Glycyrrhizic acid ammonium saltCAS# 53956-04-0 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 53956-04-0 | SDF | Download SDF |
| PubChem ID | 62074 | Appearance | White powder |
| Formula | C42H65NO16 | M.Wt | 839.96 |
| Type of Compound | Triterpenoids | Storage | Desiccate at -20°C |
| Synonyms | Ammonium Glycyrrhizinate;1407-03-0 | ||
| Solubility | Soluble in methanol and aqueous alkaline solution | ||
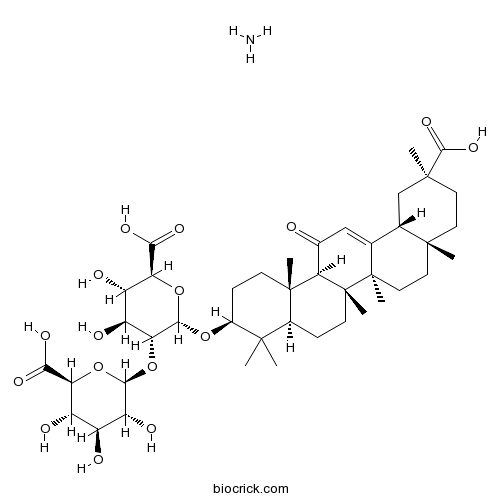
| Chemical Name | (2S,3S,4S,5R,6S)-6-[[(3S,4aR,6aR,6bS,8aS,11S,12aR,14aR,14bS)-11-carboxy-4,4,6a,6b,8a,11,14b-heptamethyl-14-oxo-2,3,4a,5,6,7,8,9,10,12,12a,14a-dodecahydro-1H-picen-3-yl]oxy]-5-[(2R,3R,4S,5S,6S)-6-carboxy-3,4,5-trihydroxyoxan-2-yl]oxy-3,4-dihydroxyoxane-2-c | ||
| SMILES | N.CC1(C)[C@H](CC[C@@]2(C)[C@H]1CC[C@]3(C)[C@@H]2C(=O)C=C4[C@@H]5C[C@](C)(CC[C@]5(C)CC[C@@]34C)C(O)=O)O[C@H]6O[C@@H]([C@@H](O)[C@H](O)[C@H]6O[C@@H]7O[C@@H]([C@@H](O)[C@H](O)[C@H]7O)C(O)=O)C(O)=O | ||
| Standard InChIKey | ILRKKHJEINIICQ-OOFFSTKBSA-N | ||
| Standard InChI | InChI=1S/C42H62O16.H3N/c1-37(2)21-8-11-42(7)31(20(43)16-18-19-17-39(4,36(53)54)13-12-38(19,3)14-15-41(18,42)6)40(21,5)10-9-22(37)55-35-30(26(47)25(46)29(57-35)33(51)52)58-34-27(48)23(44)24(45)28(56-34)32(49)50;/h16,19,21-31,34-35,44-48H,8-15,17H2,1-7H3,(H,49,50)(H,51,52)(H,53,54);1H3/t19-,21-,22-,23-,24-,25-,26-,27+,28-,29-,30+,31+,34-,35-,38+,39-,40-,41+,42+;/m0./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Glycyrrhizic acid ammonium salt has wound healing activity. |
| Targets | ROS |
| In vivo | Study of the influence of original multicomponent gels on the process of pathological scar formation using new methodological approach.[Pubmed: 25365869]Eksp Klin Farmakol. 2014;77(9):38-42.A new method of the quantitative macroscopic assessment of the process of a complex infected wound healing has been created. It was verified by example of the influence of original multicomponent gels consisting of cycloferon, amino acid glycine, glycyram (Glycyrrhizic acid ammonium salt), and vegetable oils on the process of infected wound healing and pathological scar formation. Simultaneously, the wound healing was monitored by the conventional histomorphological method. The proposed gels more effectively prevent the formation of pathological scars in comparison to reference preparation Contractubex. |
| Animal Research | Pharmacological Investigation on the Wound Healing Effects of Glycyrrhizic Acid Ammonium Salt in Rats.[Reference: WebLink]Ijrpbsonline Com., 2011.Wound healing process involves several steps, which involves coagulation, inflammation, formation of granulation tissue, matrix formation, remodeling of connective tissue, collagenisation and aquisation of wound strength.Wound healing activity of GA was Glycyrrhizic acid ammonium salt (GA) studied in terms of 1) % wound contraction, 2) epithelization, 3) wound breaking strength in incision and excision wound models. In the standard group the rats were treated with 0.0005% w/w fluticasone ointment for 10 days in incision model and for 16 days in excision model. In the test group, the rats were treated with 2% w/w GA ointment for 10 days in incision model and for 16 days in excision model.
|

Glycyrrhizic acid ammonium salt Dilution Calculator

Glycyrrhizic acid ammonium salt Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.1905 mL | 5.9527 mL | 11.9053 mL | 23.8107 mL | 29.7633 mL |
| 5 mM | 0.2381 mL | 1.1905 mL | 2.3811 mL | 4.7621 mL | 5.9527 mL |
| 10 mM | 0.1191 mL | 0.5953 mL | 1.1905 mL | 2.3811 mL | 2.9763 mL |
| 50 mM | 0.0238 mL | 0.1191 mL | 0.2381 mL | 0.4762 mL | 0.5953 mL |
| 100 mM | 0.0119 mL | 0.0595 mL | 0.1191 mL | 0.2381 mL | 0.2976 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Aristolactam BIII
Catalog No.:BCN5718
CAS No.:53948-10-0
- Aristolactam BII
Catalog No.:BCN5717
CAS No.:53948-09-7
- Aristolactam AII
Catalog No.:BCN3924
CAS No.:53948-07-5
- Corylin
Catalog No.:BCN5716
CAS No.:53947-92-5
- Apterin
Catalog No.:BCN3910
CAS No.:53947-89-0
- Euparone
Catalog No.:BCN7204
CAS No.:53947-86-7
- Crotalarine
Catalog No.:BCN2076
CAS No.:53937-97-6
- Deoxyarbutin
Catalog No.:BCC4774
CAS No.:53936-56-4
- Hederagenin 28-O-beta-D-glucopyranosyl ester
Catalog No.:BCN1423
CAS No.:53931-25-2
- (+)-Anabasine hydrochloride
Catalog No.:BCC7219
CAS No.:53912-89-3
- Pentostatin
Catalog No.:BCC1845
CAS No.:53910-25-1
- Nb-Feruloyltryptamine
Catalog No.:BCN3899
CAS No.:53905-13-8
- Ginsenoside F1
Catalog No.:BCN1244
CAS No.:53963-43-2
- Luteolin 7,3'-di-O-glucuronide
Catalog No.:BCN5396
CAS No.:53965-08-5
- Z-Phg-OH
Catalog No.:BCC2795
CAS No.:53990-33-3
- Cefaclor
Catalog No.:BCC2527
CAS No.:53994-73-3
- L-Nicotine
Catalog No.:BCN6269
CAS No.:54-11-5
- Tryptophan
Catalog No.:BCN2615
CAS No.:54-12-6
- 5-Hydroxyindole-3-Acetic Acid
Catalog No.:BCC8285
CAS No.:54-16-0
- Sodium salicylate
Catalog No.:BCC4846
CAS No.:54-21-7
- Furosemide
Catalog No.:BCC3782
CAS No.:54-31-9
- Metyrapone
Catalog No.:BCC7632
CAS No.:54-36-4
- Idoxuridine
Catalog No.:BCC4666
CAS No.:54-42-2
- Pilocarpine HCl
Catalog No.:BCC4702
CAS No.:54-71-7
[Simultaneous determination of principal components and related substances of raw material drug of ammonium glycyrrhizinate by reversed-phase high performance liquid chromatography].[Pubmed:24392624]
Se Pu. 2013 Sep;31(9):869-74.
An analytical method for the simultaneous determination of 18alpha-glycyrrhizic acid, 18beta-glycyrrhizinic acid, related substances A and B and drug quality standard by reversed-phase high performance liquid chromatography (RP-HPLC) was established. The assay was carried out on a Durashell-C18 column (250 mm x 4.6 mm, 5 microm) with 10 mmol/L ammonium perchlorate (the pH value was adjusted to 8.20 with ammonia)-methanol (48:52, v/v) as mobile phase at a flow rate of 0.80 mL/min, and the detection wavelength was set at 254 nm. The column temperature was 50 degrees C and the injection volume was 10 microL. Under the separation conditions, the calibration curves of the analytes showed good linearities within the mass concentrations of 0.50 -100 mg/L (r > 0.999 9). The detection limits for 18alpha-glycyrrhizic acid, 18beta-glycyrrhizinic acid, related substances A and B were 0.15, 0.10, 0.10, 0.15 mg/L, respectively. The average recoveries were between 97.32% and 99.33% (n = 3) with the relative standard deviations (RSDs) between 0.05% and 1.06%. The method is sensitive, reproducible, and the results are accurate and reliable. The method can be used for the determination of principal components and related substances of ammonium glycyrrhizinate for the quality control of raw material drug of ammonium glycyrrhizinate.
Cardioprotective effect of ammonium glycyrrhizinate against doxorubicin-induced cardiomyopathy in experimental animals.[Pubmed:25298583]
Indian J Pharmacol. 2014 Sep-Oct;46(5):527-30.
OBJECTIVE: The objective of this study was to evaluate the cardioprotective effect of herbal bioactive compound ammonium glycyrrhizinate against doxorubicin-induced cardiomyopathy, in experimental animals. MATERIALS AND METHODS: Ammonium glycyrrhizinate (50, 100, 200 mg/kg, p.o.) was administered for four weeks in albino rats. Cardiomyopathy was induced with a dose of 2.5 mg/kg i.p. of doxorubicin on 1(th), 7(th), 14(th), 21(th), 28(th) day in the experimental animals. At the end of the experiment, on 29(th) day, serum and heart tissues were collected and hemodynamic, biochemical and histopathological studies were carried out. RESULTS: Administration of doxorubicin in normal rats showed significant (P < 0.001) changes in body weight, feed intake, urine output, hemodynamic parameters like (blood pressure, heart rate, cardiac output) and in lipid profile (cholesterol, triglyceride, high density lipoprotein, low density lipoprotein, very low density lipoprotein) indicating cardiomyopathy symptoms. Animals treated with ammonium glycyrrhizinate significantly (P < 0.05) decreased triglyceride, cholesterol, low density lipoprotein (LDL) and very low density lipoprotein (VLDL) levels. Moreover, high density lipoprotein (HDL) levels increased in rats treated with ammonium glycyrrhizinate as compared with the normal group. CONCLUSION: Ammonium glycyrrhizinate is effective in controlling serum lipid profile and cardiac complications in experimentally induced cardiomyopathy in animals.
[Study of the influence of original multicomponent gels on the process of pathological scar formation using new methodological approach].[Pubmed:25365869]
Eksp Klin Farmakol. 2014;77(9):38-42.
A new method of the quantitative macroscopic assessment of the process of a complex infected wound healing has been created. It was verified by example of the influence of original multicomponent gels consisting of cycloferon, amino acid glycine, glycyram (ammonium salt of glycyrrhizic acid), and vegetable oils on the process of infected wound healing and pathological scar formation. Simultaneously, the wound healing was monitored by the conventional histomorphological method. The proposed gels more effectively prevent the formation of pathological scars in comparison to reference preparation Contractubex.
Study of an Acid-Free Technique for the Preparation of Glycyrrhetinic Acid from Ammonium Glycyrrhizinate in Subcritical Water.[Pubmed:26749800]
Nat Prod Commun. 2015 Nov;10(11):1801-3.
The aim of this work was to study an application of a previously developed expedient acid-free technique for the preparation of glycyrrhetinic acid from ammonium glycyrrhizinate that requires no use of acids and toxic organic solvents. Subcritical water that serves as a reactant and a solvent was used in order to obtain glycyrrhetinic acid in good yields starting from ammonium glycyrrhizinate. It has been shown that variation of only one parameter of the process (temperature) allows alteration to thecomposition of the hydrolysis products. A new method was used for the synthesis of glycyrrhetinic acid (glycyrrhizic acid aglycone) and its monoglycoside. HPLC combined with mass spectrometry and NMR spectroscopy were used to determine the quantitative and qualitative compositions of the obtained products. The method developed for the production of glycyrrhetinic acid in subcritical water is environmentally friendly and faster than conventional hydrolysis methods that use acids and-expensive and toxic organic solvents. The proposed technique has a potential for the future development of inexpensive and environmentally friendly technologies for production of new pharmaceutical plant-based substances.
Ammonium glycyrrhizinate-loaded niosomes as a potential nanotherapeutic system for anti-inflammatory activity in murine models.[Pubmed:24493924]
Int J Nanomedicine. 2014 Jan 24;9:635-51.
BACKGROUND: Liquorice extracts demonstrate therapeutic efficacy in treating dermatitis, eczema, and psoriasis when compared with corticosteroids. In this work, nonionic surfactant vesicles (niosomes, NSVs) containing polysorbate 20 (Tween 20), cholesterol, and cholesteryl hemisuccinate at different molar concentrations were used to prepare monoammonium glycyrrhizinate (AG)-loaded NSVs. The anti-inflammatory properties of AG-loaded NSVs were investigated in murine models. METHODS: The physicochemical properties of the NSVs were characterized using dynamic light scattering. The fluidity of the lipid bilayer was evaluated by measuring the fluorescence intensity of diphenylhexatriene. The drug entrapment efficiency of AG was assessed using high-performance liquid chromatography. The physicochemical stability of the NSVs was evaluated as a function of time using dynamic light scattering combined with Turbiscan Lab Expert analysis. Serum stability was determined by incubating the NSVs with 10% v/v fetal bovine serum. The cytotoxic effects of the NSVs were investigated in human dermal fibroblasts using the Trypan blue dye exclusion assay (for cell mortality) and an MTT assay (for cell viability). Release profiles for the AG-loaded NSVs were studied in vitro using cellulose membranes. NSVs showing the most desirable physicochemical properties were selected to test for in vivo anti-inflammatory activity in murine models. The anti-inflammatory activity of the NSVs was investigated by measuring edema and nociception in mice stimulated with chemical agents. RESULTS: NSVs showed favorable physicochemical properties for in vitro and in vivo administration. In addition, they demonstrated long-term stability based on Turbiscan Lab Expert analysis. The membrane fluidity of the NSVs was not affected by self-assembling of the surfactants into colloidal structures. Fluorescence anisotropy was found to be independent of the molar ratios of cholesteryl hemisuccinate and/or cholesterol during preparation of the NSVs. The anti-inflammatory AG drug showed no effect on the stability of the NSVs. In vivo experiments demonstrated that AG-loaded NSVs decreased edema and nociceptive responses when compared with AG alone and empty NSVs. In vitro and in vivo results demonstrated that pH sensitive and neutral NSVs show no statistical significant difference. CONCLUSION: NSVs were nontoxic and showed features favorable for potential administration in vivo. In addition, neutral NSVs showed signs of increased anti-inflammatory and antinociceptive responses when compared with AG.


