Paroxetine maleateHighly potent and selective 5-HT uptake inhibitor CAS# 64006-44-6 |
- Amyloid β-Protein (1-15)
Catalog No.:BCC1003
CAS No.:183745-81-5
- Beta-Amyloid (1-11)
Catalog No.:BCC1002
CAS No.:190436-05-6
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 64006-44-6 | SDF | Download SDF |
| PubChem ID | 6435921 | Appearance | Powder |
| Formula | C23H24FNO7 | M.Wt | 445.44 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in ethanol and to 100 mM in DMSO | ||
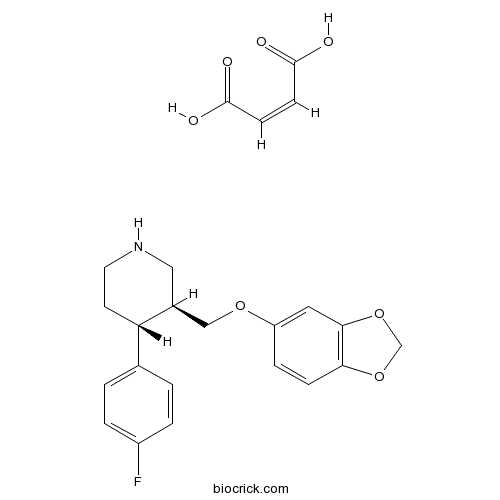
| Chemical Name | (3S,4R)-3-(1,3-benzodioxol-5-yloxymethyl)-4-(4-fluorophenyl)piperidine;(Z)-but-2-enedioic acid | ||
| SMILES | C1CNCC(C1C2=CC=C(C=C2)F)COC3=CC4=C(C=C3)OCO4.C(=CC(=O)O)C(=O)O | ||
| Standard InChIKey | AEIUZSKXSWGSRU-QXGDPHCHSA-N | ||
| Standard InChI | InChI=1S/C19H20FNO3.C4H4O4/c20-15-3-1-13(2-4-15)17-7-8-21-10-14(17)11-22-16-5-6-18-19(9-16)24-12-23-18;5-3(6)1-2-4(7)8/h1-6,9,14,17,21H,7-8,10-12H2;1-2H,(H,5,6)(H,7,8)/b;2-1-/t14-,17-;/m0./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Highly potent and selective 5-HT uptake inhibitor that binds with high affinity to the serotonin transporter (Ki = 0.05 nM). Ki values are 1.1, 350 and 1100 nM for inhibition of [3H]-5-HT, [3H]-l-NA and [3H]-DA uptake respectively. Displays minimal affinity for α1-, α2- or β-adrenoceptors, 5-HT2A, 5-HT1A, D2 or H1 receptors at concentrations below 1000 nM, however displays weak affinity for muscarinic ACh receptors (Ki = 42 nM). Antidepressant and anxiolytic in vivo. |

Paroxetine maleate Dilution Calculator

Paroxetine maleate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.245 mL | 11.2249 mL | 22.4497 mL | 44.8994 mL | 56.1243 mL |
| 5 mM | 0.449 mL | 2.245 mL | 4.4899 mL | 8.9799 mL | 11.2249 mL |
| 10 mM | 0.2245 mL | 1.1225 mL | 2.245 mL | 4.4899 mL | 5.6124 mL |
| 50 mM | 0.0449 mL | 0.2245 mL | 0.449 mL | 0.898 mL | 1.1225 mL |
| 100 mM | 0.0224 mL | 0.1122 mL | 0.2245 mL | 0.449 mL | 0.5612 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- H-D-Val-OH
Catalog No.:BCC3145
CAS No.:640-68-6
- Etimizol
Catalog No.:BCC1562
CAS No.:64-99-3
- Colchicine
Catalog No.:BCN6271
CAS No.:64-86-8
- Tolbutamide
Catalog No.:BCC5001
CAS No.:64-77-7
- Tetracycline Hydrochloride
Catalog No.:BCC1206
CAS No.:64-75-5
- Demeclocycline hydrochloride
Catalog No.:BCC5303
CAS No.:64-73-3
- Physostigmine hemisulfate
Catalog No.:BCC6724
CAS No.:64-47-1
- Lobeline Hydrochloride
Catalog No.:BCC8202
CAS No.:63990-84-1
- 13-Oxopodocarp-8(14)-en-18-oic acid
Catalog No.:BCN4009
CAS No.:63976-69-2
- Hopeyhopin
Catalog No.:BCN7533
CAS No.:63975-56-4
- Artemisinin
Catalog No.:BCN5814
CAS No.:63968-64-9
- (3R,10S)-Heptadeca-1,8-diene-4,6-diyne-3,10-diol
Catalog No.:BCC9111
CAS No.:63910-76-9
- Tamgermanetin
Catalog No.:BCN7944
CAS No.:640235-90-1
- 4(15)-Oppositene-1,7-diol
Catalog No.:BCN4180
CAS No.:640289-58-3
- Torachrysone 8-O-glucoside
Catalog No.:BCN4181
CAS No.:64032-49-1
- Boc-Lys(Ac)-OH
Catalog No.:BCC3411
CAS No.:6404-26-8
- Boc-Nle-OH
Catalog No.:BCC3296
CAS No.:6404-28-0
- Boc-ε-Acp-OH
Catalog No.:BCC3205
CAS No.:6404-29-1
- Z-D-Pro-OH
Catalog No.:BCC2752
CAS No.:6404-31-5
- Ro 04-5595 hydrochloride
Catalog No.:BCC7234
CAS No.:64047-73-0
- GANT 58
Catalog No.:BCC1587
CAS No.:64048-12-0
- Cannabispirol
Catalog No.:BCN4609
CAS No.:64052-90-0
- H-Ala(3-pyridyl)-OH.HCl
Catalog No.:BCC3321
CAS No.:64090-98-8
- Sennidin A
Catalog No.:BCN6354
CAS No.:641-12-3
Comparison of the anticholinergic effects of the serotonergic antidepressants, paroxetine, fluvoxamine and clomipramine.[Pubmed:12421645]
Eur J Pharmacol. 2002 Nov 15;454(2-3):183-8.
Paroxetine, a selective serotonin reuptake inhibitor, shows relatively high affinity for muscarinic acetylcholine receptors compared to other selective serotonin reuptake inhibitors. To determine whether paroxetine has anticholinergic effects in vivo, we examined the effects of paroxetine on oxotremorine-induced tremor, spontaneous defecation and passive avoidance performance using mice and compared the results with those using fluvoxamine, another selective serotonin reuptake inhibitor, and clomipramine, a tricyclic antidepressant with serotonin selectivity. The potency of antidepressant activity as determined in the tail suspension test was paroxetine>fluvoxamine>clomipramine. Paroxetine and clomipramine inhibited oxotremorine-induced tremor, reduced spontaneous defecation and impaired passive avoidance performance, while fluvoxamine did not have similar effects. A comparison of ED(50) values showed that the ratio of anticholinergic effect to antidepressant activity was fluvoxamine, >3.2; paroxetine, 2.1-2.6; clomipramine, <0.8. These results suggest that paroxetine may induce fewer adverse anticholinergic effects than clomipramine, but more than fluvoxamine.
Paroxetine: a review.[Pubmed:11420571]
CNS Drug Rev. 2001 Spring;7(1):25-47.
Paroxetine is a potent and selective serotonin reuptake inhibitor (SSRI) with currently approved indications for the treatment of depression, obsessive-compulsive disorder, panic disorder and social phobia. It is also used in the treatment of generalized anxiety disorder, post traumatic stress disorder, premenstrual dysphoric disorder and chronic headache. Paroxetine, a phenylpiperidine derivative, is the most potent inhibitor of the reuptake of serotonin (5-hydroxytryptamine, 5-HT) of all the currently available antidepressants including the class of SSRIs. It is a very weak inhibitor of norepinephrine (NE) uptake but it is still more potent at this site than the other SSRIs. The selectivity of paroxetine, i.e., the ratio of inhibition of uptake of norepinephrine to serotonin (NE/5-HT) is amongst the highest of the SSRIs. Paroxetine has little affinity for catecholaminergic, dopaminergic or histaminergic systems and by comparison with tricyclic antidepressants (TCAs) has, therefore, a reduced propensity to cause central and autonomic side effects. Paroxetine exhibits some affinity for the muscarinic cholinergic receptor but much less than the TCAs. In addition, the adaptive changes of somatodendritic (5-HT(1A)) and terminal (5-HT(1B/1D)) autoreceptors observed with paroxetine are different to those observed with TCAs; it also inhibits nitric oxide synthase. It is both a substrate and an inhibitor of cytochrome isoenzyme P450 2D6. Paroxetine is well absorbed orally and undergoes extensive first pass metabolism that is partially saturable. Its metabolites are pharmacologically inactive in vivo. Steady state levels are achieved after 4-14 days and an elimination half-life of 21 h is consistent with once-daily dosing. There is wide inter-individual variation in the pharmacokinetics of paroxetine in adults as well as in the young and the elderly with higher plasma concentrations and slower elimination noted in the latter. Elimination is also reduced in severe renal and hepatic impairment. Serious adverse events are, however, extremely rare even in overdose. In summary, paroxetine is well tolerated and effective in the treatment of both depressive and anxiety disorders across the age range.
Neurotransmitter receptor and transporter binding profile of antidepressants and their metabolites.[Pubmed:9400006]
J Pharmacol Exp Ther. 1997 Dec;283(3):1305-22.
Several new antidepressants that inhibit the serotonin (SERT) and norepinephrine transporters (NET) have been introduced into clinical practice the past several years. This report focuses on the further pharmacologic characterization of nefazodone and its metabolites within the serotonergic and noradrenergic systems, in comparison with other antidepressants. By use of radioligand binding assays, we measured the affinity (Ki) of 13 antidepressants and 6 metabolites for the rat and human SERT and NET. The Ki values for eight of the antidepressants and three metabolites were also determined for the rat 5-HT1A, 5-HT2A and muscarinic cholinergic receptors, the guinea pig histamine1 receptor and the human alpha-1 and alpha-2 receptors. These data are useful for predicting side effect profiles and the potential for pharmacodynamic drug-drug interactions of antidepressants. Of particular interest were the findings that paroxetine, generally thought of as a selective SERT antagonist, possesses moderately high affinity for the NET and that venlafaxine, which has been described as a "dual uptake inhibitor", possesses weak affinity for the NET. We observed significant correlations in SERT (r = 0.965) or NET (r = 0.983) affinity between rat and human transporters. Significant correlations were also observed between muscarinic cholinergic and NET affinity. There are several significant correlations between affinities for the 5-HT1A, 5-HT2A, histamine1, alpha-1 and alpha-2 receptors. These novel findings, not widely described previously, suggest that many of the individual drugs studied in these experiments possess some structural characteristic that determines affinity for several G protein-coupled, but not muscarinic, receptors.
Biochemical effects of the antidepressant paroxetine, a specific 5-hydroxytryptamine uptake inhibitor.[Pubmed:2962217]
Psychopharmacology (Berl). 1987;93(2):193-200.
Paroxetine was shown to be a potent (Ki = 1.1 nM) and specific inhibitor of [3H]-5-hydroxytryptamine (5-HT) uptake into rat cortical and hypothalamic synaptosomes in vitro. Lineweaver-Burk kinetic analysis determined that this inhibition was competitive in nature, implying a direct interaction with the 5-HT uptake transporter complex. Oral administration of paroxetine produced a dose-related inhibition of [3H]-5-HT uptake (ED50 = 1.9 mg/kg) into rat hypothalamic synaptosomes ex vivo with little effect on [3H]-l-noradrenaline (NA) uptake (ED50 greater than 30 mg/kg). This selectivity for 5-HT uptake was maintained after oral dosing for 14 days. Paroxetine (ED50 1-3 mg/kg PO) prevented the 5-HT depleting effect of p-chloroamphetamine (PCA) in rat brain, demonstrating 5-HT uptake blockade in vivo. Radioligand binding techniques in rat brain in vitro showed that paroxetine has little affinity for alpha 1, alpha 2 or beta adrenoceptors, dopamine (D2), 5-HT1, 5-HT2 or histamine (H1) receptors at concentrations below 1000 nM. Paroxetine demonstrated weak affinity for muscarinic receptors (Ki = 89 nM) but was at least 15 fold weaker than amitriptyline (Ki = 5.1 nM). Paroxetine, therefore, provides a useful pharmacological tool for investigating 5-HT systems and furthermore should be an antidepressant with reduced tricyclic-like side-effects.


