L-NicotineCAS# 54-11-5 |
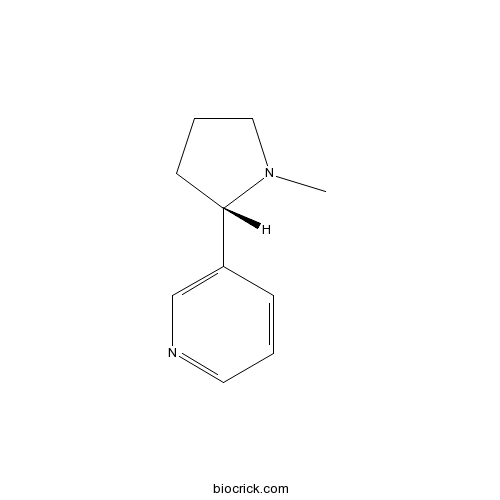
- (±)-Nicotine
Catalog No.:BCN9030
CAS No.:22083-74-5
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 54-11-5 | SDF | Download SDF |
| PubChem ID | 89594 | Appearance | Oil |
| Formula | C10H14N2 | M.Wt | 162.23 |
| Type of Compound | Alkaloids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 3-[(2S)-1-methylpyrrolidin-2-yl]pyridine | ||
| SMILES | CN1CCCC1C2=CN=CC=C2 | ||
| Standard InChIKey | SNICXCGAKADSCV-JTQLQIEISA-N | ||
| Standard InChI | InChI=1S/C10H14N2/c1-12-7-3-5-10(12)9-4-2-6-11-8-9/h2,4,6,8,10H,3,5,7H2,1H3/t10-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Nicotine is a potent inhibitor of cardiac A-type K+ channels, with blockade probably due to block of closed and open channels, this action may contribute to the ability of nicotine to affect cardiac electrophysiology and induce arrhythmias.Nicotine is able to activate mitogenic signalling pathways, which promote cell growth or survival as well as increase chemoresistance of cancer cells, nicotine activates its downstream signalling to interfere with the ubiquitination process and prevent Bcl-2 from being degraded in lung cancer cells, resulting in the increase of chemoresistance. |
| Targets | 5-HT Receptor | Dopamine Receptor | AChR | Potassium Channel |
| In vitro | Molecularly imprinted polymers as synthetic receptors for the QCM-D-based detection of L-nicotine in diluted saliva and urine samples.[Pubmed: 23754330]Anal Bioanal Chem. 2013 Aug;405(20):6479-87.Molecularly imprinted polymers (MIPs) are synthetic receptors that are able to specifically bind their target molecules in complex samples, making them a versatile tool in biosensor technology. The combination of MIPs as a recognition element with quartz crystal microbalances (QCM-D with dissipation monitoring) gives a straightforward and sensitive device, which can simultaneously measure frequency and dissipation changes. Heat-transfer-based detection of L-nicotine, histamine, and serotonin using molecularly imprinted polymers as biomimetic receptors.[Pubmed: 23685906]Anal Bioanal Chem. 2013 Aug;405(20):6453-60.
|
| In vivo | Discriminative stimulus effects of intravenous l-nicotine and nicotine analogs or metabolites in squirrel monkeys.[Pubmed: 2508155]Psychopharmacology (Berl). 1989;99(2):208-12.
|
| Cell Research | Uptake of L-nicotine and of 6-hydroxy-L-nicotine by Arthrobacter nicotinovorans and by Escherichia coli is mediated by facilitated diffusion and not by passive diffusion or active transport.[Pubmed: 19443550 ]Microbiology. 2009 Jun;155(Pt 6):1866-77.The mechanism by which L-Nicotine is taken up by bacteria that are able to grow on it is unknown. |

L-Nicotine Dilution Calculator

L-Nicotine Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 6.1641 mL | 30.8204 mL | 61.6409 mL | 123.2818 mL | 154.1022 mL |
| 5 mM | 1.2328 mL | 6.1641 mL | 12.3282 mL | 24.6564 mL | 30.8204 mL |
| 10 mM | 0.6164 mL | 3.082 mL | 6.1641 mL | 12.3282 mL | 15.4102 mL |
| 50 mM | 0.1233 mL | 0.6164 mL | 1.2328 mL | 2.4656 mL | 3.082 mL |
| 100 mM | 0.0616 mL | 0.3082 mL | 0.6164 mL | 1.2328 mL | 1.541 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Cefaclor
Catalog No.:BCC2527
CAS No.:53994-73-3
- Z-Phg-OH
Catalog No.:BCC2795
CAS No.:53990-33-3
- Luteolin 7,3'-di-O-glucuronide
Catalog No.:BCN5396
CAS No.:53965-08-5
- Ginsenoside F1
Catalog No.:BCN1244
CAS No.:53963-43-2
- Glycyrrhizic acid ammonium salt
Catalog No.:BCN5943
CAS No.:53956-04-0
- Aristolactam BIII
Catalog No.:BCN5718
CAS No.:53948-10-0
- Aristolactam BII
Catalog No.:BCN5717
CAS No.:53948-09-7
- Aristolactam AII
Catalog No.:BCN3924
CAS No.:53948-07-5
- Corylin
Catalog No.:BCN5716
CAS No.:53947-92-5
- Apterin
Catalog No.:BCN3910
CAS No.:53947-89-0
- Euparone
Catalog No.:BCN7204
CAS No.:53947-86-7
- Crotalarine
Catalog No.:BCN2076
CAS No.:53937-97-6
- Tryptophan
Catalog No.:BCN2615
CAS No.:54-12-6
- 5-Hydroxyindole-3-Acetic Acid
Catalog No.:BCC8285
CAS No.:54-16-0
- Sodium salicylate
Catalog No.:BCC4846
CAS No.:54-21-7
- Furosemide
Catalog No.:BCC3782
CAS No.:54-31-9
- Metyrapone
Catalog No.:BCC7632
CAS No.:54-36-4
- Idoxuridine
Catalog No.:BCC4666
CAS No.:54-42-2
- Pilocarpine HCl
Catalog No.:BCC4702
CAS No.:54-71-7
- Cinanserin hydrochloride
Catalog No.:BCC6653
CAS No.:54-84-2
- Isoniazid
Catalog No.:BCC9003
CAS No.:54-85-3
- Pentylenetetrazole
Catalog No.:BCC7453
CAS No.:54-95-5
- Amifampridine
Catalog No.:BCC5185
CAS No.:54-96-6
- Albendazole Oxide
Catalog No.:BCC4757
CAS No.:54029-12-8
Heat-transfer-based detection of L-nicotine, histamine, and serotonin using molecularly imprinted polymers as biomimetic receptors.[Pubmed:23685906]
Anal Bioanal Chem. 2013 Aug;405(20):6453-60.
In this work, we will present a novel approach for the detection of small molecules with molecularly imprinted polymer (MIP)-type receptors. This heat-transfer method (HTM) is based on the change in heat-transfer resistance imposed upon binding of target molecules to the MIP nanocavities. Simultaneously with that technique, the impedance is measured to validate the results. For proof-of-principle purposes, aluminum electrodes are functionalized with MIP particles, and L-Nicotine measurements are performed in phosphate-buffered saline solutions. To determine if this could be extended to other templates, histamine and serotonin samples in buffer solutions are also studied. The developed sensor platform is proven to be specific for a variety of target molecules, which is in agreement with impedance spectroscopy reference tests. In addition, detection limits in the nanomolar range could be achieved, which is well within the physiologically relevant concentration regime. These limits are comparable to impedance spectroscopy, which is considered one of the state-of-the-art techniques for the analysis of small molecules with MIPs. As a first demonstration of the applicability in biological samples, measurements are performed on saliva samples spiked with L-Nicotine. In summary, the combination of MIPs with HTM as a novel readout technique enables fast and low-cost measurements in buffer solutions with the possibility of extending to biological samples.
Discriminative stimulus effects of intravenous l-nicotine and nicotine analogs or metabolites in squirrel monkeys.[Pubmed:2508155]
Psychopharmacology (Berl). 1989;99(2):208-12.
Squirrel monkeys were trained to emit one response after IV administration of L-Nicotine (0.4 or 0.2 mumol/kg) and a different response after IV administration of saline. After stable discriminative performances were established, subjects were tested with cumulative doses of L-Nicotine (0.02-2.2 mumol/kg), d-nicotine (0.02-19.7 mumol/kg), l-nornicotine (0.2-12.0 mumol/kg), l-cotinine (56.8-567.5 mumol/kg), and dl-anabasine (0.6-19.7 mumol/kg). All of the drugs produced dose-related increases in the percentage of drug-appropriate responses emitted, from predominantly saline-appropriate responses after low doses, to predominantly drug-appropriate responses at the highest doses studied. Relative potency comparisons indicated that L-Nicotine was 28 times more potent than d-nicotine, 29 times more potent than l-nornicotine, and approximately 2000 times more potent than l-cotinine. Each of the drugs also produced decreases in rates of responding, with potency order similar to that obtained for the discriminative effects. The effects of l-cotinine may be attributed to trace amounts of L-Nicotine, which existed within the l-cotinine. The effects of dl-anabasine were lethal in one subject and were consequently not studied in the other subjects.
Uptake of L-nicotine and of 6-hydroxy-L-nicotine by Arthrobacter nicotinovorans and by Escherichia coli is mediated by facilitated diffusion and not by passive diffusion or active transport.[Pubmed:19443550]
Microbiology. 2009 Jun;155(Pt 6):1866-77.
The mechanism by which L-Nicotine is taken up by bacteria that are able to grow on it is unknown. Nicotine degradation by Arthrobacter nicotinovorans, a Gram-positive soil bacterium, is linked to the presence of the catabolic megaplasmid pAO1. l-[(14)C]Nicotine uptake assays with A. nicotinovorans showed transport of nicotine across the cell membrane to be energy-independent and saturable with a K(m) of 6.2+/-0.1 microM and a V(max) of 0.70+/-0.08 micromol min(-1) (mg protein)(-1). This is in accord with a mechanism of facilitated diffusion, driven by the nicotine concentration gradient. Nicotine uptake was coupled to its intracellular degradation, and an A. nicotinovorans strain unable to degrade nicotine (pAO1(-)) showed no nicotine import. However, when the nicotine dehydrogenase genes were expressed in this strain, import of l-[(14)C]nicotine took place. A. nicotinovorans pAO1(-) and Escherichia coli were also unable to import 6-hydroxy-L-Nicotine, but expression of the 6-hydroxy-L-Nicotine oxidase gene allowed both bacteria to take up this compound. L-Nicotine uptake was inhibited by d-nicotine, 6-hydroxy-L-Nicotine and 2-amino-L-Nicotine, which may indicate transport of these nicotine derivatives by a common permease. Attempts to correlate nicotine uptake with pAO1 genes possessing similarity to amino acid transporters failed. In contrast to the situation at the blood-brain barrier, nicotine transport across the cell membrane by these bacteria was not by passive diffusion or active transport but by facilitated diffusion.
Molecularly imprinted polymers as synthetic receptors for the QCM-D-based detection of L-nicotine in diluted saliva and urine samples.[Pubmed:23754330]
Anal Bioanal Chem. 2013 Aug;405(20):6479-87.
Molecularly imprinted polymers (MIPs) are synthetic receptors that are able to specifically bind their target molecules in complex samples, making them a versatile tool in biosensor technology. The combination of MIPs as a recognition element with quartz crystal microbalances (QCM-D with dissipation monitoring) gives a straightforward and sensitive device, which can simultaneously measure frequency and dissipation changes. In this work, bulk-polymerized L-Nicotine MIPs were used to test the feasibility of L-Nicotine detection in saliva and urine samples. First, L-Nicotine-spiked saliva and urine were measured after dilution in demineralized water and 0.1x phosphate-buffered saline solution for proof-of-concept purposes. L-Nicotine could indeed be detected specifically in the biologically relevant micromolar concentration range. After successfully testing on spiked samples, saliva was analyzed, which was collected during chewing of either nicotine tablets with different concentrations or of smokeless tobacco. The MIPs in combination with QCM-D were able to distinguish clearly between these samples: This proves the functioning of the concept with saliva, which mediates the oral uptake of nicotine as an alternative to the consumption of cigarettes.


