MetyraponeCytochrome P450 inhibitor CAS# 54-36-4 |
- Dexpramipexole dihydrochloride
Catalog No.:BCC1528
CAS No.:104632-27-1
- Dexpramipexole
Catalog No.:BCC1527
CAS No.:104632-28-2
- Cariprazine hydrochloride
Catalog No.:BCC1454
CAS No.:1083076-69-0
- Cariprazine
Catalog No.:BCC1453
CAS No.:839712-12-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 54-36-4 | SDF | Download SDF |
| PubChem ID | 4174 | Appearance | Powder |
| Formula | C14H14N2O | M.Wt | 226.27 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | Su-4885 | ||
| Solubility | DMSO : 250 mg/mL (1104.87 mM; Need ultrasonic) H2O : ≥ 38 mg/mL (167.94 mM) *"≥" means soluble, but saturation unknown. | ||
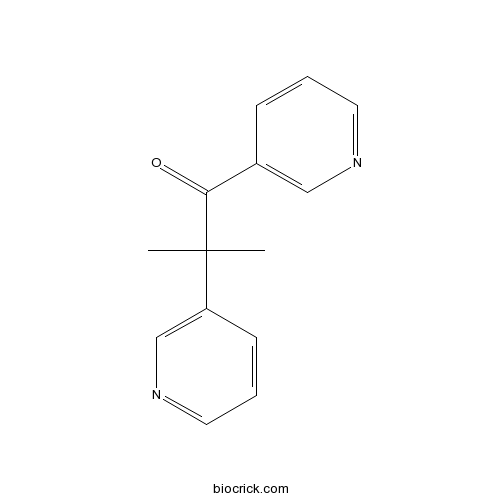
| Chemical Name | 2-methyl-1,2-dipyridin-3-ylpropan-1-one | ||
| SMILES | CC(C)(C1=CN=CC=C1)C(=O)C2=CN=CC=C2 | ||
| Standard InChIKey | FJLBFSROUSIWMA-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C14H14N2O/c1-14(2,12-6-4-8-16-10-12)13(17)11-5-3-7-15-9-11/h3-10H,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Cytochrome P450 inhibitor. Blocks glucocorticoid synthesis via inhibition of steroid 11-β hydroxylase (CYP11B1) activity (IC50 = 7.83 μM). Also inhibits CYP3A4 and cytochrome P450-mediated ω/ω-1 hydroxylase activity. |

Metyrapone Dilution Calculator

Metyrapone Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 4.4195 mL | 22.0975 mL | 44.195 mL | 88.39 mL | 110.4875 mL |
| 5 mM | 0.8839 mL | 4.4195 mL | 8.839 mL | 17.678 mL | 22.0975 mL |
| 10 mM | 0.4419 mL | 2.2097 mL | 4.4195 mL | 8.839 mL | 11.0487 mL |
| 50 mM | 0.0884 mL | 0.4419 mL | 0.8839 mL | 1.7678 mL | 2.2097 mL |
| 100 mM | 0.0442 mL | 0.221 mL | 0.4419 mL | 0.8839 mL | 1.1049 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Metyrapone is an inhibitor of cytochrome P450-mediated ω/ω-1 hydroxylase activity and CYP11B1. Target: CYP11B1 Metyrapone is a drug used in the diagnosis of adrenal insufficiency and occasionally in the treatment of Cushing's syndrome (hypercortisolism). Metyrapone blocks cortisol synthesis by reversibly inhibiting steroid 11β-hydroxylase. This stimulates ACTH secretion, which in turn increases plasma 11-Deoxycortisol levels.
- Furosemide
Catalog No.:BCC3782
CAS No.:54-31-9
- Sodium salicylate
Catalog No.:BCC4846
CAS No.:54-21-7
- 5-Hydroxyindole-3-Acetic Acid
Catalog No.:BCC8285
CAS No.:54-16-0
- Tryptophan
Catalog No.:BCN2615
CAS No.:54-12-6
- L-Nicotine
Catalog No.:BCN6269
CAS No.:54-11-5
- Cefaclor
Catalog No.:BCC2527
CAS No.:53994-73-3
- Z-Phg-OH
Catalog No.:BCC2795
CAS No.:53990-33-3
- Luteolin 7,3'-di-O-glucuronide
Catalog No.:BCN5396
CAS No.:53965-08-5
- Ginsenoside F1
Catalog No.:BCN1244
CAS No.:53963-43-2
- Glycyrrhizic acid ammonium salt
Catalog No.:BCN5943
CAS No.:53956-04-0
- Aristolactam BIII
Catalog No.:BCN5718
CAS No.:53948-10-0
- Aristolactam BII
Catalog No.:BCN5717
CAS No.:53948-09-7
- Idoxuridine
Catalog No.:BCC4666
CAS No.:54-42-2
- Pilocarpine HCl
Catalog No.:BCC4702
CAS No.:54-71-7
- Cinanserin hydrochloride
Catalog No.:BCC6653
CAS No.:54-84-2
- Isoniazid
Catalog No.:BCC9003
CAS No.:54-85-3
- Pentylenetetrazole
Catalog No.:BCC7453
CAS No.:54-95-5
- Amifampridine
Catalog No.:BCC5185
CAS No.:54-96-6
- Albendazole Oxide
Catalog No.:BCC4757
CAS No.:54029-12-8
- Etonogestrel
Catalog No.:BCC5230
CAS No.:54048-10-1
- Tofacitinib (CP-690550) Citrate
Catalog No.:BCC2189
CAS No.:540737-29-9
- Palosuran
Catalog No.:BCC4311
CAS No.:540769-28-6
- Isoastilbin
Catalog No.:BCN5719
CAS No.:54081-48-0
- 2-(1-Hydroxy-1-methylethyl)-4-methoxy-7H-furo[3,2-g][1]benzopyran-7-one
Catalog No.:BCN1422
CAS No.:54087-32-0
Effects of the combination of metyrapone and oxazepam on cocaine-induced increases in corticosterone in the medial prefrontal cortex and nucleus accumbens.[Pubmed:28024272]
Psychoneuroendocrinology. 2017 Mar;77:75-83.
We have previously demonstrated that a combination of drugs (i.e., Metyrapone and oxazepam) known to attenuate HPA-axis activity effectively decreases cocaine self-administration and cue reactivity in rats. However, we did not find changes in plasma corticosterone that matched the behavioral effects we observed, indicating that a different mechanism of action must be involved. Therefore, we hypothesized that the combination of Metyrapone and oxazepam attenuates cocaine taking and seeking by decreasing cocaine-induced increases in corticosterone in the brain. Male rats were implanted with guide cannulae targeting the medial prefrontal cortex or nucleus accumbens. After the rats recovered from surgery, the microdialysis session was conducted. Rats were housed in the experimental chamber and the dialysis probes inserted into the guide cannulae the night before the session. The following day, dialysate samples were collected over a five-hour session. Baseline samples were collected for the first two hours, every 20min. Samples were then collected following administration of cocaine (15mg/kg, ip). Before injections of cocaine, rats were pretreated with either vehicle or the combination of Metyrapone (50mg/kg, ip) and oxazepam (10mg/kg, ip). The administration of cocaine resulted in an increase in corticosterone in the medial prefrontal cortex following vehicle pretreatment, which was not observed in the nucleus accumbens. This cocaine-induced increase in corticosterone was attenuated by Metyrapone/oxazepam. Reducing cocaine-induced increases in corticosterone in the medial prefrontal cortex might represent a novel mechanism through which the combination of Metyrapone/oxazepam produces its behavioral effects.
A successful case of pregnancy in a woman with ACTH-independent Cushing's syndrome treated with ketoconazole and metyrapone.[Pubmed:28277127]
Gynecol Endocrinol. 2017 May;33(5):349-352.
Cushing's syndrome (CS) is a rare disease caused by a chronic excess of cortisol. Hypercortisolaemia may affect reproductive system leading to infertility in women. However, some of the patients remain fertile, although pregnancy is uncommon. In our report, we describe the case of a 31-years old woman suffering from hypertension, oligomenorrhea, easy bruising, muscle weakness and elevated levels of cortisol. During hospitalization, high level of serum cortisol with stiff diurnal rhythm and undetectable plasma ACTH concentration were found. The overnight 1 mg dexamethasone (DEX) suppression test and the test with 8 mg of DEX were performed - plasma cortisol levels after both doses of DEX were over expected values. Thus, the diagnosis of ACTH independent hypercortisolaemia was established. After three weeks of ketoconazole treatment, high level of beta-HCG was found corresponding to the third week of pregnancy. The ketoconazole was shift to Metyrapone but afterwards ketoconazole was added again. The treatment was well tolerated and pregnancy proceeded without complications. US scan revealed a 2 cm adenoma of the left adrenal gland, confirmed by CT. An adrenalectomy was performed. Concluding, we think that medical treatment of CS in pregnant women is well tolerated and safe both for the mother and fetus.
Prospective evaluation of a week one overnight metyrapone test with subsequent dynamic assessments of hypothalamic-pituitary-adrenal axis function after pituitary surgery.[Pubmed:28329436]
Clin Endocrinol (Oxf). 2017 Jul;87(1):35-43.
OBJECTIVE: To determine whether an overnight Metyrapone test (OMT) within the first week postpituitary surgery can definitively assess the hypothalamic-pituitary-adrenal (HPA) axis, compared with subsequent dynamic tests and glucocorticoid requirement at 6 months. DESIGN: Prospective study measuring morning cortisol levels on days 3 and 4 post-operatively, OMT day 5-7 and week 6, week 6 250 mug short Synacthen test (SST) and week 7 insulin tolerance test (ITT). PATIENTS AND MEASUREMENTS: Forty participants who underwent pituitary surgery at a single centre (Cushing's disease excluded) were followed for at least 6 months. 46% had pre-operative adrenal insufficiency. PRIMARY OUTCOME: week 1 OMT compared to glucocorticoid requirement at 6 months. SECONDARY OUTCOMES: the performance of ITT as a "definitive" test and all tests compared to glucocorticoid requirement at 6 months. RESULTS: Week 1 OMT showed concordance with ITT at week 7 of 78% and glucocorticoid requirement at 6 months of 81% respectively which was not significantly different from post-operative morning cortisol levels; 37% of participants with an abnormal OMT on day 6 had a normal OMT at week 6. All HPA axis tests showed similar concordance with glucocorticoid requirement at 6 months of 80%-85%. CONCLUSIONS: Overnight Metyrapone test within the first week after pituitary surgery was no better than an early morning cortisol level at predicting glucocorticoid requirement at 6 months. OMT at week 6 demonstrated recovery of HPA axis in a substantial proportion of participants who failed earlier assessments; thus, definitive testing should be delayed until 6 weeks post-operatively.
Postnatal treatment with metyrapone attenuates the effects of diet-induced obesity in female rats exposed to early-life stress.[Pubmed:27965205]
Am J Physiol Endocrinol Metab. 2017 Feb 1;312(2):E98-E108.
Experimental studies in rodents have shown that females are more susceptible to exhibiting fat expansion and metabolic disease compared with males in several models of fetal programming. This study tested the hypothesis that female rat pups exposed to maternal separation (MatSep), a model of early-life stress, display an exacerbated response to diet-induced obesity compared with male rats. Also, we tested whether the postnatal treatment with Metyrapone (MTP), a corticosterone synthase inhibitor, would attenuate this phenotype. MatSep was performed in WKY offspring by separation from the dam (3 h/day, postnatal days 2-14). Upon weaning, male and female rats were placed on a normal (ND; 18% kcal fat) or high-fat diet (HFD; 60% kcal fat). Nondisturbed littermates served as controls. In male rats, no diet-induced differences in body weight (BW), glucose tolerance, and fat tissue weight and morphology were found between MatSep and control male rats. However, female MatSep rats displayed increased BW gain, fat pad weights, and glucose intolerance compared with control rats (P < 0.05). Also, HFD increased plasma corticosterone (196 +/- 51 vs. 79 +/- 18 pg/ml, P < 0.05) and leptin levels (1.8 +/- 0.4 vs. 1.3 +/- 0.1 ng/ml, P < 0.05) in female MatSep compared with control rats, whereas insulin and adiponectin levels were similar between groups. Female control and MatSep offspring were treated with MTP (50 microg/g ip) 30 min before the daily separation. MTP treatment significantly attenuated diet-induced obesity risk factors, including elevated adiposity, hyperleptinemia, and glucose intolerance. These findings show that exposure to stress hormones during early life could be a key event to enhance diet-induced obesity and metabolic disease in female rats. Thus, pharmacological and/or behavioral inflection of the stress levels is a potential therapeutic approach for prevention of early life stress-enhanced obesity and metabolic disease.
Structural and dynamical basis of broad substrate specificity, catalytic mechanism, and inhibition of cytochrome P450 3A4.[Pubmed:16190729]
J Am Chem Soc. 2005 Oct 5;127(39):13634-42.
Cytochrome P450 (CYP) 3A4 is responsible for the oxidative degradation of more than 50% of clinically used drugs. By means of molecular dynamics simulations with the newly developed force field parameters for the heme-thiolate group and its dioxygen adduct, we examine the differences in structural and dynamic properties between CYP3A4 in the resting form and its complexes with the substrate progesterone and the inhibitor Metyrapone. The results indicate that the broad substrate specificity of CYP3A4 stems from the malleability of a loop (residues 211-218) that resides in the vicinity of the channel connecting the active site and bulk solvent. However, the high-amplitude motion of the flexible loop is found to be damped out upon binding of the inhibitor or the substrate in the active site. In the resting form of CYP3A4, a structural water molecule is bound to the sixth coordination position of the heme iron, stabilizing the octahedral coordination geometry. In addition to the direct coordination of Metyrapone to the heme iron, the hydrogen bond interaction between the inhibitor carbonyl group and the side chain of Ser119 also contributes significantly to stabilizing the CYP3A4-Metyrapone complex. On the other hand, progesterone is stabilized in the active site by the formation of two hydrogen bonds with Ser119 and Arg106, as well as by the van der Waals interactions with the heme and hydrophobic residues. The structural and dynamic features of the CYP3A4-progesterone complex indicate that the oxidative degradation of progesterone occurs through hydroxylation at the C16 position by the reactive oxygen coordinated to the heme iron.
Cytochrome P450-mediated prostaglandin omega/omega-1 hydroxylase activities in porcine ciliary body epithelial cells.[Pubmed:1426070]
Exp Eye Res. 1992 Aug;55(2):377-84.
The ocular hypotensive effect of topically applied prostaglandins (PGs) is well documented. Although PGs introduced in the posterior chamber accumulate in the anterior tissues (e.g. iris/ciliary complex), little is known about the metabolism of PGs by these tissues. We have recently found that non-pigmented epithelial (NPE) cells and pigmented epithelial (PE) cells are readily separated from porcine ciliary body and cytochrome P450-dependent xenobiotic metabolism is considerably higher in NPE cells than in PE cells. We have therefore investigated in this study the cytochrome P450-mediated PG omega/omega-1 hydroxylase activities of porcine ciliary epithelial cells. The NPE cells show about three times higher activities than do PE cells; the NPE cells, in fact, demonstrate the highest PG omega/omega-1 hydroxylase activities among different ocular tissues. Both omega and omega-1 hydroxylases show broad substrate specificities and hydroxylate PGA1, A2, E1, E2, and lauric acid. The omega/omega-1 hydroxylase activities of NPE and PE, as determined with PGA2 and lauric acid as substrates, are enhanced or induced by treatment of primary cultures of the individual epithelial cells with clofibrate, both activities reaching maximum levels within 48 hr of induction. The induced activities are inhibited almost completely by cycloheximide and actinomycin D. The omega/omega-1 hydroxylase activities of both NPE and PE cells require NADPH and molecular oxygen, are associated with the microsomal fraction, respond to inducers such as clofibrate, and are inhibited by Metyrapone and SKF525 A (inhibitors of P450 enzymes). These results support the suggestion that PG omega/omega-1 hydroxylations by NPE and PE are cytochrome P450-mediated reactions.(ABSTRACT TRUNCATED AT 250 WORDS)
Structure-activity relationship study of the inhibition of adrenal cortical 11 beta-hydroxylase by new metyrapone analogues.[Pubmed:6606707]
J Med Chem. 1984 Jan;27(1):15-9.
Metyrapone, 2-methyl-1,2-di-3-pyridyl-1-propanone (1a), is a potent reversible inhibitor of the cytochrome P-450 11 beta-hydroxylase enzyme system (P-450(11) beta) of the adrenal cortex. The structural features of the Metyrapone molecule have been systemically altered to determine the requirements necessary for inhibition of P-450(11) beta activity. Metyrapone and 14 analogues have been obtained or synthesized and evaluated as inhibitors using a crude, defatted bovine adrenal cortical mitochondrial preparation. The inhibition of P-450(11) beta activity with these derivatives demonstrated that (1) the A-ring phenyl derivatives 2a-d were better inhibitors than the respective dipyridyl analogues, (2) the ketone in the 1-position can be replaced by various functionalities without markedly reducing inhibition, and (3) at least one methyl group should be present in the 2-position to maintain inhibition. The observed inhibition of P450(11) beta activity with the Metyrapone analogues suggest that A-ring phenyl Metyrapone analogues 2a-d would be candidates for radioiodination and subsequently used as adrenal cortical imaging agents.


