CilostamidePDE3 inhibitor CAS# 68550-75-4 |
- Olprinone Hydrochloride
Catalog No.:BCC1821
CAS No.:119615-63-3
- GSK256066 2,2,2-trifluoroacetic acid
Catalog No.:BCC1605
CAS No.:1415560-64-3
- CDP 840 hydrochloride
Catalog No.:BCC7814
CAS No.:162542-90-7
- Nortadalafil
Catalog No.:BCC1806
CAS No.:171596-36-4
- Rolipram
Catalog No.:BCC2282
CAS No.:61413-54-5
- Oglemilast
Catalog No.:BCC1817
CAS No.:778576-62-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 68550-75-4 | SDF | Download SDF |
| PubChem ID | 2753 | Appearance | Powder |
| Formula | C20H26N2O3 | M.Wt | 342.44 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | OCP 3689 | ||
| Solubility | DMSO : 31 mg/mL (90.53 mM; Need ultrasonic and warming) | ||
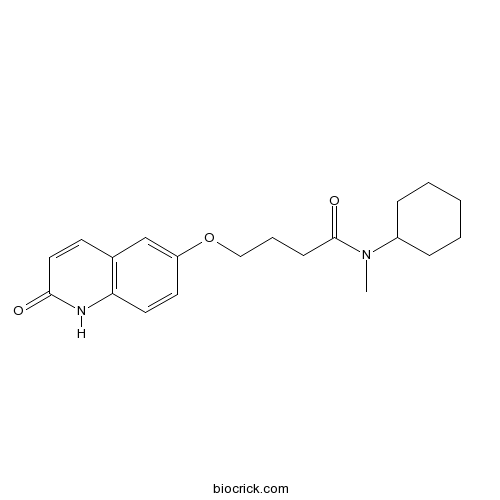
| Chemical Name | N-cyclohexyl-N-methyl-4-[(2-oxo-1H-quinolin-6-yl)oxy]butanamide | ||
| SMILES | CN(C1CCCCC1)C(=O)CCCOC2=CC3=C(C=C2)NC(=O)C=C3 | ||
| Standard InChIKey | UIAYVIIHMORPSJ-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H26N2O3/c1-22(16-6-3-2-4-7-16)20(24)8-5-13-25-17-10-11-18-15(14-17)9-12-19(23)21-18/h9-12,14,16H,2-8,13H2,1H3,(H,21,23) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Selective inhibitor of type III phosphodiesterase (PDE3). Displays moderate selectivity for PDE3A isozyme vs. PDE3B (IC50 values are 0.027 and 0.050 μM for PDE3A and PDE3B respectively). Inhibits ADP-induced platelet aggregation (IC50 = 16.8 μM); antithrombotic. Also available as part of the Phosphodiesterase Inhibitor. |

Cilostamide Dilution Calculator

Cilostamide Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.9202 mL | 14.6011 mL | 29.2022 mL | 58.4044 mL | 73.0055 mL |
| 5 mM | 0.584 mL | 2.9202 mL | 5.8404 mL | 11.6809 mL | 14.6011 mL |
| 10 mM | 0.292 mL | 1.4601 mL | 2.9202 mL | 5.8404 mL | 7.3005 mL |
| 50 mM | 0.0584 mL | 0.292 mL | 0.584 mL | 1.1681 mL | 1.4601 mL |
| 100 mM | 0.0292 mL | 0.146 mL | 0.292 mL | 0.584 mL | 0.7301 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Isoguvacine hydrochloride
Catalog No.:BCC6575
CAS No.:68547-97-7
- Angiotensin II
Catalog No.:BCC1030
CAS No.:68521-88-0
- Vigabatrin
Catalog No.:BCC2039
CAS No.:68506-86-5
- Pramiracetam
Catalog No.:BCC4928
CAS No.:68497-62-1
- WS 12
Catalog No.:BCC7543
CAS No.:68489-09-8
- Coromandaline
Catalog No.:BCN2044
CAS No.:68473-86-9
- Heliovicine
Catalog No.:BCN2047
CAS No.:68473-85-8
- 4-(p-Biphenylyl)-3-hydroxybutyric acid
Catalog No.:BCN2240
CAS No.:6845-17-6
- Isowighteone
Catalog No.:BCN4243
CAS No.:68436-47-5
- PPDA
Catalog No.:BCC5918
CAS No.:684283-16-7
- Timosaponin AI
Catalog No.:BCN7819
CAS No.:68422-00-4
- 6',7'-Dihydroxybergamottin acetonide
Catalog No.:BCN4242
CAS No.:684217-08-1
- GSK 264220A
Catalog No.:BCC6062
CAS No.:685506-42-7
- Eupatoriopicrin
Catalog No.:BCN7116
CAS No.:6856-01-5
- Pridinol Methanesulfonate
Catalog No.:BCC3845
CAS No.:6856-31-1
- Moschamine
Catalog No.:BCN3900
CAS No.:68573-23-9
- Prometaphanine
Catalog No.:BCN4244
CAS No.:6858-85-1
- PX-478 2HCl
Catalog No.:BCC6502
CAS No.:685898-44-6
- Isorhyncophylline
Catalog No.:BCN3466
CAS No.:6859-01-4
- Isorhynchophylline
Catalog No.:BCN6458
CAS No.:6859-1-4
- Xylobiose
Catalog No.:BCN8424
CAS No.:6860-47-5
- Procerine
Catalog No.:BCN2017
CAS No.:68622-81-1
- Otenabant
Catalog No.:BCC1828
CAS No.:686344-29-6
- CP-945598 HCl
Catalog No.:BCC1082
CAS No.:686347-12-6
Cilostamide and forskolin treatment during pre-IVM improves preimplantation development of cloned embryos by influencing meiotic progression and gap junction communication in pigs.[Pubmed:27056415]
Theriogenology. 2016 Aug;86(3):757-65.
This study was conducted to evaluate the effects of treatment with the cAMP modulators Cilostamide and/or forskolin during pre-IVM culture on meiotic progression, gap junction communication, intraoocyte cAMP level and glutathione content, embryonic development after parthenogenesis, and somatic cell nuclear transfer in pigs. Cumulus-oocyte complexes were cultured for 24 hours in unsupplemented medium or media containing 20 muM Cilostamide and/or 50 muM forskolin. After pre-IVM, oocytes were cultured for 41 to 44 hours in a standard IVM medium to induce oocyte maturation. When the nuclear status of oocytes was examined after pre-IVM for 24 hours, a higher (P < 0.01) proportion of oocytes treated with forskolin (85.5%) and Cilostamide + forskolin (92.6%) remained at the germinal vesicle stage compared with untreated (20.6%) and Cilostamide-treated oocytes (54.7%). cAMP level in pre-IVM oocytes was significantly increased by combined treatment with Cilostamide + forskolin (21.38 fmol/oocyte) relative to the no pre-IVM control, no treatment, Cilostamide, and forskolin groups (2.85, 1.88, 1.74, and 8.95 fmol/oocyte, respectively). Forskolin with or without Cilostamide significantly maintained open-gap junction communication relative to no treatment. Blastocyst formation in parthenogenesis was significantly (P < 0.01) improved by forskolin (65.3%) relative to other treatments (28.3% to 48.1%). Supplementation of pre-IVM with dibutyryl cAMP showed similar blastocyst formation as forskolin treatment (61.1% and 61.0%, respectively). In somatic cell nuclear transfer, simultaneous treatment with Cilostamide + forskolin significantly (P < 0.05) increased embryonic development to the blastocyst stage (42.9%) relative to the no pre-IVM, control, and Cilostamide groups (32.3, 28.6, and 32.8%, respectively). The glutathione contents in pre-IVM oocytes were increased by no treatment, forskolin, and Cilostamide + forskolin (1.38, 1.39, and 1.27 pixels/oocyte, respectively) compared with no pre-IVM and Cilostamide (1.00 and 0.99 pixels/oocyte, respectively; P < 0.05). Our results reported that the meiotic progression of immature pig oocytes could be reversibly attenuated by cAMP, whereas treatment with Cilostamide and forskolin during pre-IVM had positive effects on developmental competence of oocytes in pigs, probably by improving cytoplasmic maturation.
Supplement of cilostamide in growth medium improves oocyte maturation and developmental competence of embryos derived from small antral follicles in pigs.[Pubmed:28215673]
Theriogenology. 2017 Mar 15;91:1-8.
This study was conducted to evaluate the effects of cyclic AMP (cAMP) modulator Cilostamide (CIL) and forskolin (FSK) treatment during in vitro growth (IVG) on growth, maturation, and embryonic development of cumulus-oocyte complexes (COCs) derived from small antral follicles < 3 mm in diameter (SAFCOCs). SAFCOCs were untreated (control) or treated with 20 muM CIL and/or 50 muM FSK for 2 days for IVG. Next, IVG oocytes were cultured for maturation and then induced for parthenogenesis (PA) or used as recipient ooplasts for somatic cell nuclear transfer (SCNT). Nuclear maturation of oocytes was significantly lower in the control (49.6 +/- 9.3%) group than in other groups (67.2 +/- 5.0-79.8 +/- 7.9%). The cumulus expansion score after IVG-IVM was significantly higher in the control and CIL group than in the FSK and CIL + FSK groups. CIL significantly increased mean diameter SAF-derived oocytes (120.0 +/- 0.5 mum) compared to the control, FSK, and CIL + FSK (114.8 +/- 0.5-116.7 +/- 0.6 mum) and showed a comparable level of intracellular glutathione (GSH) contents (0.84 +/- 0.07 pixels/oocyte) to medium antral follicle (MAF)-derived oocytes (1.00 +/- 0.08 pixels/oocyte), but was higher than those of oocytes treated with FSK and CIL + FSK (0.29 +/- 0.05 and 0.37 +/- 0.05 pixels/oocyte, respectively). CIL treatment significantly increased blastocyst formation (55.1 +/- 4.7%) after PA relative to the control (29.4 +/- 6.4%), FSK (34.8 +/- 7.1%), and CIL + FSK (41.1 +/- 5.2%). A higher proportion of oocytes treated with CIL, FSK, and CIL + FSK (73.3 +/- 1.7-82.8 +/- 9.1%) remained at the germinal vesicle stage after IVG culture than control oocytes (40.0 +/- 5.0%). Following SCNT, blastocyst formation of SAFCOCs treated with CIL (22.4 +/- 6.3%) was higher than that of oocytes (0-10.4 +/- 5.3%) in control, FSK, and CIL + FSK, but similar to that (25.3 +/- 3.5%) of MAF-derived COCs not cultured for IVG. The cAMP level of SAFCOCs before IVG was 0.1 +/- 0.03 fmol/oocyte. After 2 days of IVG culture, cAMP level was increased significantly by treatment with FSK and CIL + FSK (3.0 +/- 0.57 and 12.1 +/- 0.62 fmol/oocyte, respectively) relative to the control and CIL treatment (0.1 +/- 0.03 and 0.3 +/- 0.04 fmol/oocyte, respectively). Our results demonstrate that CIL treatment during IVG improves the low developmental competence of SAFCOCs to levels comparable to MAFCOCs by allowing oocyte growth while inhibiting premature meiotic maturation, probably via maintenance of cAMP concentrations at appropriate levels.
Effects of cilostamide and/or forskolin on the meiotic resumption and development competence of growing ovine oocytes selected by brilliant cresyl blue staining.[Pubmed:26879998]
Theriogenology. 2016 May;85(8):1483-90.
The relevance of low developmental competence of in vitro-matured oocyte to the incomplete/delayed cytoplasmic maturation, and the heterogeneity of retrieved oocytes is well established in several species. A short phase of prematuration culture was used to allow better oocyte cytoplasmic maturation. The preselection of growing and fully grown oocytes has been proposed to improve developmental competency. This study investigated the effects of phosphodiesterase type 3-specific inhibitor, Cilostamide, and adenylate cyclase activator, forskolin, on the resumption of meiosis and developmental competence of growing ovine oocytes selected by brilliant cresyl blue (BCB) staining. Results indicate that Cilostamide, forskolin, and their combination significantly (P < 0.05) increased the percentage of growing (BCB-) oocytes maintained at the germinal vesicle stage. However, only forskolin significantly (P < 0.05) increased the yield and quality of blastocysts derived from BCB- oocytes compared with non-BCB-treated oocytes. We conclude that a short prematuration culture with forskolin may improve the in vitro developmental competency of growing oocytes in ovine.
Effect of the meiotic inhibitor cilostamide on resumption of meiosis and cytoskeletal distribution in buffalo oocytes.[Pubmed:27616355]
Anim Reprod Sci. 2016 Nov;174:37-44.
Improving the quality of in vitro maturated buffalo oocytes is essential for embryo production. We report here the effects on microtubules and microfilaments in oocytes and embryo development that result from treating buffalo oocytes with the phosphodiesterase 3 (PDE3) inhibitor Cilostamide. Addition of 20muM or 50muM Cilostamide for 24h during in vitro maturation showed no differences in the percentage of oocytes arrested at the germinal vesicle (GV) stage. When 20muM Cilostamide was added to the pre-maturation culture for 6h, 12h or 24h and continued for another 24h without Cilostamide, oocytes resumed meiosis, but with significantly lower (P<0.01) maturation rates in the 24h group than that in the other two groups. During oocyte maturation in vitro, no microtubules were detected before GV breakdown (GVBD). After GVBD, microtubules combined with condensed chromatin to form the meiotic metaphase spindle. Microfilaments covered a thick area around the cellular cortex and overlying chromosomes. Cilostamide had no effects on microtubules and microfilaments in metaphase II oocytes, and there were no significant differences in the rates of cleavage, blastocyst formation and number of blastocyst cells between oocytes treated pre-maturation with inhibitor for 6h and those of the control group (P>0.05). In summary, Cilostamide reversibly arrested the resumption of meiosis without any adverse impact on the dynamic changes in microtubules and microfilaments in buffalo oocytes and their in vitro developmental capacity.
Potent effects of novel anti-platelet aggregatory cilostamide analogues on recombinant cyclic nucleotide phosphodiesterase isozyme activity.[Pubmed:10644042]
Biochem Pharmacol. 2000 Feb 15;59(4):347-56.
The inhibitory potential of novel anti-platelet aggregatory Cilostamide analogues on phosphodiesterase (PDE) isozyme activities was investigated with recombinant PDE isozymes expressed in a baculovirus/ Sf9 expression system. The recombinant enzymes (PDE1-PDE5 and PDE7) showed Km values and sensitivities to selective inhibitors similar to those reported previously for native enzymes purified from tissues. The cyclooctylurea derivative OPC-33540 (6-[3-[3-cyclooctyl-3-[(1R*,2R*)-2-hydroxycyclohexyl]ureido]-propoxy]-2(1H)-quino linone) inhibited recombinant PDE3A (IC50 = 0.32 nM) more potently and selectively than the classical PDE3 inhibitors Cilostamide, cilostazol, milrinone, and amrinone. The cyclopropylurea derivative OPC-33509 [(-)-6-[3-[3-cyclopropyl-3-[(1R,2R)-2-hydroxycyclohexyl]ureido]-propoxy]-2(1H)-qu inolinone] was less potent (IC50 = 0.10 microM) than OPC-33540, demonstrating that the cyclooctyl moiety was important for a potent inhibitory effect. In platelets, OPC-33540 potentiated cyclic AMP accumulation concentration-dependently in both the absence and the presence of 3 nM prostaglandin E1 (PGE1) (doubling concentrations: 32.5 and 6.2 nM, respectively). OPC-33540 inhibited thrombin-induced platelet aggregation potently (Ic50 = 27.8 nM). The anti-platelet aggregation effect also was stimulated in the presence of 3 nM PGE1 (IC50 = 6.0 nM). There was a good correlation between the IC50 values of PDE3 inhibitors in this study for recombinant PDE3A activity and their IC50 values for thrombin-induced platelet aggregation (r = 0.998). These data demonstrated that OPC-33540 is a highly selective and potent PDE3 inhibitor and a useful probe for identification of the intracellular functions of PDE3.


