2',4'-Dihydroxy-3',6'-dimethoxychalconeCAS# 129724-43-2 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 129724-43-2 | SDF | Download SDF |
| PubChem ID | 98218661 | Appearance | Yellow powder |
| Formula | C17H16O5 | M.Wt | 300.3 |
| Type of Compound | Chalcones | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
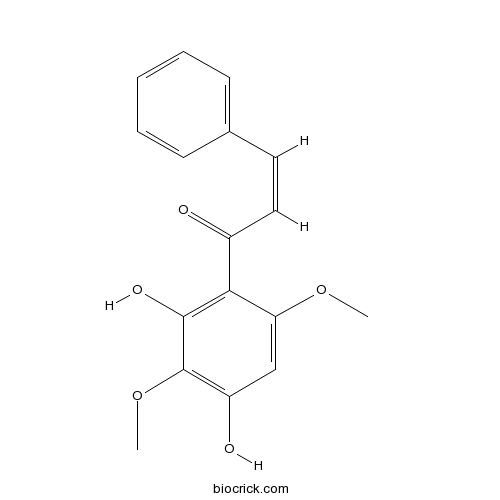
| Chemical Name | (Z)-1-(2,4-dihydroxy-3,6-dimethoxyphenyl)-3-phenylprop-2-en-1-one | ||
| SMILES | COC1=CC(=C(C(=C1C(=O)C=CC2=CC=CC=C2)O)OC)O | ||
| Standard InChIKey | BXMWIRIMYNWIGQ-HJWRWDBZSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. 2',4'-Dihydroxy-3',6'-dimethoxychalcone has cytotoxic activities against cancer cells. 2. 2',4'-Dihydroxy-3',6'-dimethoxychalcone has anti-inflammatory activity and anti-cholinesterase activity, it has significant NO inhibitory activity. |
| Targets | NO |

2',4'-Dihydroxy-3',6'-dimethoxychalcone Dilution Calculator

2',4'-Dihydroxy-3',6'-dimethoxychalcone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.33 mL | 16.65 mL | 33.3 mL | 66.6001 mL | 83.2501 mL |
| 5 mM | 0.666 mL | 3.33 mL | 6.66 mL | 13.32 mL | 16.65 mL |
| 10 mM | 0.333 mL | 1.665 mL | 3.33 mL | 6.66 mL | 8.325 mL |
| 50 mM | 0.0666 mL | 0.333 mL | 0.666 mL | 1.332 mL | 1.665 mL |
| 100 mM | 0.0333 mL | 0.1665 mL | 0.333 mL | 0.666 mL | 0.8325 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Aripiprazole
Catalog No.:BCC5034
CAS No.:129722-12-9
- Dofequidar
Catalog No.:BCC4176
CAS No.:129716-58-1
- ZD 7114 hydrochloride
Catalog No.:BCC6852
CAS No.:129689-28-7
- 3,4-Dihydroxybisabola-1,10-diene
Catalog No.:BCN7326
CAS No.:129673-87-6
- 3-Hydroxybisabola-1,10-dien-9-one
Catalog No.:BCN7325
CAS No.:129673-86-5
- GR 82334
Catalog No.:BCC5802
CAS No.:129623-01-4
- Nevirapine
Catalog No.:BCC3820
CAS No.:129618-40-2
- Satraplatin
Catalog No.:BCC5356
CAS No.:129580-63-8
- Goniopypyrone
Catalog No.:BCN3957
CAS No.:129578-07-0
- Tolfenpyrad
Catalog No.:BCC8069
CAS No.:129558-76-5
- Verteporfin
Catalog No.:BCC3690
CAS No.:129497-78-5
- 3,4-Diacetoxycinnamamide
Catalog No.:BCN6157
CAS No.:129488-34-2
- 3-Hydroxylanost-9(11)-24-dien-26-oic acid
Catalog No.:BCN1586
CAS No.:129724-83-0
- Anemoside A3
Catalog No.:BCN2328
CAS No.:129724-84-1
- Anemoside B4
Catalog No.:BCN1276
CAS No.:129741-57-7
- ODM-201
Catalog No.:BCC3796
CAS No.:1297538-32-9
- MC 976
Catalog No.:BCC1734
CAS No.:129831-99-8
- Amicarbazone
Catalog No.:BCC5464
CAS No.:129909-90-6
- Dapoxetine HCl
Catalog No.:BCC5064
CAS No.:129938-20-1
- Iloperidone hydrochloride
Catalog No.:BCC4212
CAS No.:1299470-39-5
- Senecionine
Catalog No.:BCN2129
CAS No.:130-01-8
- 1,4-Naphthoquinone
Catalog No.:BCN8420
CAS No.:130-15-4
- Thioridazine HCl
Catalog No.:BCC3869
CAS No.:130-61-0
- Protopine
Catalog No.:BCN6165
CAS No.:130-86-9
Cytotoxicity and modes of action of 4'-hydroxy-2',6'-dimethoxychalcone and other flavonoids toward drug-sensitive and multidrug-resistant cancer cell lines.[Pubmed:25442273]
Phytomedicine. 2014 Oct 15;21(12):1651-7.
INTRODUCTION: Resistance of cancer to chemotherapy is a main cause in treatment failure. Naturally occurring chalcones possess a wide range of biological activities including anti-cancer effects. In this work, we evaluated the antiproliferative activity of three chalcones [4'-hydroxy-2',6'-dimethoxychalcone (1), cardamomin (2), 2',4'-dihydroxy-3',6'-dimethoxychalcone (3)], and four flavanones [(S)-(-)-pinostrobin (4), (S)-(-)-onysilin (5) and alpinetin (6)] toward nine cancer cell lines amongst which were multidrug resistant (MDR) types. METHODS: The resazurin reduction assay was used to detect the antiproliferative activity of the studied samples whilst flow cytometry for the mechanistic studies of the most active molecule (1). RESULTS: IC50 values in a range of 2.54 muM against CEM/ADR5000 leukemia cells to 58.63 muM toward hepatocarcinoma HepG2 cells were obtained with 1. The lowest IC50 values of 8.59 muM for 2 and 10.67 muM for 3 were found against CCRF-CEM cells leukemia cells, whilst the corresponding values were above 80 muM for 4 and 6. P-glycoprotein-expressing and multidrug-resistant CEM/ADR5000 cells were much more sensitive toward compound 1 than toward doxorubicin and low cross-resistance or even collateral sensitivity was observed in other drug-resistent cell lines to this compound. Normal liver AML12 cells were more resistant to the studied compounds than HepG2 liver cancer cells, indicating tumor specificity at least to some extent. Compound 1 arrested the cell cycle between Go/G1 phase, strongly induced apoptosis via disrupted mitochondrial membrane potential (MMP) and increased production of reactive oxygen species (ROS) in the studied leukemia cell line. CONCLUSIONS: Chalcone 1 was the best tested cytotoxic molecule and further studies will be performed in order to envisage its possible use in the fight against multifactorial resistant cancer cells.
Cytotoxicity and antimicrobial activity of the methanol extract and compounds from Polygonum limbatum.[Pubmed:22495442]
Planta Med. 2012 May;78(8):787-92.
The present study was designed to investigate the antimicrobial activity and the cytotoxicity of the methanol extract (PLA) as well as fractions (PLA1-4) and compounds [cardamomin (1), (+/-)-polygohomoisoflavanone (2), (S)-(-)-pinostrobin (3), 2',4'-dihydroxy-3',6'-dimethoxychalcone (4), (2S)-(-)-5-hydroxy-6,7-dimethoxyflavanone (5), and (2S)-(-)-5,7-dimethoxyflavanone (6)] obtained from leaves of Polygonum limbatum. The microbroth dilution was used to determine the minimal inhibitory concentration (MIC) of the samples against 11 microbial strains including Candida albicans, C. krusei, C. tropicalis, Aspergillus fumigatus, Pseudomonas aeruginosa, Escherichia coli, vancomycin-resistant Enterococcus faecalis (VRE), Staphylococcus aureus, methicillin-resistant S. aureus (MRSA), S.epidermidis, and Mycobacterium tuberculosis H37Rv. The sulphorhodamine B cell growth inhibition assay was used to assess the cytotoxicity of the above samples on lung A549 adenocarcinoma, breast carcinoma MCF-7, prostate carcinoma PC-3, cervical carcinoma HeLa, and the acute monocytic leukemia cell line THP-1. The results of the MIC determination indicated that, apart from fraction PLA3, all other fractions as well as PLA and compound 3 were selectively active. MIC values were noted on 100 % of the 11 tested microorganisms for fraction PLA3, 72.7 % for PLA, fraction PLA2, and compound 4, 63.6 % for PLA1, and 54.5 % for fraction PLA4. The results of the cytotoxicity assay revealed that, except for A459 cells, more than 50 % inhibition of the proliferation was obtained with each of the tested samples on at least one of the four other cell lines. IC(5)(0) values below 4 microg/mL were obtained with 1 and 4 on THP-1 cells. The overall results of the present study provided baseline information for the possible use of Polygonum limbatum as well as some of the isolated compounds for the control of cancer diseases and mostly leukemia.
Anti-inflammatory and anticholinesterase activity of six flavonoids isolated from Polygonum and Dorstenia species.[Pubmed:26048035]
Arch Pharm Res. 2017 Oct;40(10):1129-1134.
This study was aimed at investigating the anti-inflammatory and anticholinesterase activity of six naturally occurring flavonoids: (-) pinostrobin (1), 2',4'-dihydroxy-3',6'-dimethoxychalcone (2), 6-8-diprenyleriodictyol (3), isobavachalcone (4), 4-hydroxylonchocarpin (5) and 6-prenylapigenin (6). These compounds were isolated from Dorstenia and Polygonum species used traditionally to treat pain. The anti-inflammatory activity was determined by using the Griess assay and the 15-lipoxygenase inhibitory activity was determined with the ferrous oxidation-xylenol orange assay. Acetylcholinesterase inhibition was determined by the Ellman's method. At the lowest concentration tested (3.12 microg/ml), compounds 2, 3 and 4 had significant NO inhibitory activity with 90.71, 84.65 and 79.57 % inhibition respectively compared to the positive control quercetin (67.93 %). At this concentration there was no significant cytotoxicity against macrophages with 91.67, 72.86 and 70.86 % cell viability respectively, compared to 73.1 % for quercetin. Compound 4 had the most potent lipoxygenase inhibitory activity (IC50 of 25.92 microg/ml). With the exception of (-) pinostrobin (1), all the flavonoids had selective anticholinesterase activity with IC50 values ranging between 5.93 and 8.76 microg/ml compared to the IC50 4.94 microg/ml of eserine the positive control. These results indicate that the studied flavonoids especially isobavachalcone are potential anti-inflammatory natural products that may have the potential to be developed as therapeutic agents against inflammatory conditions and even Alzheimer's disease.


