PhiKan 083p53 stabilizer CAS# 880813-36-5 |
- Nutlin-3a chiral
Catalog No.:BCC1812
CAS No.:675576-98-4
- p53 and MDM2 proteins-interaction-inhibitor chiral
Catalog No.:BCC1830
CAS No.:939981-37-0
- p53 and MDM2 proteins-interaction-inhibitor racemic
Catalog No.:BCC1831
CAS No.:939983-14-9
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 880813-36-5 | SDF | Download SDF |
| PubChem ID | 4722579 | Appearance | Powder |
| Formula | C16H18N2 | M.Wt | 238.3 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 130 mg/mL (545.46 mM; Need ultrasonic) H2O : < 0.1 mg/mL (insoluble) | ||
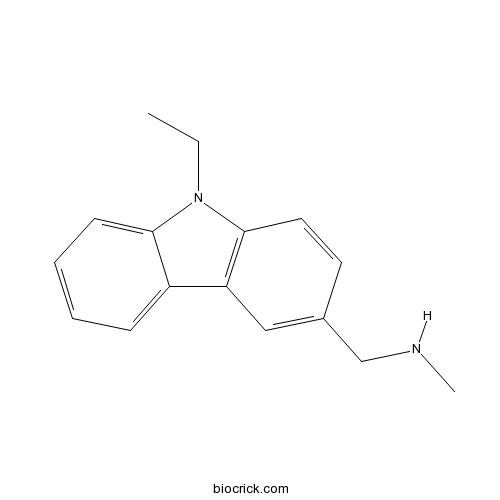
| Chemical Name | 1-(9-ethylcarbazol-3-yl)-N-methylmethanamine | ||
| SMILES | CCN1C2=C(C=C(C=C2)CNC)C3=CC=CC=C31 | ||
| Standard InChIKey | LBPNOEAFWYTTEB-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C16H18N2/c1-3-18-15-7-5-4-6-13(15)14-10-12(11-17-2)8-9-16(14)18/h4-10,17H,3,11H2,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | p53 stabilizing agent; preferentially binds mutated (Y220C) p53 over wild-type p53 at a site distinct from functional DNA/protein interaction regions. Slows rate of thermal denaturation of p53-Y220C. |

PhiKan 083 Dilution Calculator

PhiKan 083 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.1964 mL | 20.982 mL | 41.9639 mL | 83.9278 mL | 104.9098 mL |
| 5 mM | 0.8393 mL | 4.1964 mL | 8.3928 mL | 16.7856 mL | 20.982 mL |
| 10 mM | 0.4196 mL | 2.0982 mL | 4.1964 mL | 8.3928 mL | 10.491 mL |
| 50 mM | 0.0839 mL | 0.4196 mL | 0.8393 mL | 1.6786 mL | 2.0982 mL |
| 100 mM | 0.042 mL | 0.2098 mL | 0.4196 mL | 0.8393 mL | 1.0491 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Ki: ≈ 150 M
PhiKan 083 is a critical p53 stabilizing agent by bonding its mutation Y220C
The mutation Y220C of p53 exists a surface cavity that destabilizes the protein by 4 kcal/mol, at a site that is not functional. A p53 stabilizing agent binding it is able to slow rate of thermal denaturation [1].
In vitro: Isothermal titration of PhiKan083 with T-p53C-Y220C exhibited 1:1 stoichiometry and a Kd of 125 ± 10 M. PhiKan083 stabilized T-p53C-Y220C in a concentration-dependent manner. In a simple binding model for PhiKan083 (T-p53C-Y220C at 310 K (37°C)), in the absence of ligand, the protein gave a half-life of 3.8 min, while it increased to 15.7 min at saturating concentrations of PhiKan083. This is a proposed anticancer drug that could be developed for the Y220C mutant using p53 stabilizing agent. The site of mutation in the oncogenic Y220C mutant PhiKan083 binds with reasonable affinity is a druggable target. Interestingly, the site of mutation does not exist in a region of the protein that is functionally important, it will be an excellent target for drug stabilization therapy. This suggests the crystal structure of the complex will be a fresh start for further rounds of drug design and refinement [1].
In vivo: So far, no study in vivo has been conducted.
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] Boeckler FM, Joerger AC, Jaggi G, Rutherford TJ, Veprintsev DB, Fersht AR. Targeted rescue of a destabilized mutant of p53 by an in silico screened drug. Proc Natl Acad Sci U S A. 2008 Jul 29; 105(30):10360-5.
- Naloxonazine dihydrochloride
Catalog No.:BCC6710
CAS No.:880759-65-9
- AMD-070 hydrochloride
Catalog No.:BCC1358
CAS No.:880549-30-4
- Fmoc-Hyp-OH
Catalog No.:BCC3254
CAS No.:88050-17-3
- Neuropathiazol
Catalog No.:BCC5375
CAS No.:880090-88-0
- 1,2-Benzenedicarboxylic acid
Catalog No.:BCN3151
CAS No.:88-99-3
- BHQ
Catalog No.:BCC6982
CAS No.:88-58-4
- 2-Acetylthiophene
Catalog No.:BCC8517
CAS No.:88-15-3
- Furan-2-carboxylic acid
Catalog No.:BCN4557
CAS No.:88-14-2
- Ingenol-5,20-acetonide-3-O-angelate
Catalog No.:BCN8458
CAS No.:87980-68-5
- UFP 803
Catalog No.:BCC5917
CAS No.:879497-82-2
- Prionoid E
Catalog No.:BCN3161
CAS No.:879324-78-4
- Prionoid D
Catalog No.:BCN3160
CAS No.:879324-77-3
- 4-Hydroxybenzaldehyde rhamnoside
Catalog No.:BCN7625
CAS No.:88086-86-6
- Phrixotoxin 3
Catalog No.:BCC6328
CAS No.:880886-00-0
- Notoginsenoside Fa
Catalog No.:BCN3854
CAS No.:88100-04-3
- HDS 029
Catalog No.:BCC7441
CAS No.:881001-19-0
- 30-Hydroxygambogic acid
Catalog No.:BCN3081
CAS No.:881027-36-7
- Notoginsenoside Fe
Catalog No.:BCN3852
CAS No.:88105-29-7
- Valganciclovir
Catalog No.:BCC2026
CAS No.:88110-89-8
- JNJ-26854165 (Serdemetan)
Catalog No.:BCC2240
CAS No.:881202-45-5
- Notoginsenoside Fc
Catalog No.:BCN3853
CAS No.:88122-52-5
- KU-0060648
Catalog No.:BCC1110
CAS No.:881375-00-4
- Daphniyunnine A
Catalog No.:BCN4428
CAS No.:881388-87-0
- Daphniyunnine B
Catalog No.:BCN4429
CAS No.:881388-88-1
Risk Factors for Infection After Knee Arthroscopy: Analysis of 595,083 Cases From 3 United States Databases.[Pubmed:27407027]
Arthroscopy. 2016 Dec;32(12):2556-2561.
PURPOSE: To identify and quantify patient- and procedure-related risk factors for post-arthroscopic knee infections using a large dataset. METHODS: An administrative health care database including 8 years of records from 2 large commercial insurers and Medicare (a 5% random sample) was queried to identify all knee arthroscopies performed on patients aged at least 15 years using Current Procedural Terminology (CPT) codes. Each CPT code was designated as a high- or low-complexity procedure, with the former typically requiring accessory incisions or increased operative time. Deep infections were identified by a CPT code for incision and drainage within 90 days of surgery. Superficial infections were identified by International Classification of Diseases, Ninth Revision infection codes without any record of incision and drainage. Patients were compared based on age, sex, body mass index, tobacco use, presence of diabetes, and Charlson Comorbidity Index. RESULTS: A total of 526,537 patients underwent 595,083 arthroscopic knee procedures. Deep postoperative infections occurred at a rate of 0.22%. Superficial infections occurred at a rate of 0.29%. Tobacco use and morbid obesity were the largest risk factors for deep and superficial infections, respectively (P < .001; relative risk of 1.90 and 2.19, respectively). There were also higher infection rates among patients undergoing relatively high-complexity arthroscopies, men, obese patients, diabetic patients, and younger patients (in order of decreasing relative risk). Increased Charlson Comorbidity Index was associated with superficial and total infections (P < .001). CONCLUSIONS: Post-arthroscopic knee infections were more frequent among morbidly obese patients, tobacco users, patients undergoing relatively complex procedures, men, obese patients, diabetic patients, relatively young patients, and patients with increased comorbidity burdens in this study population. This knowledge may allow more informed preoperative counseling, aid surgeons in patient selection, and facilitate infection prevention by targeting individuals with higher inherent risk. LEVEL OF EVIDENCE: Level IV, cross-sectional study.
Prognostic Relevance of Mucinous Subtype in a Population-based Propensity Score Analysis of 40,083 Rectal Cancer Patients.[Pubmed:26714956]
Ann Surg Oncol. 2016 May;23(5):1576-86.
BACKGROUND: The prognostic relevance of mucinous histology in colorectal cancer remains unclear, especially for rectal neoplasms. The objective of this study was to evaluate if mucinous subtype has a relevant impact on overall survival (OS) and cancer-specific survival (CSS) of patients with adenocarcinomas of the rectum. METHODS: On the basis of the data set of the Surveillance, Epidemiology, and End Results Program of the National Cancer Institute of the United States, patients with rectal cancer between 2004 and 2011 were identified. Risk-adjusted Cox regression analysis and propensity score methods were used to assess OS and CSS. RESULTS: In total, 40,083 patients with stage I-IV rectal cancer, of whom 2483 (6.2 %) had mucinous histology, were included in this study. In unadjusted analysis, the 5-year OS and CSS for patients with a mucinous adenocarcinoma was 54.3 % [95 % confidence interval (CI) 52.0-56.7] and 61.4 % (95 % CI 59.1-63.9) compared to 66.4 % (95 % CI 65.8-67.0) and 74.5 % (95 % CI 73.9-75.1) for patients with nonmucinous adenocarcinoma (P < 0.001). The survival disadvantage persisting in risk-adjusted Cox proportional hazard regression analysis [hazard ratio (HR) 1.23, 95 % CI 1.15-1.31, P < 0.001 and 1.25, 95 % CI 1.16-1.35, P < 0.001) disappeared after propensity score matching (OS: HR = 0.96, 95 % CI 0.76-1.21, P = 0.722; CSS: HR 1.06, 95 % CI 0.80-1.40, P = 0.693). CONCLUSIONS: This population-based, propensity score matched analysis shows that mucinous histology itself does not constrain survival in rectal cancer patients. Therefore, treatment decisions should not be different according to mucinous histology.
Vaccination With Heterologous HIV-1 Envelope Sequences and Heterologous Adenovirus Vectors Increases T-Cell Responses to Conserved Regions: HVTN 083.[Pubmed:26475930]
J Infect Dis. 2016 Feb 15;213(4):541-50.
BACKGROUND: Increasing the breadth of human immunodeficiency virus type 1 (HIV-1) vaccine-elicited immune responses or targeting conserved regions may improve coverage of circulating strains. HIV Vaccine Trials Network 083 tested whether cellular immune responses with these features are induced by prime-boost strategies, using heterologous vectors, heterologous inserts, or a combination of both. METHODS: A total of 180 participants were randomly assigned to receive combinations of adenovirus vectors (Ad5 or Ad35) and HIV-1 envelope (Env) gene inserts (clade A or B) in a prime-boost regimen. RESULTS: T-cell responses to heterologous and homologous insert regimens targeted a similar number of epitopes (ratio of means, 1.0; 95% confidence interval [CI], .6-1.6; P = .91), but heterologous insert regimens induced significantly more epitopes that were shared between EnvA and EnvB than homologous insert regimens (ratio of means, 2.7; 95% CI, 1.2-5.7; P = .01). Participants in the heterologous versus homologous insert groups had T-cell responses that targeted epitopes with greater evolutionary conservation (mean entropy [+/-SD], 0.32 +/- 0.1 bits; P = .003), and epitopes recognized by responders provided higher coverage (49%; P = .035). Heterologous vector regimens had higher numbers of total, EnvA, and EnvB epitopes than homologous vector regimens (P = .02, .044, and .045, respectively). CONCLUSIONS: These data demonstrate that vaccination with heterologous insert prime boosting increased T-cell responses to shared epitopes, while heterologous vector prime boosting increased the number of T-cell epitopes recognized. CLINICAL TRIALS REGISTRATION: NCT01095224.
[Status assessment of preconception health risk exposure among 2 030 083 males in 31 provinces during 2010-2012].[Pubmed:25877026]
Zhonghua Yi Xue Za Zhi. 2015 Jan 20;95(3):176-80.
OBJECTIVE: To assess the exposure status of preconception health risk of Chinese males aged 21-49 years in 31 provinces and analyze the changes of their exposure status during 2010-2012. METHODS: The "Core Indicators of Preconception Health Risk Exposure Status in Chinese reproductive couples" were used to evaluate the preconception health risk of males aged 21-49 years who participated in National Free Preconception Health Examination Project. From January 1, 2010 to December 31, 2012, a total of 2 030 083 males were recruited from 220 pilot counties of 31 provinces. The risks involved three aspects closely correlated with offspring health: behavioral risk, infectious risk and genetic risk. RESULTS: The overall smoking rate of Chinese rural males aged 21-49 years was 31.19% (622 837/1 997 051). They smoked a daily average of 10 cigarettes. The median number of cigarettes smoked was 10 per day and it increased with age. The overall drinking rate was 31.81% (634 556/1 994 925). Meanwhile, median alcohol consumption increased with age. The rate of males with a history of birth defect, family inbreeding and familial genetic disease accounted for 0.12% (2 344/1 994 324), 0.20% (4 043/1 997 707) and 0.94% (18 736/2 000 005) respectively. The rates of males with a history of hepatitis B and sexually transmitted diseases were 0.67% (13 317/1 988 904) and 0.05% (912/1 988 904) respectively. HBsAg positive rate was 6.32% (124 274/1 965 821). And the positive screening rate of Treponema pallidum was 0.37% (7 169/1 947 021). Among three types of risks, the highest risk was behavioral risk while the lowest risk genetic risk. Behavioral risk, genetic risk and sexually transmitted diseases in infectious risk were all lowest in 2012 while HBV infection in infectious risk was lowest in 2010. CONCLUSION: The general status of preconception health is fair for rural males aged 21-49 years in 31 provinces. However, there are still some behavior, genetic and infectious risks. The preconception health risk of rural males showed an overall declining trend during 2010-2012.
Pharmacological reactivation of mutant p53: from protein structure to the cancer patient.[Pubmed:20498645]
Oncogene. 2010 Jul 29;29(30):4245-52.
The p53 tumor suppressor pathway blocks tumor development by triggering apoptosis or cellular senescence in response to oncogenic stress. A large fraction of human tumors carry p53 mutations that disrupt DNA binding of p53 and transcriptional regulation of target genes. Reconstitution of wild-type p53 in vivo triggers rapid elimination of tumors. Therefore, pharmacological reactivation of mutant p53 is a promising strategy for novel cancer therapy. Several approaches for identification of small molecules that target mutant p53 have been applied, including rational design and screening of chemical libraries. The compound PhiKan083 binds with high affinity to a crevice created by the Y220C mutation in p53 and stabilizes the mutant protein. The compound PRIMA-1 (p53 reactivation and induction of massive apoptosis) restores wild-type conformation to mutant p53 by binding to the core and induces apoptosis in human tumor cells. The PRIMA-1 analog APR-246 is currently tested in a clinical trial. Successful development of mutant p53-reactivating anticancer drugs should have a major impact on the treatment of cancer.
Targeted rescue of a destabilized mutant of p53 by an in silico screened drug.[Pubmed:18650397]
Proc Natl Acad Sci U S A. 2008 Jul 29;105(30):10360-5.
The tumor suppressor p53 is mutationally inactivated in approximately 50% of human cancers. Approximately one-third of the mutations lower the melting temperature of the protein, leading to its rapid denaturation. Small molecules that bind to those mutants and stabilize them could be effective anticancer drugs. The mutation Y220C, which occurs in approximately 75,000 new cancer cases per annum, creates a surface cavity that destabilizes the protein by 4 kcal/mol, at a site that is not functional. We have designed a series of binding molecules from an in silico analysis of the crystal structure using virtual screening and rational drug design. One of them, a carbazole derivative (PhiKan083), binds to the cavity with a dissociation constant of approximately 150 muM. It raises the melting temperature of the mutant and slows down its rate of denaturation. We have solved the crystal structure of the protein-PhiKan083 complex at 1.5-A resolution. The structure implicates key interactions between the protein and ligand and conformational changes that occur on binding, which will provide a basis for lead optimization. The Y220C mutant is an excellent "druggable" target for developing and testing novel anticancer drugs based on protein stabilization. We point out some general principles in relationships between binding constants, raising of melting temperatures, and increase of protein half-lives by stabilizing ligands.


