Retigabine dihydrochlorideAntiepileptic compound CAS# 150812-13-8 |
- Etifoxine
Catalog No.:BCC1560
CAS No.:21715-46-8
- Etomidate
Catalog No.:BCC1150
CAS No.:33125-97-2
- Acamprosate calcium
Catalog No.:BCC1327
CAS No.:77337-73-6
- Flumazenil
Catalog No.:BCC1259
CAS No.:78755-81-4
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 150812-13-8 | SDF | Download SDF |
| PubChem ID | 6918229 | Appearance | Powder |
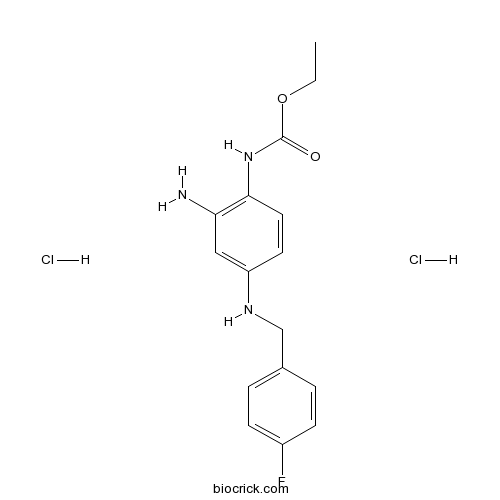
| Formula | C16H20Cl2FN3O2 | M.Wt | 376.25 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | >18.8mg/mL in DMSO | ||
| Chemical Name | ethyl N-[2-amino-4-[(4-fluorophenyl)methylamino]phenyl]carbamate;dihydrochloride | ||
| SMILES | CCOC(=O)NC1=C(C=C(C=C1)NCC2=CC=C(C=C2)F)N.Cl.Cl | ||
| Standard InChIKey | WSGFOWNASITQHJ-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C16H18FN3O2.2ClH/c1-2-22-16(21)20-15-8-7-13(9-14(15)18)19-10-11-3-5-12(17)6-4-11;;/h3-9,19H,2,10,18H2,1H3,(H,20,21);2*1H | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Retigabine dihydrochloride Dilution Calculator

Retigabine dihydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.6578 mL | 13.289 mL | 26.5781 mL | 53.1561 mL | 66.4452 mL |
| 5 mM | 0.5316 mL | 2.6578 mL | 5.3156 mL | 10.6312 mL | 13.289 mL |
| 10 mM | 0.2658 mL | 1.3289 mL | 2.6578 mL | 5.3156 mL | 6.6445 mL |
| 50 mM | 0.0532 mL | 0.2658 mL | 0.5316 mL | 1.0631 mL | 1.3289 mL |
| 100 mM | 0.0266 mL | 0.1329 mL | 0.2658 mL | 0.5316 mL | 0.6645 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
IC50: N/A
Retigabine is a first-in-class K+ channel (KCNQ) opener. KCNQ channels are reported to be expressed predominantly in neurons and are critical determinants of cellular excitability, as shown by the occurrence of human genetic mutations in KCNQ channels which underlie inheritable disorders including the syndrome of benign familial neonatal convulsions.
In vitro: Retigabine was found to combine a novel mode of actions, which were namely potassium channel opening (KCNQ2, KCNQ3 as well as KCNQ4 channels). Retigabine also showed activities with some potentiation of gamma amino butyric acid (GABA)-evoked currents at its higher concentrations [1].
In vivo: Animal models of epileptic seizures showed that retigabine treatment was effective at an oral dose as low as 0.01 mg/kg. Studies performed in mice also indicated that combining retigabine with another anticonvulsant agent leads to an additive effect [1].
Clinical trial: A clinical interaction study showed that there was no pharmacokinetic interaction between phenobarbitone and retigabine in healthy subjects. Thus, no dosage adjustment is likely to be necessary when phenobarbitone and retigabine are coadministered to patients [1].
Reference:
[1] Ferron GM,Patat A,Parks V,Rolan P,Troy SM. Lack of pharmacokinetic interaction between retigabine and phenobarbitone at steady-state in healthy subjects. Br J Clin Pharmacol.2003 Jul;56(1):39-45.
- Retigabine
Catalog No.:BCC6427
CAS No.:150812-12-7
- Tropicamide
Catalog No.:BCC4574
CAS No.:1508-75-4
- Oxybutynin chloride
Catalog No.:BCC4149
CAS No.:1508-65-2
- Gap19
Catalog No.:BCC5599
CAS No.:1507930-57-5
- Drynachromoside A
Catalog No.:BCN7891
CAS No.:1507388-29-5
- Calyxamine B
Catalog No.:BCN1671
CAS No.:150710-72-8
- 5-Methoxycanthin-6-one
Catalog No.:BCN3326
CAS No.:15071-56-4
- Tolvaptan
Catalog No.:BCC5096
CAS No.:150683-30-0
- Fmoc-Lys(Dde)-OH
Catalog No.:BCC3518
CAS No.:150629-67-7
- epi-Eudesmol
Catalog No.:BCN1670
CAS No.:15051-81-7
- L-NIL hydrochloride
Catalog No.:BCC5706
CAS No.:150403-89-7
- o-Methoxycinnamaldehyde
Catalog No.:BCN3860
CAS No.:1504-74-1
- 2,3,24-Trihydroxyolean-12-en-28-oic acid
Catalog No.:BCN1559
CAS No.:150821-16-2
- Nitenpyram
Catalog No.:BCC5559
CAS No.:150824-47-8
- EGF816
Catalog No.:BCC6428
CAS No.:1508250-71-2
- Boc-Orn(Fmoc)-OH
Catalog No.:BCC3429
CAS No.:150828-96-9
- Micromelin
Catalog No.:BCN1672
CAS No.:15085-71-9
- Uralenol-3-methylether
Catalog No.:BCN7993
CAS No.:150853-98-8
- Bis(phenylacetyl) disulfide
Catalog No.:BCC8887
CAS No.:15088-78-5
- Euonymine
Catalog No.:BCN3084
CAS No.:150881-01-9
- Aurantiamide benzoate
Catalog No.:BCN8043
CAS No.:150881-02-0
- D-allo-Ile-OH
Catalog No.:BCC2966
CAS No.:1509-35-9
- Tirofiban hydrochloride monohydrate
Catalog No.:BCC2003
CAS No.:150915-40-5
- 2,4,4'-Trihydroxydihydrochalcone
Catalog No.:BCN7365
CAS No.:15097-74-2
Activation of peripheral KCNQ channels attenuates inflammatory pain.[Pubmed:24555569]
Mol Pain. 2014 Feb 21;10:15.
BACKGROUND: Refractory chronic pain dramatically reduces the quality of life of patients. Existing drugs cannot fully achieve effective chronic pain control because of their lower efficacy and/or accompanying side effects. Voltage-gated potassium channels (KCNQ) openers have demonstrated their analgesic effect in preclinical and clinical studies, and are thus considered to be a potential therapeutic target as analgesics. However, these drugs exhibit a narrow therapeutic window due to their imposed central nerve system (CNS) side effects. To clarify the analgesic effect by peripheral KCNQ channel activation, we investigated whether the analgesic effect of the KCNQ channel opener, retigabine, is inhibited by intracerebroventricular (i.c.v.) administration of the KCNQ channel blocker, 10, 10-bis (4-Pyridinylmethyl)-9(10H) -anthracenone dihydrochloride (XE-991) in rats. RESULTS: Oral administration (p.o.) of retigabine showed an anticonvulsant effect on maximal electronic seizures and an analgesic effect on complete Freund's adjuvant-induced thermal hyperalgesia. However, impaired motor coordination and reduced exploratory behavior were also observed at the analgesic doses of retigabine. Administration (i.c.v.) of XE-991 reversed the retigabine-induced anticonvulsant effect, impaired motor coordination and reduced exploratory behavior but not the analgesic effect. Moreover, intraplantar administration of retigabine or an additional KCNQ channel opener, N-(6-Chloro-pyridin-3-yl)-3,4-difluoro-benzamide (ICA-27243), inhibited formalin-induced nociceptive behavior. CONCLUSIONS: Our findings suggest that the peripheral sensory neuron is the main target for KCNQ channel openers to induce analgesia. Therefore, peripheral KCNQ channel openers that do not penetrate the CNS may be suitable analgesic drugs as they would prevent CNS side effects.
The effects of D-23129, a new experimental anticonvulsant drug, on neurotransmitter amino acids in the rat hippocampus in vitro.[Pubmed:8991783]
Epilepsy Res. 1995 Nov;22(3):167-73.
D-23129 [N-(2-amino-4-(4-fluorobenzylamino)phenyl)carbamic acid ethyl ester] and D-20443 (dihydrochloride of D-23129) are promising anticonvulsant compounds with a broad spectrum activity in animal models of epilepsy. Their effects on de novo synthesis of excitatory (glutamate and aspartate) and inhibitory (GABA) amino acids were studied in rat hippocampal slices. Like phenytoin, carbamazepine, lamotrigine, losigamone, U54494A, and flupirtine, D-23129 and D-20443 were effective in preventing the effects of a chemoconvulsant, 4-aminopyridine, on de novo synthesis of the three amino acids. However, unlike the other compounds, D-23129 and D-20443 also preferentially increased the concentrations of newly synthesized GABA. Their effect on the neosynthesis of GABA was unique, dose dependent, and not tetrodotoxin sensitive. A total of 15 compounds (including standard, new and candidate anticonvulsants) either had no effect on new GABA or decreased it. Therefore, D-23129 and D-20443 exhibited two different effects on de novo synthesis of neurotransmitter amino acids, both of which could potentially be anticonvulsant in nature.
Gateways to clinical trials.[Pubmed:15834459]
Methods Find Exp Clin Pharmacol. 2005 Jan-Feb;27(1):49-77.
Gateways to Clinical Trials is a guide to the most recent clinical trials reported in current literature and congresses. The data in the following tables have been retrieved from the Clinical Trials Knowledge Area of Prous Science Integrity, the drug discovery and development portal, http://integrity.prous.com. This issue focuses on the following selection of drugs:[188Re]-HDD; A-179578, adalimumab, AK-602, albumin interferon alfa, alfimeprase, amelubant, anakinra, anti-CD2 MAb, APD-356, aripiprazole, atvogen; Bimatoprost, bimosiamose, BLP-25, brivaracetam; Caspofungin acetate, cilansetron, CMV vaccine (bivalent), conivaptan hydrochloride, Cypher; Darbepoetin alfa, darifenacin hydrobromide, D-D4FC, decitabine, dnaJP1, doranidazole, dronedarone hydrochloride; Efalizumab, efaproxiral sodium, emtricitabine, Endeavor, entecavir, erlotinib hydrochloride, escitalopram oxalate, etoricoxib, etravirine, ezetimibe; Fampridine, fenretinide, ferumoxtran-10, forodesine hydrochloride; Gantacurium chloride, gemi-floxacin mesilate, Glyminox, GW-501516; HBV-ISS, hepavir B, human insulin, HuMax-CD20, hyaluronic acid, HyCAMP; Icatibant, IDEA-070, IGN-311, imatinib mesylate, insulin detemir, insulin glargine, insulin glulisine; Lapatinib, lasofoxifene tartrate, LB-80380, liarozole fumarate, liposome encapsulated doxorubicin, lumiracoxib, LY-570310; MC-1, melatonin, merimepodib, metanicotine, midostaurin; Natalizumab, nicotine conjugate vaccine, NYVAC-HIV C; Patupilone, peginterferon alfa-2a, peginterferon alfa-2b, peginterferon alfa-2b/ribavirin, pelitinib, Peru-15, pexelizumab, PHP, pimecrolimus, prednisolone sodium metasulfobenzoate; Recombinant alfa1-antitrypsin (AAT), retigabine, rHA influenza vaccine, rifalazil, rofecoxib, rosiglitazone maleate/Metformin hydrochloride, rostaporfin, rosuvastatin calcium, rubitecan; Selenite sodium, semilente insulin, SMP-797, sorafenib; Talampanel, tenofovir disoproxil fumarate, TER-199, tiotropium bromide, torcetrapib, treprostinil sodium, TTA; ValboroPro, valdecoxib, val-mCyd, valtorcitabine dihydrochloride: XP-828L.
Gateways to clinical trials.[Pubmed:20140276]
Methods Find Exp Clin Pharmacol. 2009 Dec;31(10):661-700.
[Methoxy-(11)C]PD-153035, 2-Methoxyestradiol; Adalimumab, Adecatumumab, Adefovir dipivoxil, ADH-1, ADX-10059, Aflibercept, AIR-human growth hormone, Aliskiren fumarate, AMG-221, Amlodipine besylate/olmesartan medoxomil, Aprepitant; Bavituximab, Bevacizumab, Bexarotene, BIBW-2992, BMS-690514, Bortezomib, Bosentan, Briakinumab; Capecitabine, Certolizumab pegol, Cetuximab, Cholecalciferol, Choline fenofibrate, Chorionic gonadotropin (human), Cixutumumab, Clopidogrel, CP-690550 citrate; Dabigatran, Dacetuzumab, Daclizumab, Dapagliflozin, Darbepoetin alfa, Dasatinib, Denosumab; Efavirenz, Elisidepsin, Enoxaparin, Enzastaurin hydrochloride, Eribulin mesilate, Erlotinib hydrochloride, Everolimus, Exenatide; Fenobam, Figitumumab, Filibuvir, Fondaparinux sodium, Fresolimumab; Gefitinib, Golimumab, Golnerminogene pradenovec; Ifosfamide, Imatinib mesylate, Ipilimumab, Ivabradine hydrochloride, Ixabepilone; Lapatinib ditosylate, Lenalidomide, Levocetirizine dihydrochloride, Liposomal vincristine, Liraglutide; M-118, Masitinib mesylate, Metformin hydrochloride, Micafungin sodium, Moxifloxacin hydrochloride; Neratinib; Oblimersen sodium, Ofatumumab, Olmesartan medoxomil; Paclitaxel nanoparticles, Palifosfamide lysine, Panobacumab, Panobinostat, Patupilone, Peginterferon alfa-2a, Pegylated arginine deiminase 20000, Piclozotan hydrochloride hydrate, Pixantrone maleate, Prasterone, Prasugrel, Prednisone, Progesterone, Prucalopride, pVGI.1 (VEGF-2); Retigabine, rhFSH, Rituximab, Rivaroxaban, Rosuvastatin calcium; Salinosporamide A, Selumetinib, Sipuleucel-T, Somatropin, Sorafenib, SSR-244738, Sunitinib malate; Tamoxifen citrate, Teduglutide, Telavancin hydrochloride, Telmisartan, Telmisartan/amlodipine, Telmisartan/hydrochlorothiazide, Temsirolimus, Tenofovir disoproxil fumarate, Tipifarnib, Tolvaptan, Trastuzumab, Trastuzumab-MCC-DM1, Travoprost, Tremelimumab; Valsartan/amlodipine besylate, Valsartan/amlodipine besylate/hydrochlorothiazide, Valsartan/hydrochlorothiazide, Vandetanib, Vorinostat.


