1-HexadecanolCAS# 36653-82-4 |
- CYT387 sulfate salt
Catalog No.:BCC1506
CAS No.:1056636-06-6
- Baricitinib phosphate
Catalog No.:BCC1401
CAS No.:1187595-84-1
- JAK2 Inhibitor V, Z3
Catalog No.:BCC1667
CAS No.:195371-52-9
- Bardoxolone methyl
Catalog No.:BCC1400
CAS No.:218600-53-4
- Ruxolitinib (INCB018424)
Catalog No.:BCC1276
CAS No.:941678-49-5
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 36653-82-4 | SDF | Download SDF |
| PubChem ID | 2682 | Appearance | Powder |
| Formula | C16H34O | M.Wt | 242.44 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in DMSO > 10 mM | ||
| Chemical Name | hexadecan-1-ol | ||
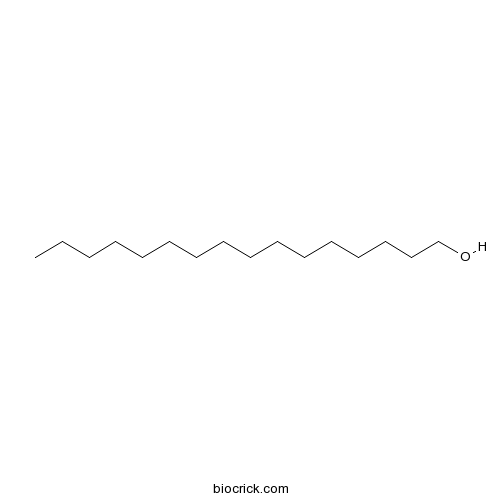
| SMILES | CCCCCCCCCCCCCCCCO | ||
| Standard InChIKey | BXWNKGSJHAJOGX-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C16H34O/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17/h17H,2-16H2,1H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

1-Hexadecanol Dilution Calculator

1-Hexadecanol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.1247 mL | 20.6237 mL | 41.2473 mL | 82.4946 mL | 103.1183 mL |
| 5 mM | 0.8249 mL | 4.1247 mL | 8.2495 mL | 16.4989 mL | 20.6237 mL |
| 10 mM | 0.4125 mL | 2.0624 mL | 4.1247 mL | 8.2495 mL | 10.3118 mL |
| 50 mM | 0.0825 mL | 0.4125 mL | 0.8249 mL | 1.6499 mL | 2.0624 mL |
| 100 mM | 0.0412 mL | 0.2062 mL | 0.4125 mL | 0.8249 mL | 1.0312 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
1-Hexadecanol is a fatty alcohol used to make other chemicals.
- Semialactone
Catalog No.:BCN5422
CAS No.:366450-46-6
- 1,4-Benzodioxan-2-carboxylic acid
Catalog No.:BCC8421
CAS No.:3663-80-7
- Mubritinib (TAK 165)
Catalog No.:BCC4513
CAS No.:366017-09-6
- AY 9944 dihydrochloride
Catalog No.:BCC3940
CAS No.:366-93-8
- Procarbazine HCl
Catalog No.:BCC4452
CAS No.:366-70-1
- H-Arg-OEt.2HCl
Catalog No.:BCC2860
CAS No.:36589-29-4
- Ac-Tyr-OEt.H2O
Catalog No.:BCC3121
CAS No.:36546-50-6
- Cycloheterophyllin
Catalog No.:BCN4640
CAS No.:36545-53-6
- Guaiacin
Catalog No.:BCN4127
CAS No.:36531-08-5
- Serpentinine
Catalog No.:BCN5325
CAS No.:36519-42-3
- Emiline
Catalog No.:BCN2080
CAS No.:36506-99-7
- beta-Costic acid
Catalog No.:BCN5324
CAS No.:3650-43-9
- Rivaroxaban
Catalog No.:BCC2292
CAS No.:366789-02-8
- Cussosaponin C
Catalog No.:BCN2895
CAS No.:366814-42-8
- Pulchinenoside E
Catalog No.:BCN8165
CAS No.:366814-43-9
- Isorhamnetin 3-glucuronide
Catalog No.:BCN2709
CAS No.:36687-76-0
- Benzyl 2,4-dihydroxyphenyl ketone
Catalog No.:BCC8867
CAS No.:3669-41-8
- 8-Shogaol
Catalog No.:BCN3266
CAS No.:36700-45-5
- Beta-Solamarine
Catalog No.:BCN2693
CAS No.:3671-38-3
- Boc-Phe-Osu
Catalog No.:BCC2601
CAS No.:3674-06-4
- Boc-D-Phe-Osu
Catalog No.:BCC2600
CAS No.:3674-18-8
- Lurasidone
Catalog No.:BCC9013
CAS No.:367514-87-2
- Lurasidone HCl
Catalog No.:BCC4458
CAS No.:367514-88-3
- AR-M 1896
Catalog No.:BCC5931
CAS No.:367518-31-8
1H NMR investigations of inclusion complexes between beta-cyclodextrin and 1-hexadecanol.[Pubmed:18045091]
Curr Drug Discov Technol. 2007 Dec;4(4):295-7.
The inclusion complex between beta-CD and 1-Hexadecanol is synthesized and identified via (1)H NMR spectrum. The possible conformation of the inclusion complex is figured out. Via MM2 calculations, the possibility of complexation is verified.
Metabolic engineering of Saccharomyces cerevisiae to improve 1-hexadecanol production.[Pubmed:25466225]
Metab Eng. 2015 Jan;27:10-19.
Fatty alcohols are important components of a vast array of surfactants, lubricants, detergents, pharmaceuticals and cosmetics. We have engineered Saccharomyces cerevisiae to produce 1-Hexadecanol by expressing a fatty acyl-CoA reductase (FAR) from barn owl (Tyto alba). In order to improve fatty alcohol production, we have manipulated both the structural genes and the regulatory genes in yeast lipid metabolism. The acetyl-CoA carboxylase gene (ACC1) was over-expressed, which improved 1-Hexadecanol production by 56% (from 45mg/L to 71mg/L). Knocking out the negative regulator of the INO1 gene in phospholipid metabolism, RPD3, further enhanced 1-Hexadecanol production by 98% (from 71mg/L to 140mg/L). The cytosolic acetyl-CoA supply was next engineered by expressing a heterologous ATP-dependent citrate lyase, which increased the production of 1-Hexadecanol by an additional 136% (from 140mg/L to 330mg/L). Through fed-batch fermentation using resting cells, over 1.1g/L 1-Hexadecanol can be produced in glucose minimal medium, which represents the highest titer reported in yeast to date.
Metabolic engineering of Saccharomyces cerevisiae to produce 1-hexadecanol from xylose.[Pubmed:26830023]
Microb Cell Fact. 2016 Feb 1;15:24.
BACKGROUND: An advantageous but challenging approach to overcome the limited supply of petroleum and relieve the greenhouse effect is to produce bulk chemicals from renewable materials. Fatty alcohols, with a billion-dollar global market, are important raw chemicals for detergents, emulsifiers, lubricants, and cosmetics production. Microbial production of fatty alcohols has been successfully achieved in several industrial microorganisms. However, most of the achievements were using glucose, an edible sugar, as the carbon source. To produce fatty alcohols in a renewable manner, non-edible sugars such as xylose will be a more appropriate feedstock. RESULTS: In this study, we aim to engineer a Saccharomyces cerevisiae strain that can efficiently convert xylose to fatty alcohols. To this end, we first introduced the fungal xylose utilization pathway consisting of xylose reductase (XR), xylitol dehydrogenase (XDH), and xylulose kinase (XKS) into a fatty alcohol-producing S. cerevisiae strain (XF3) that was developed in our previous studies to achieve 1-Hexadecanol production from xylose at 0.4 g/L. We next applied promoter engineering on the xylose utilization pathway to optimize the expression levels of XR, XDH, and XKS, and increased the 1-Hexadecanol titer by 171 %. To further improve the xylose-based fatty alcohol production, two optimized S. cerevisiae strains from promoter engineering were evolved with the xylose as the sole carbon source. We found that the cell growth rate was improved at the expense of decreased fatty alcohol production, which indicated 1-Hexadecanol was mainly produced as a non-growth associated product. Finally, through fed-batch fermentation, we successfully achieved 1-Hexadecanol production at over 1.2 g/L using xylose as the sole carbon source, which represents the highest titer of xylose-based 1-Hexadecanol reported in microbes to date. CONCLUSIONS: A fatty alcohol-producing S. cerevisiae strain was engineered in this study to produce 1-Hexadecanol from xylose. Although the xylose pathway we developed in this study could be further improved, this proof-of-concept study, for the first time to our best knowledge, demonstrated that the xylose-based fatty alcohol could be produced in S. cerevisiae with potential applications in developing consolidated bioprocessing for producing other fatty acid-derived chemicals.
Effects of dioctadecyl dimethyl ammonium chloride on the rheological behavior of behenyl trimethyl ammonium chloride/1-hexadecanol/water ternary system.[Pubmed:16839564]
J Colloid Interface Sci. 2006 Oct 1;302(1):341-6.
The effects of dioctadecyl dimethyl ammonium chloride (DODAC) on the rheological properties of ternary systems consisting of behenyl trimethyl ammonium chloride (C22TAC), 1-Hexadecanol (C16OH), and water are studied to improve the long-term stability and to establish the preparation method of cosmetic products. The basic ternary systems behave as solids at low stresses, due to the formation of lamella liquid crystals called alpha gel. The additions of DODAC to ternary systems cause the rupture of alpha gel structures and transformation to vesicles. The structures of molecular assemblies are confirmed through particle size distribution, differential scanning calorimetry, and freeze fracture electron microscopy. The vesicle structures formed in coexistence of single-chain surfactant and double-chain surfactant are highly stable. Because the quaternary systems are considered to be constructed by the mixtures of alpha gel and vesicles at the appropriate concentrations of DODAC, the rheology can be controlled by the structural balance between them.


