Ethyl cinnamateCAS# 103-36-6 |
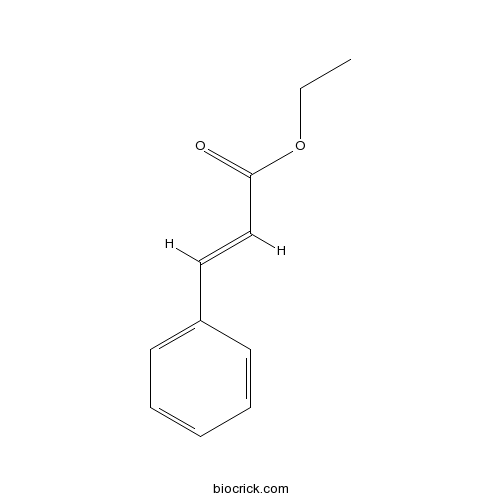
- (Z)-ethyl cinnamate
Catalog No.:BCN9200
CAS No.:4192-77-2
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 103-36-6 | SDF | Download SDF |
| PubChem ID | 637758 | Appearance | Oil |
| Formula | C11H12O2 | M.Wt | 176.21 |
| Type of Compound | Phenylpropanoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | ethyl (E)-3-phenylprop-2-enoate | ||
| SMILES | CCOC(=O)C=CC1=CC=CC=C1 | ||
| Standard InChIKey | KBEBGUQPQBELIU-CMDGGOBGSA-N | ||
| Standard InChI | InChI=1S/C11H12O2/c1-2-13-11(12)9-8-10-6-4-3-5-7-10/h3-9H,2H2,1H3/b9-8+ | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Ethyl cinnamate has antifungal, and vasorelaxant effects, it can inhibit the tonic contractions induced by high K+ and phenylephrine (PE) in a concentration-dependent manner, with respective IC50 values of 0.30 +/- 0.05 mM and 0.38 +/- 0.04 mM. Ethyl cinnamate can lead to the damage of cell membrane system and metabolic disorder through inducing lipid peroxidation via initiating ROS overproduction.Ethyl cinnamate has acute inhibition to the maximum quantum yield and the potential activity of photosystem II of Chlorella pyrenoidosa. |
| Targets | ROS | NO | Calcium Channel | Potassium Channel | Antifection |
| In vitro | Toxic Effects of Ethyl Cinnamate on the Photosynthesis and Physiological Characteristics of Chlorella vulgaris Based on Chlorophyll Fluorescence and Flow Cytometry Analysis.[Pubmed: 26101784]ScientificWorldJournal. 2015;2015:107823.The toxic effects of Ethyl cinnamate on the photosynthetic and physiological characteristics of Chlorella vulgaris were studied based on chlorophyll fluorescence and flow cytometry analysis.
Effects of allelochemicals ethyl cinnamate on the growth and physiological characteristics of Chlorella pyrenoidosa.[Pubmed: 23487932]Huan Jing Ke Xue. 2013 Jan;34(1):156-62.The effects of Ethyl cinnamate on the growth and physiological characteristics of Chlorella pyrenoidosa were studied.
The allelopathic mechanisms were explored, from views of chlorophyll a content, antioxidant enzyme activities, reactive oxygen species (ROS) level, malondialdehyde (MDA) content and photosynthetic activity.
Antifungal properties of Ocimum gratissimum essential oil (ethyl cinnamate chemotype).[Pubmed: 11449510]Fitoterapia. 2000 Sep;71(5):567-9.
Largely widespread in tropical countries, Ocimum gratissimum has been claimed for many uses in folk medicine. Recent research on its essential oils showed five chemotypes.
|
| In vivo | Fragrance material review on ethyl cinnamate.[Pubmed: 18037216]Food Chem Toxicol. 2007;45 Suppl 1:S90-4.A toxicologic and dermatologic review of Ethyl cinnamate when used as a fragrance ingredient is presented. |
| Animal Research | Vasorelaxant effects of ethyl cinnamate isolated from Kaempferia galanga on smooth muscles of the rat aorta.[Pubmed: 12143006]Planta Med. 2002 Jul;68(7):655-7.From the rhizomes of Kaempferia galanga, Ethyl cinnamate (EC) was isolated and its vasorelaxant effect was examined on the rat aorta.
|

Ethyl cinnamate Dilution Calculator

Ethyl cinnamate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 5.675 mL | 28.3752 mL | 56.7505 mL | 113.5009 mL | 141.8762 mL |
| 5 mM | 1.135 mL | 5.675 mL | 11.3501 mL | 22.7002 mL | 28.3752 mL |
| 10 mM | 0.5675 mL | 2.8375 mL | 5.675 mL | 11.3501 mL | 14.1876 mL |
| 50 mM | 0.1135 mL | 0.5675 mL | 1.135 mL | 2.27 mL | 2.8375 mL |
| 100 mM | 0.0568 mL | 0.2838 mL | 0.5675 mL | 1.135 mL | 1.4188 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Methyl cinnamate
Catalog No.:BCN5043
CAS No.:103-26-4
- Monobenzone
Catalog No.:BCC3818
CAS No.:103-16-2
- H-DL-Phg-OH
Catalog No.:BCC3317
CAS No.:103-01-5
- Trelagliptin succinate
Catalog No.:BCC2015
CAS No.:1029877-94-8
- INCB28060
Catalog No.:BCC3793
CAS No.:1029712-80-8
- Scutellaric acid
Catalog No.:BCN5843
CAS No.:102919-76-6
- MDL 73005EF hydrochloride
Catalog No.:BCC6636
CAS No.:102908-60-1
- Pexidartinib (PLX3397)
Catalog No.:BCC6405
CAS No.:1029044-16-3
- 17-Hydroxy sprengerinin C
Catalog No.:BCN2755
CAS No.:1029017-75-1
- Lycopsamine
Catalog No.:BCN1999
CAS No.:10285-07-1
- Intermedine
Catalog No.:BCN1997
CAS No.:10285-06-0
- MLN8237 (Alisertib)
Catalog No.:BCC2166
CAS No.:1028486-01-2
- Benzyl cinnamate
Catalog No.:BCN5042
CAS No.:103-41-3
- Scutebarbatine M
Catalog No.:BCN8327
CAS No.:960302-92-5
- N-Methylbenzylamine
Catalog No.:BCN1790
CAS No.:103-67-3
- Phenylacetic Acid
Catalog No.:BCC8349
CAS No.:103-82-2
- 4'-Methylacetanilide
Catalog No.:BCC8714
CAS No.:103-89-9
- Acetaminophen
Catalog No.:BCC5269
CAS No.:103-90-2
- Dehydroheliobuphthalmin
Catalog No.:BCN5844
CAS No.:103001-05-4
- Alterlactone
Catalog No.:BCN7261
CAS No.:1030376-89-6
- MK-4305
Catalog No.:BCC1760
CAS No.:1030377-33-3
- CTCE 9908
Catalog No.:BCC6366
CAS No.:1030384-98-5
- Ethyl 3-(pyridin-2-ylamino)propanoate
Catalog No.:BCC8973
CAS No.:103041-38-9
- Daptomycin
Catalog No.:BCC1057
CAS No.:103060-53-3
Fragrance material review on ethyl cinnamate.[Pubmed:18037216]
Food Chem Toxicol. 2007;45 Suppl 1:S90-4.
A toxicologic and dermatologic review of Ethyl cinnamate when used as a fragrance ingredient is presented.
Antifungal properties of Ocimum gratissimum essential oil (ethyl cinnamate chemotype).[Pubmed:11449510]
Fitoterapia. 2000 Sep;71(5):567-9.
Largely widespread in tropical countries, Ocimum gratissimum has been claimed for many uses in folk medicine. Recent research on its essential oils showed five chemotypes. An Indian chemotype, with a high level of Ethyl cinnamate, presents, in vitro, an interesting spectrum of antifungal properties.
Vasorelaxant effects of ethyl cinnamate isolated from Kaempferia galanga on smooth muscles of the rat aorta.[Pubmed:12143006]
Planta Med. 2002 Jul;68(7):655-7.
From the rhizomes of Kaempferia galanga, Ethyl cinnamate (EC) was isolated and its vasorelaxant effect was examined on the rat aorta. EC inhibited the tonic contractions induced by high K+ and phenylephrine (PE) in a concentration-dependent manner, with respective IC50 values of 0.30 +/- 0.05 mM and 0.38 +/- 0.04 mM. The relaxant effect against PE-induced contractions was greater in the presence of endothelium. Pre-treatment of the aorta with methylene blue and indomethacin significantly reduced the relaxant effect. These results suggest that the inhibitory effects of EC may involve inhibition of Ca2+ influx into vascular cells and release of nitric oxide (NO) and prostacyclin from the endothelial cells. Thus, the vasorelaxant effect of EC mediated through multiple pathways may explain the traditional use of the parent plant in treating hypertension.
Toxic Effects of Ethyl Cinnamate on the Photosynthesis and Physiological Characteristics of Chlorella vulgaris Based on Chlorophyll Fluorescence and Flow Cytometry Analysis.[Pubmed:26101784]
ScientificWorldJournal. 2015;2015:107823.
The toxic effects of Ethyl cinnamate on the photosynthetic and physiological characteristics of Chlorella vulgaris were studied based on chlorophyll fluorescence and flow cytometry analysis. Parameters, including biomass, F(v)/F(m) (maximal photochemical efficiency of PSII), capital EF, Cyrillic(PSII) (actual photochemical efficiency of PSII in the light), FDA, and PI staining fluorescence, were measured. The results showed the following: (1) The inhibition on biomass increased as the exposure concentration increased. 1 mg/L Ethyl cinnamate was sufficient to reduce the total biomass of C. vulgaris. The 48-h and 72-h EC50 values were 2.07 mg/L (1.94-2.20) and 1.89 mg/L (1.82-1.97). (2) After 24 h of exposure to 2-4 mg/L Ethyl cinnamate, the photosynthesis of C. vulgaris almost ceased, manifesting in capital EF, Cyrillic(PSII) being close to zero. After 72 h of exposure to 4 mg/L Ethyl cinnamate, the Fv /Fm of C. vulgaris dropped to zero. (3) Ethyl cinnamate also affected the cellular physiology of C. vulgaris, but these effects resulted in the inhibition of cell yield rather than cell death. Exposure to Ethyl cinnamate resulted in decreased esterase activities in C. vulgaris, increased average cell size, and altered intensities of chlorophyll a fluorescence. Overall, esterase activity was the most sensitive variable.


