from seeds of the wild parsnip (Pastinaca sativa L. s.l.).[Pubmed:30562630]
J Chromatogr B Analyt Technol Biomed Life Sci. 2019 Jan 15;1105:54-66.
Although the wild parsnip (Pastinaca sativa L. s.l.) fruits are known to contain linear and angular furanocoumarins, the individual components of the seeds have not been fully identified and quantitated, and, in the case of immature seeds, reported. In view of this, the main furanocoumarin compounds were extracted using pyridine, and were isolated using semi-preparative high-performance liquid chromatography. The structural elucidation of isolated compounds was done based on detailed spectral analysis conducted by liquid chromatography-electrospray ionization-mass spectrometry (LC-ESI/MS), (1)H and (13)C NMR and, where possible, by gas chromatography-mass spectrometry (GC-MS). The quantitative analysis of furanocoumarin compounds in the wild parsnip was conducted by analytical ultra-performance liquid chromatography (UPLC-DAD), calculated against the standard curves of isolated compounds. The total yields of furanocoumarin compounds from the seeds after extraction with pyridine were 107.2-222.8mgg(-1) (fresh weight) and 50.2-66.4mgg(-1) (soluble dry matter). Thirteen furanocoumarins were identified. The main compounds (percentage in FW) in immature seeds were bergapten (40.8), pimpinellin (10.5), methoxsalen (5.7), isopimpinellin (4.3), imperatorin (3.2), and phellopterin (7.2). Seven constituents previously not described in P. sativa seeds and its products were identified, namely, byakangelicol (14.4), heraclenin (8.5), isobergapten (2.5), byakangelicin (1.3), (-)-Heraclenol (0.5), psoralen (0.3), and isobyakangelicin (0.8). The latter is a new compound of the Apiaceae family. Extraction of immature seeds using pyridine gave a much higher yield and a greater variety of furanocoumarins. This indicates that the wild parsnip, along with other Apiaceae family plants, may be an important source of bioactive compounds.
New coumarin from the roots of Prangos pabularia.[Pubmed:29224384]
Nat Prod Res. 2018 Oct;32(19):2325-2332.
The new coumarin 1, yuganin A (7-methoxy-8-((1S,2S)-1,2,3-trihydroxy-3-methylbutyl)-2H-chromen-2-one) along with nine known coumarins, (-)-Heraclenol 3'-O-beta-D-glucopyranoside (2), oxypeucedanin hydrate 3'-O-beta-D-glucopyranoside (3), (-)-Heraclenol (4), oxypeucedanin hydrate (5), osthole (6), oxypeucedanin (7), heraclenin (8), isoimperatorin (9), imperatorin (10) and the disaccharide sucrose (11), have been isolated from the roots of Prangos pabularia, and the structures of these isolated compounds were elucidated by spectroscopic means, especially, UV, HR-ESIMS, and 1D and 2D NMR spectroscopy. Furthermore, the anti-melanogenic effect of yuganin A and its inhibitory effect on B16 cells were evaluated. Yuganin A may be useful in the treatment of hyperpigmentation and as a skin-whitening agent in the cosmetics industry.
[Chemical constituents from lipophilic parts in roots of Angelica dahurica cv.Yubaizhi].[Pubmed:28822155]
Zhongguo Zhong Yao Za Zhi. 2017 Jun;42(11):2102-2109.
The chemical constituents from lipophilic parts in the roots of Angelica dahurica cv. Yubaizhi were studied in this paper. The compounds were separated and purified by repeated column chromatographic methods on silica gel and HPLC, and the chemical structures of compounds were determined by spectral data analyses. Thirty-three compounds were obtained and identified as isoimperatorin (1), imperatorin (2), stigmasterol (3), isooxypeucedanin (4), pabulenol (5), psoralen (6), bergapten (7), isodemethylfuropinarine (8), phellopterin (9), osthenol (10), alloimperatorin (11), xanthotoxin (12), xanthotoxol (13), isopimpinellin (14), alloisoimperatorin (15), beta-sitosterol (16), oxyalloimperatorin (17), pabularinone (18), 5-hydroxy-8-methoxypsoralen (19), columbianetin (20), heracol (21), isogosferol (22), 2''R-neobyakangelicol (23), byakangelicin ethoxide (24), byakangelicin (25), oxypeucedanin hydrate (26), uracil (27), umbelliferone (28), bergaptol (29), demethylfuropinarine (30), isobyakangelicol (31), oxypeucedanin ethanolate (32), (-)-Heraclenol (33). Among them, compounds 8, 10, 17, 21, and 30 were obtained from the roots of title plant for the first time.
[Chemical constituents from lipophilic parts in roots of Angelica dahurica var. formosana cv. Chuanbaizhi].[Pubmed:26552172]
Zhongguo Zhong Yao Za Zhi. 2015 Jun;40(11):2148-56.
The chemical constituents from lipophilic parts in the roots of Angelica dahurica var. formosana cv. Chuanbaizhi were studied in this paper. The compounds were separated and purified by repeated column chromatographic methods on silica gel and HPLC, and the chemical structures of compounds were determined by spectral data analyses. Twenty-nine compounds were obtained and identified as isoimperatorin (1), beta-sitosterol (2), imperatorin (3), bergapten (4), osthenol (5), xanthotoxin (6), isoimpinellin (7), dehydrogeijerin (8), phellopterin (9), isodemethylfuropinarine (10), 7-demethylsuberosin (11), alloimperatorin (12), xanthotoxol (13), isooxypeucedanin (14), alloisoimperatorin (15), demethylfuropinarine (16), 5-hydroxy-8-methoxypsoralen (17), oxypeucedanin methanolate (18), pabulenol (19), byakangelicin (20), marmesin (21), (+) -decursinol (22), (-)-Heraclenol (23), oxypeucedanin hydrate (24), marmesinin (25), ulopterol (26), erythro-guaiacylglycerol-beta-ferulic acid ether (27), threo-guaiacylglycerol-beta-ferulic acid ether (28), and uracil (29). Compounds 5, 8, 11, 18, 21-23, and 26-28 were obtained from the roots of title plant for the first time.
Isolation, cytotoxicity evaluation and HPLC-quantification of the chemical constituents from Prangos pabularia.[Pubmed:25314269]
PLoS One. 2014 Oct 14;9(10):e108713.
Phytochemical analysis of the dichloromethane:methanol (1:1) extract of root parts of Prangos pabularia led to the isolation of twelve cytotoxic constituents, viz., 6-hydroxycoumarin (1), 7-hydroxycoumarin (2), (-)-Heraclenol-glycoside (3), xanthotoxol (4), (-)-Heraclenol (5), oxypeucedanin hydrate (6), 8-((3,3-dimethyloxiran-2-yl)methyl)-7-methoxy-2H-chromen-2-one (7), oxypeucedanin hydrate monoacetate (8), xanthotoxin (9), 4-((2-hydroxy-3-methylbut-3-en-1-yl)oxy)-7H-furo[3,2-g]chromen-7-one (10), imperatorin (11) and osthol (12). The isolates were identified using spectral techniques in the light of literature. 3-(4,5-dimethyl thiazol-2yl)-2,5-diphenyltetrazolium bromide (MTT) cytotoxicity screening of the isolated constituents was carried out against six human cancer cell lines including lung (A549 and NCI-H322), epidermoid carcinoma (A431), melanoma (A375), prostate (PC-3) and Colon (HCT-116) cell lines. Osthol (12) exhibited the highest cytotoxicity with IC50 values of 3.2, 6.2, 10.9, 14.5, 24.8, and 30.2 microM against epidermoid carcinoma (A431), melanoma (A375), lung (NCI-H322), lung (A549), prostate (PC-3) and colon (HCT-116) cell lines respectively. Epidermoid carcinoma cell line A431 was sensitive to most of the compounds followed by lung (A549) cancer cell line. Finally a simple and reliable HPLC method was developed (RP-HPLC-DAD) and validated for the simultaneous quantification of these cytotoxic constituents in Prangos pabularia. The extract was analyzed using a reversed-phase Agilent ZORBAX eclipse plus column C18 (4.6x250 mm, 5 microm) at 250 nm wavelength using a gradient water-methanol solvent system at a flow rate of 0.8 ml/min. The RP-HPLC method is validated in terms of recovery, linearity, accuracy and precision (intra and inter-day validation). This method, because of shorter analysis time, makes it valuable for the commercial quality control of Prangos pabularia extracts and its future pharmaceutical preparations.
In vitro and in vivo antiproliferative effect of a combination of ultraviolet-A and alkoxy furocoumarins isolated from Umbelliferae medicinal plants, in melanoma cells.[Pubmed:23802687]
Photochem Photobiol. 2013 Sep-Oct;89(5):1216-25.
We examined the effects of six furocoumarins with alkoxy groups at the C-5 or C-8 position isolated from Umbelliferae medicinal plants on cell proliferation, and their mechanisms of action against B16F10 melanoma cells or in melanin-possessing hairless mice implanted with B16F10 cells, under UVA irradiation. Three furocoumarins with an alkoxy group at C-5, isoimperatorin (1), oxypeucedanin (2) and oxypeucedanin hydrate (3), showed antiproliferative activity and caused G2/M arrest at concentrations of 0.1-10.0 mum. Furthermore, three furocoumarins with an alkoxy group at C-8, imperatorin (4), heraclenin (5) and (-)-Heraclenol (6), inhibited the proliferation of melanoma cells and cell cycle at G2/M at concentrations of 0.1-1.0 mum. UVA plus 1, 2, 3, 4 and 6 reduced tumor growth and final tumor weight in B16F10-bearing mice at a dose of 0.3, 0.5 or 1.0 mg kg(-1) (intraperitoneal injection). UVA plus 1, 3 and 6 increased Chk1 phosphorylation and reduced cdc2 (Thr 161) phosphorylation in melanoma cells. We suggest that the antitumor actions of UVA plus furocoumarins with an alkoxy group at C-5 or C-8 were due to G2/M arrest of the cell cycle by an increase in phosphor-Chk1 and decrease in phospho-cdc2.
[A new natural product from root of Angelica dahurica cv. Qibaizhi].[Pubmed:23234138]
Zhongguo Zhong Yao Za Zhi. 2012 Aug;37(16):2400-7.
OBJECTIVE: To study the chemical constituents of the root of Angelica dahurica cv. Qibaizhi. METHOD: The compounds were isolated and purified by the methods of solvent extraction and chromatographic technique, and their structures were identified on the basis of the analyses of spectral data. RESULT: Twenty-seven compounds were obtained and identified as isoimperatorin (1), alloisoimperatorin (2), oxypeucedanin hydrate (3), byakangelicin (4), alloimperatorin (5), beta-sitosterol (6), dibutylphthalate (7), 6-(3,3-dimethylallyloxy) -7-hydroxycoumarin {7-hydroxy-6-[ (3-methyl-2-butenyl) oxy] -coumarin, 8}, xanthotoxol (9), 5-hydroxy-8-methoxypsoralen ( 5-hydroxyxanthotoxin, 10), smyrindiol (11), umbelliferone (12), bergapten (13), imperatorin (14), phellopterin (15), isoimpinellin (16), 6-acyl-7-methoxycoumarin (17), psoralen (18), isoscopletin (19), scoparone (20), xanthotoxin (21), angelol A (22), oxypeucedanin ethanolate (oxypeucedanin hydrate-3"-ethyl ether, 23), dehydrogeijerin (pablohopin, 24) , ulopterol (25), (-)-Heraclenol (26), and pabulenol (27), respectively. CONCLUSION: Compound 8 was a new natural product and the above-mentioned all the compounds except for compounds 1, 3 and 14 were isolated from the root of A. dahurica cv. Qibaizhi for the first time.
Phytotoxic furanocoumarins from the shoots of Semenovia transiliensis.[Pubmed:23157001]
Nat Prod Commun. 2012 Oct;7(10):1327-30.
Discovery of novel, natural herbicides has become important to manage increasing weed resistance to synthetic herbicides and environmental issues. The systematic bioassay-guided fractionation and purification of the methylene chloride/methanol extract of the shoots of Semenovia transiliensis led to the isolation of several phytotoxic compounds. Lactuca sativa L. (lettuce, a dicot) and Agrostis stolonifera L. (bentgrass, a monocot) bioassays were used to identify and isolate the phytotoxic fractions. A number of furanocoumarin compounds isolated from S. transiliensis shoots were phytotoxic to both test species. These included psoralen, isopsoralen, heratomin, isopentenyloxyisobergapten, imperatorin, bergapten, xanthotoxin, heraclenin, and (-)-Heraclenol. All the active secondary metabolites isolated from the shoots of S. transiliensis were furanocoumarins. Identification of these was accomplished using mass spectrometry and 1- and 2-dimensional NMR techniques. Phytotoxic activity o f isolated compounds w a s evaluated in a dose-response manner from 0.3 to 1000 microM. Ingeneral, all of the compounds were more active on A. stolonifera than L. sativa. Bergaptin and xanthotoxin were the most active of the compounds, with moderate activity at 100 microM. Imperatorin and xanthotoxin inhibited growth of Lemna paucicostata Hegelm. by 50% at 29 and 60 microM, respectively. Our results show that S. transiliensis is rich in furanocoumarins, which are probably involved in various aspects of the chemical ecology of the species. Unfortunately, the general cytotoxicity of furanocoumarins makes them an unlikely candidate for pesticide discovery.
Bioactivity-guided fractionation for the butyrylcholinesterase inhibitory activity of furanocoumarins from Angelica archangelica L. roots and fruits.[Pubmed:21786787]
J Agric Food Chem. 2011 Sep 14;59(17):9186-93.
Isolation and identification of the inhibitors of butyrylcholinesterase (BChE), obtained from the extracts of roots and fruits of Angelica archangelica L., are reported. Our results confirmed the weak inhibitory effect of Angelica roots on acetylcholinesterase activity. BChE inhibition was much more pronounced at a concentration of 100 mug/mL for hexane extracts and attained a higher rate than 50%. The TLC bioautography guided fractionation and spectroscopic analysis led to the isolation and identification of imperatorin from the fruit's hexane extract and of (-)-Heraclenol-2'-O-angelate from the root's hexane extract. Both compounds showed significant BChE inhibition activity with IC(50) = 14.4 +/- 3.2 muM and IC(50) = 7.5 +/- 1.8 muM, respectively. Only C8-substituted and C5-unsubstituted furanocoumarins were active, which could supply information about the initial structures of specific BChE inhibitors.
Vasorelaxation by extracts of Casimiroa spp. in rat resistance vessels and pharmacological study of cellular mechanisms.[Pubmed:21236328]
J Ethnopharmacol. 2011 Apr 12;134(3):637-43.
AIM OF THE STUDY: Casimiroa spp. are Mexican plants traditionally used for treatment of hypertension. To study their antihypertensive action, we determined the arterial dilatation induced by extracts from leaves and seeds of Casimiroa calderoniae F. Chiang & Medrano, Casimiroa edulis Llave et Lex, and Casimiroa pubescens Ramirez. MATERIALS AND METHODS: The vascular effects of Casimiroa spp. extracts were investigated on rat caudal and aortic arteries. In addition, the extracts were characterized by HPLC using (-)-Heraclenol, isopimpinellin, heraclenin and phellopterin as external standards. The methanolic extract of Casimiroa pubescens seeds (Cp12) was also studied by H-NMR and LC-MS (ESI-TOF) for the determination of casimiroin and zapotin. RESULTS: The hexanic and methanolic extracts of Casimiroa spp. (20 mug/ml) showed vasorelaxation in arterial tissues precontracted by phenylephrine (0.5 muM); the extracts from seeds always caused a greater relaxation in comparison to those from leaves. The most active were the methanolic seed extracts of Casimiroa edulis (Ce8) and Casimiroa pubescens (Cp12). To study the pharmacological mechanisms of vasodilatation we used various inhibitors selective to different receptor subtypes or intracellular enzymes. The vasorelaxant effect of Ce8 (20 mug/ml) remained unaffected by the pretreatment with pyrilamine (10 muM), an antagonist of histamine H(1) receptors, but was inhibited by atropine (0.1 muM), a muscarinic receptor antagonist. Therefore, to determine muscarinic receptor subtypes, we used pirenzepine (1 muM), a selective inhibitor of M(1) receptor, and 4-diphenylacetoxyl-N-methylpiperidine methiodide (DAMP, 0.01 muM), a selective inhibitor of M(3) receptor. Only the latter reduced the vasodilatation by Ce8 and Cp12. To investigate the role of the nitric oxide synthase (NOS), we used N(G)-nitro-l-arginine methyl ester (l-NAME, 10 muM), a selective NOS inhibitor, which decreased the dilatation induced by Ce8 and Cp12. Finally, we studied the action of (1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one) (ODQ, 3 muM), a selective guanylyl cyclase inhibitor, which inhibited the dilatation by Casimiroa extracts. CONCLUSION: The data show that methanolic seed extracts of Casimiroa edulis (Ce8) and Casimiroa pubescens (Cp12) induce vasorelaxation by M(3) receptor through the activation of cGMP-dependent NO signaling. These results support the traditional use of Casimiroa decoctions for antihypertensive treatments in the Mexican ethnomedicine.
[Chemical constituents of Incarvillea younghusbandii].[Pubmed:20349717]
Zhongguo Zhong Yao Za Zhi. 2010 Jan;35(1):58-62.
OBJECTIVE: To study the chemical constituents of Incarvillea younghusbandii. METHOD: The chemical constituents were isolated by various column chromatographic methods and structurally identified by NMR and MS evidence. RESULT: Fifteen compounds were obtained and identified as isobergapten (1), sphondin (2), imperatorin (3), xanthotoxin (4), phellopterin (5), (-)-Heraclenol (6), rivulobirin A (7), methyl oleanolate (8), methyl caffeate (9), grevillic acid (10), boschniakinic acid (11), tert-O-beta-D-glucopyranosyl-(R)-heraclenol (12), 5-methoxy-8-O-beta-D-glucopyranosyloxypsoralen (13), 1'-O-beta-D-glucopyranosyl-3-hydroxynodakenetin (14) and phenylethyl-O-beta-D-glucopyranosyl-(1-->2)-beta-D-glucopyranoside (15). CONCLUSION: All of these compounds were isolated from this plant for the first time and most of them are furocoumarins.
Coumarins from Angelica lucida L.--antibacterial activities.[Pubmed:19701119]
Molecules. 2009 Jul 27;14(8):2729-34.
The first phytochemical investigation of the fruits of Angelica lucida has led to the isolation and characterization of five known coumarins (imperatorin, isoimperatorin, (-)-Heraclenol, oxypeucedanin hydrate and heraclenin). All isolated compounds were identified by means of spectral and literature data. The extracts and the isolated constituents from A. lucida have been also evaluated for their antimicrobial activity against six Gram positive and negative bacteria, two oral pathogens and three human pathogenic fungi, exhibiting an interesting antimicrobial profile.
Screening of potential chemopreventive compounds from Poncirus trifoliata Raf.[Pubmed:17020159]
Pharmazie. 2006 Sep;61(9):796-8.
Chemopreventive agents induce a battery of genes whose protein products can protect cells from chemical-induced carcinogenesis. In this study, we isolated three different coumarins compounds (1; poncimarin, 2; (-)-Heraclenol 3'-methyl ester and 3; oxypeucedanin methanolate) from Poncirus trifoliata Raf., and studied whether these compounds increase glutathione S-transferase (GST) expression and activity in the H4IIE cell-line (a rat hepatocyte cell line). CDNB (1-chloro-2,4-dinitrobenzene; GST subtype-nonspecific) and NBD (7-chloro-4-nitrobenzo-2-oxa-1,3-diazole; GSTalpha-type-specific) assays revealed that compound 1 most potently increased GST enzyme activities. Western blot analysis using subtype-specific antibodies confirmed that these three coumarins also selectively increased GSTalpha-protein expression, and that compound 1 most actively induced GSTalpha. In contrast, the expressions of the GSTmu and GSTmu subtypes were not altered by these three coumarins. Reporter gene analysis using an antioxidant response element (ARE) containing construct and subcellular fractionation assays, revealed that GSTalpha-induction by compound 1 might be associated with Nrf2/ARE activation. These results suggest that these three coumarin compounds from Poncirus trifoliata Raf possess phase II enzyme inducible functions, and in particular, that poncimarin has chemopreventive potential.
Candibirin A, a furanocoumarin dimer isolated from Heracleum candicans WALL.[Pubmed:15528835]
Acta Crystallogr C. 2004 Nov;60(Pt 11):o833-5.
Candibirin A [systematic name: 9,9'-(1,4-dioxane-2,5-diyldimethylenedioxy)di(7H-furo[3,2-g]chromen-7-one)], a new furanocoumarin dimer, was isolated from Heracleum candicans WALL. (1)H NMR and MS spectra had indicated that the title compound was a dimer of heraclenin or (-)-Heraclenol, but the linkage structure and its chirality were undetermined. The dioxane linkage, having the R,R configuration, has now been elucidated from dimethyl sulfoxide-solvated crystals, C(32)H(28)O(10).2C(2)H(6)OS. Candibirin A is thus a dimerization product from heraclenin formed by reaction at the epoxy group. Dimethylformamide-solvated crystals, C(32)H(28)O(10).C(3)H(7)NO, adopt a different conformation, with a folded structure that differs from the extended structure in the dimethyl sulfoxide solvate. However, the puckering of the dioxane linker unit is very similar in the two conformers. This result shows that the rotation of the ether bonds, in the linker between the furanocoumarin and dioxane moieties, causes the conformational flexibility of (I).


