Aristolactam FICAS# 112501-42-5 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 112501-42-5 | SDF | Download SDF |
| PubChem ID | 3081016 | Appearance | Yellow powder |
| Formula | C16H11NO3 | M.Wt | 265.3 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
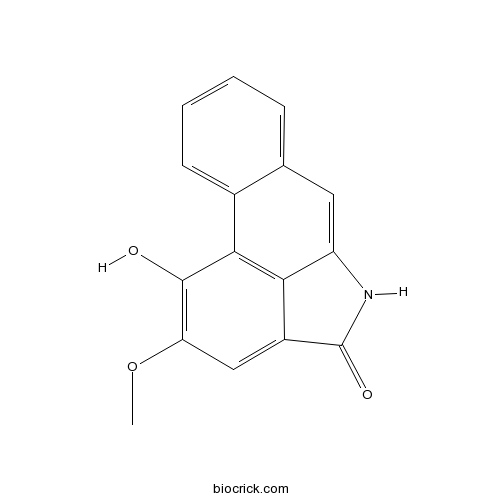
| SMILES | COC1=C(C2=C3C(=C1)C(=O)NC3=CC4=CC=CC=C42)O | ||
| Standard InChIKey | KBGNBPGXVKPRQI-UHFFFAOYSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Aristolactam FI shows platelet aggregation inhibitory activity. 2. Aristolactam FI are potential cancer chemotherapeutic and chemopreventive agents. |

Aristolactam FI Dilution Calculator

Aristolactam FI Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.7693 mL | 18.8466 mL | 37.6932 mL | 75.3864 mL | 94.2329 mL |
| 5 mM | 0.7539 mL | 3.7693 mL | 7.5386 mL | 15.0773 mL | 18.8466 mL |
| 10 mM | 0.3769 mL | 1.8847 mL | 3.7693 mL | 7.5386 mL | 9.4233 mL |
| 50 mM | 0.0754 mL | 0.3769 mL | 0.7539 mL | 1.5077 mL | 1.8847 mL |
| 100 mM | 0.0377 mL | 0.1885 mL | 0.3769 mL | 0.7539 mL | 0.9423 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 6-Aldehydo-isoophiopogonone A
Catalog No.:BCN6629
CAS No.:112500-90-0
- 5-Aminoisoquinoline
Catalog No.:BCC8736
CAS No.:1125-60-6
- 4,4-Pentamethylenepiperidine hydrochloride
Catalog No.:BCC6059
CAS No.:1125-01-5
- H-Glu(OcHex)-OH
Catalog No.:BCC2929
CAS No.:112471-82-6
- U-54494A hydrochloride
Catalog No.:BCC6668
CAS No.:112465-94-8
- 3-Phenyl-1-(pyrrol-1-yl)propan-1-one
Catalog No.:BCN4005
CAS No.:112448-69-8
- AZ3146
Catalog No.:BCC3731
CAS No.:1124329-14-1
- Ganoderic acid Jb
Catalog No.:BCN7972
CAS No.:112430-68-9
- Ganoderic acid T-Q
Catalog No.:BCN3209
CAS No.:112430-66-7
- Ganoderic acid TN
Catalog No.:BCN2443
CAS No.:112430-64-5
- 2',5,7-Trihydroxy-8-methoxyflavanone
Catalog No.:BCN6004
CAS No.:112408-71-6
- 3'-Deoxy-4-O-methylsappanol
Catalog No.:BCN3675
CAS No.:112408-68-1
- Picrasidine S
Catalog No.:BCN6006
CAS No.:112503-87-4
- Absouline
Catalog No.:BCN1954
CAS No.:112513-33-4
- Isoabsouline
Catalog No.:BCN1955
CAS No.:112513-34-5
- Neolinine
Catalog No.:BCN6564
CAS No.:112515-37-4
- Tubuloside A
Catalog No.:BCN2806
CAS No.:112516-05-9
- Citrusinol
Catalog No.:BCN8083
CAS No.:112516-43-5
- CI994 (Tacedinaline)
Catalog No.:BCC2159
CAS No.:112522-64-2
- ent-16alpha,17-Dihydroxyatisan-3-one
Catalog No.:BCN6607
CAS No.:112523-91-8
- Pioglitazone HCl
Catalog No.:BCC2278
CAS No.:112529-15-4
- 4-O-Methylepisappanol
Catalog No.:BCN3674
CAS No.:112529-37-0
- Mps1-IN-1
Catalog No.:BCC5590
CAS No.:1125593-20-5
- A 804598
Catalog No.:BCC6198
CAS No.:1125758-85-1
Total synthesis of aristolactams via a one-pot suzuki-miyaura coupling/aldol condensation cascade reaction.[Pubmed:18642834]
Org Lett. 2008 Aug 21;10(16):3543-6.
A direct one-pot synthesis of phenanthrene lactams, which employs a Suzuki-Miyaura coupling/aldol condensation cascade reaction of isoindolin-1-one with 2-formylphenylboronic acid, has been developed. The approach is used to efficiently produce a number of natural aristolactams, such as aristolactam BII (cepharanone B), aristolactam BIII, Aristolactam FI (piperolactam A), N-methyl piperolactam A, and sauristolactam.
Synthesis of aristolactam analogues and evaluation of their antitumor activity.[Pubmed:19394218]
Bioorg Med Chem Lett. 2009 Jun 1;19(11):3036-40.
A series of natural aristolactams and their analogues have been prepared and evaluated for antitumor activity against human cancer cells, including multi-drug resistant cell lines. Naturally occurring aristolactams, such as aristolactam BII (cepharanone B), aristolactam BIII, Aristolactam FI (piperolactam A), N-methyl piperolactam A, and sauristolactam showed moderate antitumor activities in selected cell lines. However, several synthetic aristolactam derivatives exhibited potent antitumor activities against a broad array of cancer cell lines with GI(50) values in the submicromolar range.


