ARRY-380Tyrosine kinase HER2 and p95-HER2 inhibitor CAS# 937265-83-3 |
- CO-1686 (AVL-301)
Catalog No.:BCC1490
CAS No.:1374640-70-6
- Lapatinib Ditosylate
Catalog No.:BCC2083
CAS No.:388082-78-8
- BMS-690514
Catalog No.:BCC1430
CAS No.:859853-30-8
- TAK-285
Catalog No.:BCC3860
CAS No.:871026-44-7
- BMS-599626 Hydrochloride
Catalog No.:BCC1426
CAS No.:873837-23-1
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 937265-83-3 | SDF | Download SDF |
| PubChem ID | 42598643 | Appearance | Powder |
| Formula | C29H27N7O4S | M.Wt | 569.63 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | >28.5mg/mL in DMSO | ||
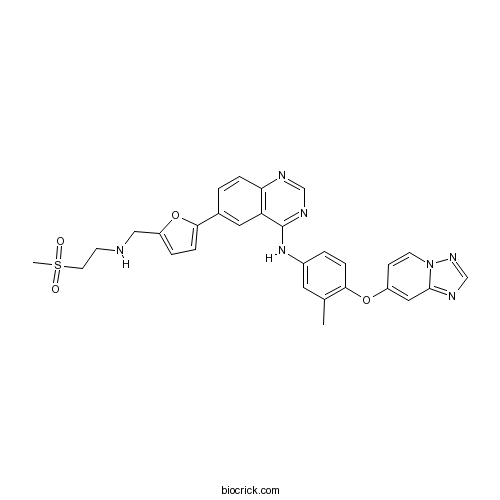
| Chemical Name | 6-[5-[(2-methylsulfonylethylamino)methyl]furan-2-yl]-N-[3-methyl-4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)phenyl]quinazolin-4-amine | ||
| SMILES | CC1=C(C=CC(=C1)NC2=NC=NC3=C2C=C(C=C3)C4=CC=C(O4)CNCCS(=O)(=O)C)OC5=CC6=NC=NN6C=C5 | ||
| Standard InChIKey | QVMNYGOVNWWFKF-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C29H27N7O4S/c1-19-13-21(4-7-26(19)39-22-9-11-36-28(15-22)32-18-34-36)35-29-24-14-20(3-6-25(24)31-17-33-29)27-8-5-23(40-27)16-30-10-12-41(2,37)38/h3-9,11,13-15,17-18,30H,10,12,16H2,1-2H3,(H,31,33,35) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | ARRY-380 is an orally bioavailable, potent, selective, small-molecule inhibitor of tyrosine kinase HER2 and p95-HER2 with an IC50 of 8 and 7 nM, respectively. | |||||
| Targets | p95-HER2 | HER2 | EGFR | |||
| IC50 | 7 nM | 8 nM | 4 μM | |||

ARRY-380 Dilution Calculator

ARRY-380 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.7555 mL | 8.7776 mL | 17.5553 mL | 35.1105 mL | 43.8881 mL |
| 5 mM | 0.3511 mL | 1.7555 mL | 3.5111 mL | 7.0221 mL | 8.7776 mL |
| 10 mM | 0.1756 mL | 0.8778 mL | 1.7555 mL | 3.5111 mL | 4.3888 mL |
| 50 mM | 0.0351 mL | 0.1756 mL | 0.3511 mL | 0.7022 mL | 0.8778 mL |
| 100 mM | 0.0176 mL | 0.0878 mL | 0.1756 mL | 0.3511 mL | 0.4389 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
IC50: 8 nM (HER2)
HER2 is a member of the human epidermal growth factor receptor (HER/EGFR/ERBB) family. Amplification or overexpression of this oncogene has been shown to play an important role in the development and progression of certain aggressive types of breast cancer. ARRY-380 is an orally bioavailable inhibitor of the human epidermal growth factor receptor tyrosine kinase ErbB-2 (also called HER2) with potential antineoplastic activity.
In vitro: ARRY-380 is reported to be a reversible, ATP-competitive inhibitor with nanomolar activity against HER2 enzyme. In cell-based assays, ARRY-380 is ~500-fold selective for HER2 vs. EGFR and is equipotent against truncated p95-HER2 [1].
In vivo: ARRY-380 treatment significantly enhances survival in two ErbB2 driven intracranial tumor xenograft models, with superior activity compared to other ErbB2 agents in these studies. Additionally, ARRY-380 has demonstrated durable clinical activity in heavily pre-treated patients with ErbB2+ MBC. These preclinical and clinical data suggest that ARRY-380 may provide benefit to patients with ErbB2+ MBC with brain metastases. These preclinical and clinical data suggest that ARRY-380 may provide benefit to patients with ErbB2+ MBC with brain metastases and warrants further study [2].
Clinical trial: In a phase 1 clinical trial, 15 patients have been treated in 5 dosing cohorts at doses of 25 to 300 mg BID. No DLTs have been observed and drug-related adverse events have included Grade 1 nausea, rash and fatigue and Grade 2 fatigue in 2 patients at the 200 mg BID dose level. Preliminary PK analyses indicate a trend for increasing Cmax and AUC with increasing dose, a median Tmax of 2 hours and a mean t1/2 of 4.6 hours across all cohorts. Two patients with HER2+ breast cancer have had stable disease for ≥ 4 months with no significant toxicity. One of these two patients had a notable reduction in liver metastases (28%) after 2 cycles of ARRY-380 and is currently on study.These findings indicate ARRY-380 has demonstrated an acceptable safety and PK profile and preliminary signs of clinical benefit. Dose escalation continues to determine the MTD [3].
References:
[1] S. L. Moulder, V. Borges, S. K. L. Chia, T. Baetz, E. Barrett, J. Garrus, K. Guthrie, C. Kass, E. Laird, J. Lyssikatos, F. Marmsater, E. Wallace. ARRY-380, a Selective HER2 Inhibitor: From Drug Design to Clinical Evaluation. Poster of AACR-NCI-EORTC, Nov 12-16, 2011, San Francisco, CA.
[2] Victoria Dinkel, Deborah Anderson, Shannon Winski, Jim Winkler, Kevin Koch and Patrice Lee. ARRY-380, a potent, small molecule inhibitor of ErbB2, increases survival in intracranial ErbB2+ xenograft models in mice. Poster available at www.arraybiopharma.com
[3] S. Chia, T. Baetz, S. D'Aloisio, G. Fernetich, B. Freeman, E. Barrett, C. Kass, J. Kang, B. Sajan, S. Moulder, and J. Garrus. A Phase 1 Study To Assess the Safety, Tolerability and Pharmacokinetics of ARRY-380 – An Oral Inhibitor of HER2. Cancer Res 2009;69(24 Suppl):Abstract nr 5111.
- NSC 95397
Catalog No.:BCC7109
CAS No.:93718-83-3
- GSK690693
Catalog No.:BCC2483
CAS No.:937174-76-0
- Leuconolam
Catalog No.:BCN4482
CAS No.:93710-27-1
- TC-H 106
Catalog No.:BCC2426
CAS No.:937039-45-7
- Magnolignan C
Catalog No.:BCN4085
CAS No.:93697-42-8
- OSI-027
Catalog No.:BCC4603
CAS No.:936890-98-1
- Forsythoside E
Catalog No.:BCN2782
CAS No.:93675-88-8
- Rengyol
Catalog No.:BCN4481
CAS No.:93675-85-5
- Magnolignan A
Catalog No.:BCN4084
CAS No.:93673-81-5
- VX-809
Catalog No.:BCC3712
CAS No.:936727-05-8
- LCZ696
Catalog No.:BCC5505
CAS No.:936623-90-4
- PCI-32765 (Ibrutinib)
Catalog No.:BCC1266
CAS No.:936563-96-1
- SB1317
Catalog No.:BCC1925
CAS No.:937270-47-8
- Pacritinib (SB1518)
Catalog No.:BCC4558
CAS No.:937272-79-2
- Magnaldehyde D
Catalog No.:BCN4070
CAS No.:93753-33-4
- GRP (human)
Catalog No.:BCC5810
CAS No.:93755-85-2
- Jangomolide
Catalog No.:BCN4483
CAS No.:93767-25-0
- Neogambogic acid
Catalog No.:BCN2321
CAS No.:93772-31-7
- Neurodazine
Catalog No.:BCC7738
CAS No.:937807-66-4
- gamma-Secretase Modulators
Catalog No.:BCC1586
CAS No.:937812-80-1
- Roxatidine Acetate HCl
Catalog No.:BCC4534
CAS No.:93793-83-0
- 3-Prenyl-2,4,6-trihydroxybenzophenone
Catalog No.:BCN1303
CAS No.:93796-20-4
- 22-beta-Acetoxyglycyrrhizin
Catalog No.:BCN7904
CAS No.:938042-17-2
- ATPγS tetralithium salt
Catalog No.:BCC7855
CAS No.:93839-89-5
Phase I Study of ONT-380, a HER2 Inhibitor, in Patients with HER2(+)-Advanced Solid Tumors, with an Expansion Cohort in HER2(+) Metastatic Breast Cancer (MBC).[Pubmed:28053022]
Clin Cancer Res. 2017 Jul 15;23(14):3529-3536.
Purpose: ONT-380 (ARRY-380) is a potent and selective oral HER2 inhibitor. This Phase I study determined the MTD, pharmacokinetics (PK) and antitumor activity of ONT-380 in HER2-positive advanced solid tumors, with an expansion cohort of patients with HER2(+) MBC.Experimental Design: ONT-380 was administered twice daily (BID) in continuous 28-day cycles. After a modified 3+3 dose-escalation design determined the MTD, the expansion cohort was enrolled. PK properties of ONT-380 and a metabolite were determined. Response was evaluated by Response Evaluation Criteria in Solid Tumors (RECIST).Results: Fifty patients received ONT-380 (escalation = 33; expansion = 17); 43 patients had HER2(+) MBC. Median prior anticancer regimens = 5. Dose-limiting toxicities of increased transaminases occurred at 800 mg BID, thus 600 mg BID was the MTD. Common AEs were usually Grade 1/2 in severity and included nausea (56%), diarrhea (52%), fatigue (50%), vomiting (40%) constipation, pain in extremity and cough (20% each). 5 patients (19%) treated at MTD had grade 3 AEs (increased transaminases, rash, night sweats, anemia, and hypokalemia). The half-life of ONT-380 was 5.38 hours and increases in exposure were approximately dose proportional. In evaluable HER2(+) MBC (n = 22) treated at doses >/= MTD, the response rate was 14% [all partial response (PR)] and the clinical benefit rate (PR + stable disease >/= 24 weeks) was 27%.Conclusions: ONT-380 had a lower incidence and severity of diarrhea and rash than that typically associated with current dual HER2/EGFR inhibitors and showed notable antitumor activity in heavily pretreated HER2(+) MBC patients, supporting its continued development. Clin Cancer Res; 23(14); 3529-36. (c)2017 AACR.


