KumatakeninCAS# 3301-49-3 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 3301-49-3 | SDF | Download SDF |
| PubChem ID | 5318869 | Appearance | Yellow powder |
| Formula | C17H14O6 | M.Wt | 314.3 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
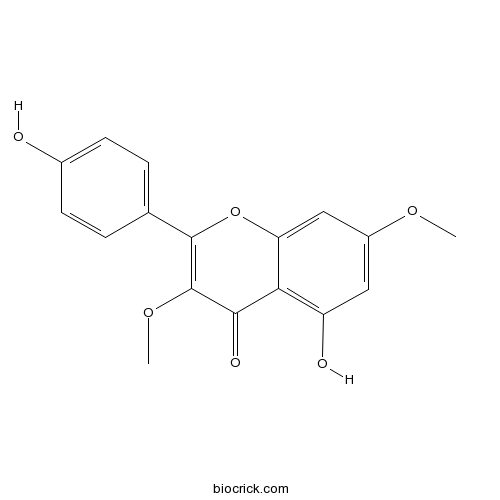
| Chemical Name | 5-hydroxy-2-(4-hydroxyphenyl)-3,7-dimethoxychromen-4-one | ||
| SMILES | COC1=CC(=C2C(=C1)OC(=C(C2=O)OC)C3=CC=C(C=C3)O)O | ||
| Standard InChIKey | BJBUTJQYZDYRMJ-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C17H14O6/c1-21-11-7-12(19)14-13(8-11)23-16(17(22-2)15(14)20)9-3-5-10(18)6-4-9/h3-8,18-19H,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Kumatakenin can suppress >80% of the SOS-inducing activity of Trp-P-1 at <0.06 micromol/mL. |
| Targets | Antifection |
| In vitro | Antimutagenic activity of flavonoids from Pogostemon cablin.[Pubmed: 10725128]J Agric Food Chem. 2000 Mar;48(3):642-7.A methanol extract from Pogostemon cablin showed a suppressive effect on umu gene expression of SOS response in Salmonella typhimurium TA1535/pSK1002 against the mutagen 2-(2-furyl)-3-(5-nitro-2-furyl)acrylamide (furylfuramide). |
| Structure Identification | Zhongguo Zhong Yao Za Zhi. 2014 Jul;39(14):2674-8.A new flavonone from seeds of Alpinia katsumadai and its neuroprotective effect on PC12 cells.[Pubmed: 25272494]A new flavonone, named as (2R, 3S)-pinobanksin-3-cinnamate(1), together with six known compounds, pinocem-brin (2), pinobanksin (3), 3-O-acetylpinobanksin (4), galangin (5), Kumatakenin(6), and 3-methylkaempferol (7), were isolated from a 95% ethanol extract of seeds of Alpinia katsumadai through a combination of various chromatographic techniques, including silica gel and Sephadex LH-20. Pharmacogn Mag. 2010 Oct;6(24):339-44.Phytoconstituents from Alpinia purpurata and their in vitro inhibitory activity against Mycobacterium tuberculosis.[Pubmed: 21120040]Alpinia purpurata or red ginger was studied for its phytochemical constituents as part of our growing interest on Philippine Zingiberaceae plants that may exhibit antimycobacterial activity. |

Kumatakenin Dilution Calculator

Kumatakenin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.1817 mL | 15.9084 mL | 31.8167 mL | 63.6335 mL | 79.5418 mL |
| 5 mM | 0.6363 mL | 3.1817 mL | 6.3633 mL | 12.7267 mL | 15.9084 mL |
| 10 mM | 0.3182 mL | 1.5908 mL | 3.1817 mL | 6.3633 mL | 7.9542 mL |
| 50 mM | 0.0636 mL | 0.3182 mL | 0.6363 mL | 1.2727 mL | 1.5908 mL |
| 100 mM | 0.0318 mL | 0.1591 mL | 0.3182 mL | 0.6363 mL | 0.7954 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Nicarbazin
Catalog No.:BCC9101
CAS No.:330-95-0
- 3,4-Secocucurbita-4,24-diene-3,26,29-trioic acid
Catalog No.:BCN1458
CAS No.:329975-47-5
- TTP 22
Catalog No.:BCC2017
CAS No.:329907-28-0
- Viomycin
Catalog No.:BCC3930
CAS No.:32988-50-4
- Tobramycin
Catalog No.:BCC4739
CAS No.:32986-56-4
- 10-Deacetylbaccatin III
Catalog No.:BCN5251
CAS No.:32981-86-5
- Methyl (2R,3S)-3-(benzoylamino)-2-hydroxy-3-phenylpropanoate
Catalog No.:BCN8520
CAS No.:32981-85-4
- Cinaciguat
Catalog No.:BCC1484
CAS No.:329773-35-5
- 2-Acetyl-3-ethylpyrazine
Catalog No.:BCC8512
CAS No.:32974-92-8
- Pinoresinol diacetate
Catalog No.:BCN5250
CAS No.:32971-25-8
- FPR A14
Catalog No.:BCC7498
CAS No.:329691-12-5
- 7-O-Methylaloeresin A
Catalog No.:BCN2849
CAS No.:329361-25-3
- ent-16beta,17-Dihydroxy-19-kauranoic acid
Catalog No.:BCN1457
CAS No.:3301-61-9
- Cyanidin-3-O-sambubioside chloride
Catalog No.:BCN3150
CAS No.:33012-73-6
- SU6656
Catalog No.:BCC6392
CAS No.:330161-87-0
- TC HSD 21
Catalog No.:BCC6228
CAS No.:330203-01-5
- Boc-β-Ala-OH
Catalog No.:BCC3051
CAS No.:3303-84-2
- Aloe-emodin-8-O-beta-D-glucopyranoside
Catalog No.:BCN1456
CAS No.:33037-46-6
- H-Orn(Z)-OH
Catalog No.:BCC3003
CAS No.:3304-51-6
- Peramivir
Catalog No.:BCC1846
CAS No.:330600-85-6
- TCTU
Catalog No.:BCC2689
CAS No.:330641-16-2
- HCTU
Catalog No.:BCC2818
CAS No.:330645-87-9
- KH 7
Catalog No.:BCC7787
CAS No.:330676-02-3
- Paclitaxel
Catalog No.:BCN4650
CAS No.:33069-62-4
Phytoconstituents from Alpinia purpurata and their in vitro inhibitory activity against Mycobacterium tuberculosis.[Pubmed:21120040]
Pharmacogn Mag. 2010 Oct;6(24):339-44.
Alpinia purpurata or red ginger was studied for its phytochemical constituents as part of our growing interest on Philippine Zingiberaceae plants that may exhibit antimycobacterial activity. The hexane and dichloromethane subextracts of the leaves were fractionated and purified using silica gel chromatography to afford a mixture of C(28)-C(32) fatty alcohols, a 3-methoxyflavone and two steroidal glycosides. The two latter metabolites were spectroscopically identified as Kumatakenin (1), sitosteryl-3-O-6-palmitoyl-beta-D-glucoside (2) and b-sitosteryl galactoside (3) using ultraviolet (UV), infrared (IR), electron impact mass spectrometer (EIMS) and nuclear magnetic resonance (NMR) experiments, and by comparison with literature data. This study demonstrates for the first time the isolation of these constituents from A. purpurata. In addition to the purported anti-inflammatory activity, its phytomedicinal potential to treat tuberculosis is also described.
A new flavonone from seeds of Alpinia katsumadai and its neuroprotective effect on PC12 cells.[Pubmed:25272494]
Zhongguo Zhong Yao Za Zhi. 2014 Jul;39(14):2674-8.
A new flavonone, named as (2R, 3S)-pinobanksin-3-cinnamate(1), together with six known compounds, pinocem-brin (2), pinobanksin (3), 3-O-acetylpinobanksin (4), galangin (5), Kumatakenin(6), and 3-methylkaempferol (7), were isolated from a 95% ethanol extract of seeds of Alpinia katsumadai through a combination of various chromatographic techniques, including silica gel and Sephadex LH-20. The structure of compound 1 was elucidated by spectroscopic data analysis. Compound 1 exhibits a potent neuroprotective effect against the corticosterone-damaged PC12 cells, which may be underlying the effect by scavenging intracellular ROS.
Antimutagenic activity of flavonoids from Pogostemon cablin.[Pubmed:10725128]
J Agric Food Chem. 2000 Mar;48(3):642-7.
A methanol extract from Pogostemon cablin showed a suppressive effect on umu gene expression of SOS response in Salmonella typhimurium TA1535/pSK1002 against the mutagen 2-(2-furyl)-3-(5-nitro-2-furyl)acrylamide (furylfuramide). The methanol extract was re-extracted with hexane, dichloromethane, butanol, and water. A dichloromethane fraction showed a suppressive effect. Suppressive compounds against furylfuramide in the dichloromethane fraction were isolated by SiO(2) column chromatography and identified as 7,4'-di-O-methyleriodictyol (1), 7, 3',4'-tri-O-methyleriodictyol (2), and 3,7,4'-tri-O-methylkaempferol (3). In addition, three flavonoids, ombuine (4), pachypodol (5), and Kumatakenin (6), were isolated and identified from the dichrolomethane fraction. Compounds 1 and 3 suppressed >50% of the SOS-inducing activity at <0.6 micromol/mL, and the ID(50) values of both compounds were 0.25 micromol/mL. Compound 2 showed a weakly suppressive effect (17%) at a concentration of 0.6 micromol/mL, and compounds 4-6 did not. These compounds were also assayed with 3-amino-1,4-dimethyl-5H-pyrido[4,3-b]indole (Trp-P-1), which requires liver metabolizing enzymes. Compounds 3-6 suppressed >80% of the SOS-inducing activity of Trp-P-1 at <0.06 micromol/mL, and compounds 1 and 2 suppressed 87 and 63% at a concentration of 0.3 micromol/mL. In addition, these compounds were assayed with activated Trp-P-1, and the suppressed effects of these compounds were further decreased when compared to Trp-P-1. The antimutagenic activities of these compounds against furylfuramide, Trp-P-1, and activated Trp-P-1 were assayed by the Ames test using S. typhimurium TA100.


