3CAIAKT inhibitor CAS# 28755-03-5 |
- Pyridostatin
Catalog No.:BCC1875
CAS No.:1085412-37-8
- 360A iodide
Catalog No.:BCC1308
CAS No.:737763-37-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 28755-03-5 | SDF | Download SDF |
| PubChem ID | 152961 | Appearance | Powder |
| Formula | C10H8ClNO | M.Wt | 193.63 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | DMSO : 250 mg/mL (1291.12 mM; Need ultrasonic) | ||
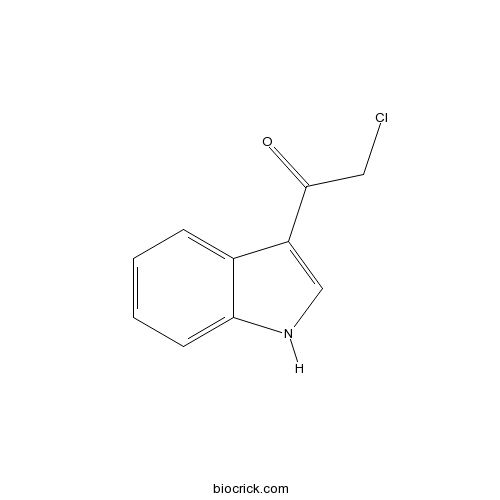
| Chemical Name | 2-chloro-1-(1H-indol-3-yl)ethanone | ||
| SMILES | C1=CC=C2C(=C1)C(=CN2)C(=O)CCl | ||
| Standard InChIKey | LLZQFAXTCYDVTR-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C10H8ClNO/c11-5-10(13)8-6-12-9-4-2-1-3-7(8)9/h1-4,6,12H,5H2 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 3CAI is a potent and specific AKT1 and AKT2 inhibitor.In Vitro:3CAI is a potential inhibitor of AKT. Based on these screening data, the effect of 3CAI on the kinase activities of AKT1, MEK1, JNK1, ERK1 and TOPK is tested using in vitro kinase assays. The results show that 3CAI (1 μM) suppresses only AKT1 kinase activity and the other kinases tested are not affected by 3CAI. 3CAI is a much more potent AKT1 inhibitor than PI3K (60% inhibition at 1 vs 10 μM, respectively). 3CAI substantially suppresses AKT1 activity as well as AKT2 activity in a dose dependent manner. 3CAI inhibits down-stream targets of AKT and induces apoptosis. AKT-mediated phosphorlyation site of mTOR (Ser2448) and GSK3β (Ser9) are substantially decreased by 3CAI in a time-dependent manner. Furthermore, pro-apoptotic marker proteins p53 and p21 are also upregulated by 3CAI after 12 or 24 h of treatment. HCT116 and HT29 colon cancer cells are seeded on 6 cm dishes in 1% FBS/McCoy's 5A (HCT116) with 3CAI (4 μM), I3C or the AKT inhibitor and then incubated for 4 days. Results show that the number of apoptotic cells is significantly increased by 3CAI in HCT116 and HT29 colon cancer cells compared with untreated control cells[1].In Vivo:To examine the antitumor activity of 3CAI in vivo, HCT116 cancer cells are injected into the right flank of individual athymic nude mice. Mice are orally administered 3CAI at 20 or 30 mg/kg, I3C at 100 mg/kg, or vehicle 5 times a week for 21 days. Treatment of mice with 30 mg/kg of 3CAI significantly suppresses HCT116 tumor growth by 50% relative to the vehicle-treated group (p<0.05). Remarkably, mice seem to tolerate treatment with these doses of 3CAI without overt signs of toxicity or significant loss of body weight compared with vehicle-treated group. Expression of these AKT-target proteins is strongly suppressed by 30 mg/kg of 3CAI in tumor tissues[1]. References: | |||||

3CAI Dilution Calculator

3CAI Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 5.1645 mL | 25.8224 mL | 51.6449 mL | 103.2898 mL | 129.1122 mL |
| 5 mM | 1.0329 mL | 5.1645 mL | 10.329 mL | 20.658 mL | 25.8224 mL |
| 10 mM | 0.5164 mL | 2.5822 mL | 5.1645 mL | 10.329 mL | 12.9112 mL |
| 50 mM | 0.1033 mL | 0.5164 mL | 1.0329 mL | 2.0658 mL | 2.5822 mL |
| 100 mM | 0.0516 mL | 0.2582 mL | 0.5164 mL | 1.0329 mL | 1.2911 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Target: Akt
IC50: 1 μM
3CAI is a potent allosteric and specific inhibitor of AKT, which directly binds with AKT1 or AKT2 in an ATP noncompetitive manner and shows 60% inhibition of AKT1 kinase activity at 1 μM [1]. The serine/threonine kinase AKT includes three members, AKT1, AKT2, and AKT3. AKT plays a critical role in promoting transformation and chemoresistance through inducing proliferation and inhibiting apoptosis. Therefore, AKT is regarded as a potential target for cancer therapy.
In vitro: 3CAI (1 and 4 μM) significantly inhibited AKT1 kinase activity and decreased expression of AKT direct downstream targets including mTOR and GSK3β, respectively. 3CAI (4 μM) efficiently induced growth inhibition and apoptosis in HCT116 or HCT29 colon cancer cells [1].
In vivo: 3CAI (30 mg/kg, oral administration) significantly suppressed colon cancer growth in an in vivo xenograft mouse model and inhibited the expression of AKT-target protein such as mTOR and GSK3βin HCT116 colon tumor tissues [1].
Reference:
1. Kim DJ, Reddy K, Kim MO, Li Y, Nadas J, Cho YY, et al. (3-Chloroacetyl)-indole, a novel allosteric AKT inhibitor, suppresses colon cancer growth in vitro and in vivo. Cancer Prev Res (Phila). 2011;4(11):1842-51.
- Scriptaid
Catalog No.:BCC2163
CAS No.:287383-59-9
- Oxcarbazepine
Catalog No.:BCC5077
CAS No.:28721-07-5
- MA 2029
Catalog No.:BCC7983
CAS No.:287206-61-5
- PD 180970
Catalog No.:BCC3894
CAS No.:287204-45-9
- NCX 4040
Catalog No.:BCC7944
CAS No.:287118-97-2
- Ezatiostat hydrochloride
Catalog No.:BCC4259
CAS No.:286942-97-0
- Fesoterodine Fumarate
Catalog No.:BCC4584
CAS No.:286930-03-8
- 6'-Amino-3',4'-(methylenedioxy)acetophenone
Catalog No.:BCC8760
CAS No.:28657-75-2
- Epoxylathyrol
Catalog No.:BCN5382
CAS No.:28649-60-7
- Euphorbiasteroid
Catalog No.:BCN2781
CAS No.:28649-59-4
- Multicaulisin
Catalog No.:BCN7840
CAS No.:286461-76-5
- L-838,417
Catalog No.:BCC7617
CAS No.:286456-42-6
- Apigenin 5-O-beta-D-glucopyranoside
Catalog No.:BCN5185
CAS No.:28757-27-9
- Rosuvastatin
Catalog No.:BCC4139
CAS No.:287714-41-4
- 4,5,6,7-Tetrahydrothieno [3,2,c]pyridine hydrochloride
Catalog No.:BCC8664
CAS No.:28783-41-7
- Nordihydrocapsaicin
Catalog No.:BCN2387
CAS No.:28789-35-7
- H-Cys(Acm)-OH.HCl
Catalog No.:BCC2903
CAS No.:28798-28-9
- Peptone, bacteriological
Catalog No.:BCC1210
CAS No.:288-88-0
- Tetrazole
Catalog No.:BCC2847
CAS No.:288-94-8
- Heraclenin
Catalog No.:BCN5187
CAS No.:2880-49-1
- 4,7-Bis(5-bromo-2-thienyl)-2,1,3-benzothiadiazole
Catalog No.:BCC8668
CAS No.:288071-87-4
- Fraxinellone
Catalog No.:BCN1272
CAS No.:28808-62-0
- Cixiophiopogon A
Catalog No.:BCN2778
CAS No.:288143-27-1
- SB408124
Catalog No.:BCC4972
CAS No.:288150-92-5
Excited-State Proton Transfer in 3-Cyano-7-azaindole: From Aqueous Solution to Ice.[Pubmed:29457903]
J Phys Chem A. 2018 Mar 8;122(9):2479-2484.
We investigated the excited-state proton transfer (ESPT) reaction for 3-cyano-7-azaindole (3CAI) in aqueous solution and in ice. 3CAI undergoes water-catalyzed ESPT in the aqueous solution, giving normal (355 nm) and proton transfer tautomer ( approximately 472 nm) emission bands. Detailed temperature-dependent studies showed that the values of activation free energy (Delta G(double dagger)) were similar between N-H and N-D isotopes. Therefore, water-catalyzed ESPT involves a stepwise mechanism incorporating solvation equilibrium ( Keq) to form a 1:1 (molar ratio) water:3CAI cyclic hydrogen-bonded complex as an intermediate, followed by perhaps proton tunneling reaction. In sharp contrast, 3CAI in ice undergoes entirely different photophysical properties, in which 3CAI self-organizes to form a double-hydrogen-bonded dimers at the grain boundary of the polycrystalline. Upon excitation, the dimer proceeds with a fast excited-state double proton transfer reaction, giving rise to solely a tautomer emission ( approximately 450 nm). The distinct difference in ESPT properties between water and ice makes azaindoles feasible for the investigation of water-ice interface property.
TRPC3- and ETB receptor-mediated PI3K/AKT activation induces vasogenic edema formation following status epilepticus.[Pubmed:28764936]
Brain Res. 2017 Oct 1;1672:58-64.
Status epilepticus (SE, a prolonged seizure activity) is a high risk factor of developing vasogenic edema, which leads to secondary complications following SE. In the present study, we investigated whether transient receptor potential canonical channel-3 (TRPC3) may link vascular endothelial growth factor (VEGF) pathway to NFkappaB/ETB receptor axis in the rat piriform cortex during vasogenic edema formation. Following SE, TRPC3 and ETB receptor independently activated phosphatidylinositol 3 kinase (PI3K)/AKT/eNOS signaling pathway. SN50 (a NFkappaB inhibitor) attenuated the up-regulations of eNOS, TRPC3 and ETB receptor expressions following SE, accompanied by reductions in PI3K/AKT phosphorylations. Inhibition of SE-induced VEGF over-expression by leptomycin B also abrogated PI3K and AKT phosphorylations, but not TRPC3 expression. Wortmannin (a PI3K inhibitor) and 3CAI (an AKT inhibitor) effectively inhibited up-regulation of eNOS expressions and vasogenic edema lesion following SE. These findings indicate that PI3K/AKT may be common down-stream molecules for TRPC3- and ETB receptor signaling pathways during vasogenic edema formation. In addition, the present data demonstrate for the first time that TRPC3 may integrate VEGF- and NFkappaB-mediated vasogenic edema formation following SE. Thus, we suggest that PI3K/AKT signaling pathway may be one of considerable therapeutic targets for vasogenic edema.
(3-Chloroacetyl)-indole, a novel allosteric AKT inhibitor, suppresses colon cancer growth in vitro and in vivo.[Pubmed:21885813]
Cancer Prev Res (Phila). 2011 Nov;4(11):1842-51.
Indole-3-carbinol (I3C) is produced in Brassica vegetables such as broccoli and cabbage and has been shown to inhibit proliferation and induce apoptosis in various cancer cells, including breast, prostate, colon, and leukemia. However, only high doses of I3C were shown to inhibit cell proliferation (IC(50) = 200-300 mumol/L). Our goal here was to develop a more potent antitumor agent by modifying the structure of I3C. We created I3C derivatives and found that (3-chloroacetyl)-indole (3CAI) more strongly inhibited colon cancer cell growth than I3C. In addition, by screening 85 kinases in a competitive kinase assay, we found that 3CAI was a specific AKT inhibitor. AKT is a serine/threonine kinase that plays a pivotal role in promoting transformation and chemoresistance by inducing proliferation and inhibiting apoptosis. Therefore, AKT is regarded as a critical target for cancer therapy. 3ICA, a derivative of I3C, is a potent and specific AKT inhibitor. This compound showed significant inhibition of AKT in an in vitro kinase assay and suppressed expression of AKT direct downstream targets such as mTOR and GSK3beta as well as induced growth inhibition and apoptosis in colon cancer cells. In addition, oral administration of this potent AKT inhibitor suppressed cancer cell growth in an in vivo xenograft mouse model.


