MenisdaurilideCAS# 67765-59-7 |
- Aquilegiolide
Catalog No.:BCN9602
CAS No.:94481-79-5
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 67765-59-7 | SDF | Download SDF |
| PubChem ID | 155054 | Appearance | Powder |
| Formula | C8H8O3 | M.Wt | 152.15 |
| Type of Compound | Other NPs | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
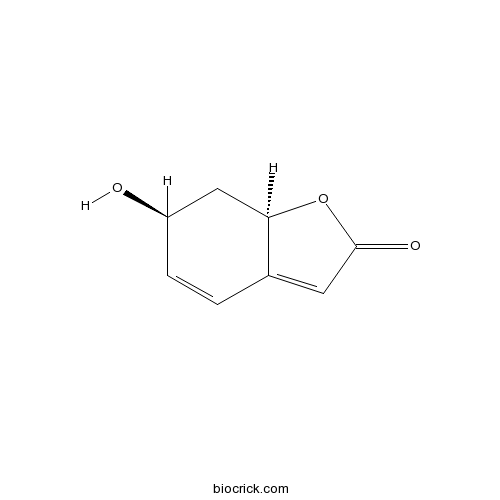
| Chemical Name | (6S,7aR)-6-hydroxy-7,7a-dihydro-6H-1-benzofuran-2-one | ||
| SMILES | C1C(C=CC2=CC(=O)OC21)O | ||
| Standard InChIKey | RAXNUTINVDSFEU-RNFRBKRXSA-N | ||
| Standard InChI | InChI=1S/C8H8O3/c9-6-2-1-5-3-8(10)11-7(5)4-6/h1-3,6-7,9H,4H2/t6-,7-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Menisdaurilide Dilution Calculator

Menisdaurilide Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 6.5725 mL | 32.8623 mL | 65.7246 mL | 131.4492 mL | 164.3115 mL |
| 5 mM | 1.3145 mL | 6.5725 mL | 13.1449 mL | 26.2898 mL | 32.8623 mL |
| 10 mM | 0.6572 mL | 3.2862 mL | 6.5725 mL | 13.1449 mL | 16.4312 mL |
| 50 mM | 0.1314 mL | 0.6572 mL | 1.3145 mL | 2.629 mL | 3.2862 mL |
| 100 mM | 0.0657 mL | 0.3286 mL | 0.6572 mL | 1.3145 mL | 1.6431 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Aquilegiolide
Catalog No.:BCN9602
CAS No.:94481-79-5
- 16-Oxoserratenediol
Catalog No.:BCN9601
CAS No.:24513-52-8
- Norushinsunine
Catalog No.:BCN9600
CAS No.:3175-84-6
- 3β-Hydroxy-7β,25-dimethoxycucurbita-5,23-dien-19-al
Catalog No.:BCN9599
CAS No.:85372-69-6
- (2R,3R)-Glucodistylin
Catalog No.:BCN9598
CAS No.:27297-45-6
- Sinococuline
Catalog No.:BCN9597
CAS No.:109351-36-2
- Tannagine
Catalog No.:BCN9596
CAS No.:123750-34-5
- Junosine
Catalog No.:BCN9595
CAS No.:103956-34-9
- Citrusinine I
Catalog No.:BCN9594
CAS No.:86680-32-2
- 6a,7-Dehydroboldine
Catalog No.:BCN9593
CAS No.:91599-23-4
- Boschnaloside
Catalog No.:BCN9592
CAS No.:72963-55-4
- Taccaoside E
Catalog No.:BCN9591
CAS No.:1858199-00-4
- 7,7'-Dihydrotaiwanin C
Catalog No.:BCN9604
CAS No.:216955-79-2
- Stepharine
Catalog No.:BCN9605
CAS No.:2810-21-1
- Taiwanin C
Catalog No.:BCN9606
CAS No.:14944-34-4
- Beiwutine
Catalog No.:BCN9607
CAS No.:76918-93-9
- Apigenin 7-O-(2'',6''-di-O-E-p-coumaroyl)glucoside
Catalog No.:BCN9608
CAS No.:1448779-19-8
- Karavilagenin F
Catalog No.:BCN9609
CAS No.:1639024-15-9
- 3-O-(p-Hydroxybenzoyl)serratriol
Catalog No.:BCN9610
CAS No.:1448534-93-7
- 5-Methoxyjusticidin A
Catalog No.:BCN9611
CAS No.:205505-62-0
- Taiwanin E
Catalog No.:BCN9612
CAS No.:22743-05-1
- 6,7-Di-O-acetylsinococuline
Catalog No.:BCN9613
CAS No.:1054312-81-0
- Illiciumlignan D
Catalog No.:BCN9614
CAS No.:2237239-36-8
- Tuberculatin
Catalog No.:BCN9615
CAS No.:90706-10-8
Preparation of menisdaurigenin and related compounds.[Pubmed:30094599]
J Nat Med. 2019 Jan;73(1):236-243.
Menisdaurin (1), a cyano glucoside, was first isolated in 1978 from Menispermum dauricum (Menispermaceae) and named after the plant. It has been also isolated from several plant sources. The stereochemistry of the aglycone part was first reported as (Z,4R,6S)-enantiomer of (4,6-dihydroxy-2-cyclohexen-1-ylidene)acetonitrile based on the CD spectrum of Menisdaurilide (2), the alpha,beta-unsaturated gamma-lactone obtained by an acid hydrolysis of menisdaurin. Later, the absolute stereochemistry was revised as (Z,4S,6R) by X-ray crystal analysis of 1 isolated from Saniculiphyllum guangxiens. The aglycone part of menisdaurin (1) has not been obtained from 1, because an acid hydrolysis of 1 gave Menisdaurilide (2), and enzymatic hydrolysis with emulsin did not give the aglycone. On the other hand, a compound named coculauril (3) was isolated from Cocculus lauriforius. This compound has the same planner structure corresponding to the aglycone of 1, but the stereochemistry was reported to be (E,4R,6S). Here, we confirmed the absolute stereochemistry of 1 by Mosher's method to be (Z,4S,6R), and prepared the aglycone of 1, i.e., menisdaurigenin (4) by an enzymatic hydrolysis of 1. We also revealed that 4 is a different compound from 3 and unstable in water and MeOH.
Four New Cyclohexylideneacetonitrile Derivatives from the Hypocotyl of Mangrove (Bruguiera gymnorrhiza).[Pubmed:26274945]
Molecules. 2015 Aug 12;20(8):14565-75.
Four new cyclohexylideneacetonitrile derivatives 1-4, named menisdaurins B-E, as well as three known cyclohexylideneacetonitrile derivatives--menisdaurin (5), coclauril (6), and Menisdaurilide (7)--were isolated from the hypocotyl of a mangrove (Bruguiera gymnorrhiza). The structures of the isolates were elucidated on the basis of extensive spectroscopic analysis. Compounds 1-7 showed anti-Hepatitis B virus (HBV) activities, with EC50 values ranging from 5.1 +/- 0.2 mug/mL to 87.7 +/- 5.8 mug/mL.
Diastereoselective synthesis of allosecurinine and viroallosecurinine from menisdaurilide.[Pubmed:18781804]
J Org Chem. 2008 Oct 3;73(19):7657-62.
A new and versatile synthetic route to Securinega alkaloids is reported. The first synthesis of allosecurinine has been accomplished in seven steps and 40% yield, starting from (+)-Menisdaurilide, using a vinylogous Mannich reaction as the key transformation. Similarly, viroallosecurinine has been synthesized from (-)-Menisdaurilide.
Discovery of the apoptosis-inducing activity and high accumulation of the butenolides, menisdaurilide and aquilegiolide in Dicentra spectabilis.[Pubmed:17999351]
Planta Med. 2007 Dec;73(15):1543-47.
The occurrence of two butenolides, Menisdaurilide and aquilegiolide, in commercial specimens of Dicentra spectabilis is reported for the first time; a rapid and direct isolation protocol is described. The ability of these lactones to induce apoptosis in human tumour cell lines at 10 microM concentration is also described. The high abundance and apoptosis-inducing activity reported here indicates that these constituents play a more significant role than the hormonal action previously attributed to them.
7,8-Dihydrobenzofuranones from Ouratea reticulata.[Pubmed:11543976]
Fitoterapia. 2001 Aug;72(6):706-8.
Two known 7,8-dihydrobenzofuranones, aquilegiolide (1) and Menisdaurilide (2), were isolated from Ouratea reticulata root bark.


