Neuropeptide FFEndogenous NPFF1 and NPFF2 agonist CAS# 99566-27-5 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 99566-27-5 | SDF | Download SDF |
| PubChem ID | 123797 | Appearance | Powder |
| Formula | C54H76N14O10 | M.Wt | 1081.28 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 0.50 mg/ml in water | ||
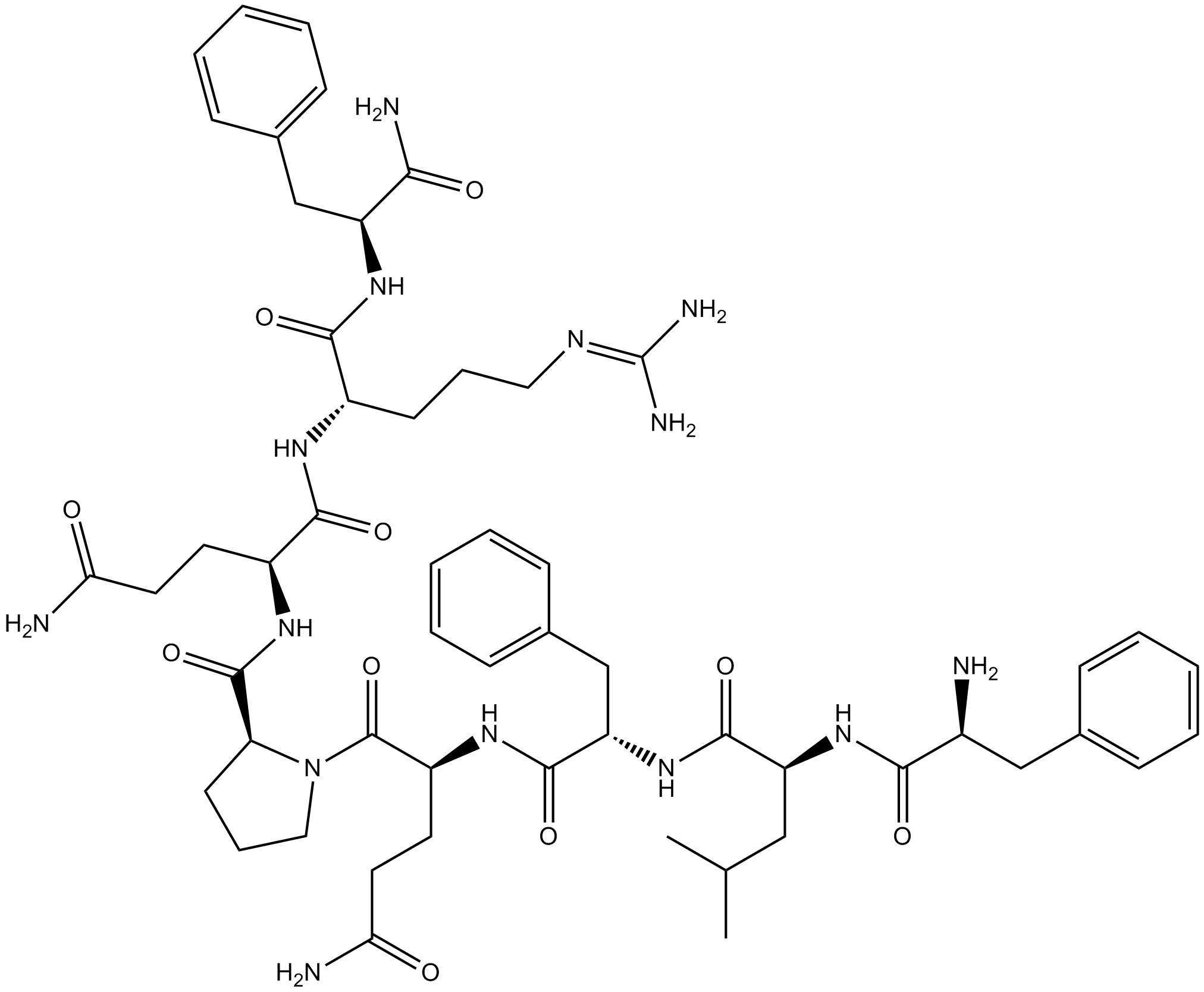
| Sequence | FLFQPQRF (Modifications: Phe-8 = C-terminal amide) | ||
| Chemical Name | (2S)-2-[[(2S)-1-[(2S)-5-amino-2-[[(2S)-2-[[(2S)-2-[(2-amino-3-phenylpropanoyl)amino]-4-methylpentanoyl]amino]-3-phenylpropanoyl]amino]-5-oxopentanoyl]pyrrolidine-2-carbonyl]amino]-N-[(2S)-1-[(1-amino-1-oxo-3-phenylpropan-2-yl)amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]pentanediamide | ||
| SMILES | CC(C)CC(C(=O)NC(CC1=CC=CC=C1)C(=O)NC(CCC(=O)N)C(=O)N2CCCC2C(=O)NC(CCC(=O)N)C(=O)NC(CCCN=C(N)N)C(=O)NC(CC3=CC=CC=C3)C(=O)N)NC(=O)C(CC4=CC=CC=C4)N | ||
| Standard InChIKey | HWYCFZUSOBOBIN-FPAYTFQGSA-N | ||
| Standard InChI | InChI=1S/C54H76N14O10/c1-32(2)28-41(66-47(72)36(55)29-33-14-6-3-7-15-33)50(75)67-42(31-35-18-10-5-11-19-35)51(76)64-39(23-25-45(57)70)53(78)68-27-13-21-43(68)52(77)63-38(22-24-44(56)69)49(74)62-37(20-12-26-61-54(59)60)48(73)65-40(46(58)71)30-34-16-8-4-9-17-34/h3-11,14-19,32,36-43H,12-13,20-31,55H2,1-2H3,(H2,56,69)(H2,57,70)(H2,58,71)(H,62,74)(H,63,77)(H,64,76)(H,65,73)(H,66,72)(H,67,75)(H4,59,60,61)/t36?,37-,38-,39-,40?,41-,42-,43-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Endogenous antiopioid peptide and agonist at NPFF1 and NPFF2 receptors (Ki values are 2.82 and 0.21 nM respectively). Exhibits anorexigenic effects following i.c.v. administration. |

Neuropeptide FF Dilution Calculator

Neuropeptide FF Molarity Calculator

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- K 252a
Catalog No.:BCC7152
CAS No.:99533-80-9
- L-651,582
Catalog No.:BCC7561
CAS No.:99519-84-3
- 3-Ethoxy-4-ethoxycarbonyl phenylacetic acid
Catalog No.:BCC8629
CAS No.:99469-99-5
- 1-Chloroethyl cyclohexyl carbonate
Catalog No.:BCC8463
CAS No.:99464-83-2
- Ampiroxicam
Catalog No.:BCC4426
CAS No.:99464-64-9
- BTZO 1
Catalog No.:BCC7886
CAS No.:99420-15-2
- Methyl rosmarinate
Catalog No.:BCN4536
CAS No.:99353-00-1
- Kadsurin A
Catalog No.:BCN6515
CAS No.:99340-07-5
- Venlafaxine Hydrochloride
Catalog No.:BCC2513
CAS No.:99300-78-4
- Levodropropizine
Catalog No.:BCC4520
CAS No.:99291-25-5
- Proglumide sodium salt
Catalog No.:BCC5768
CAS No.:99247-33-3
- Mulberrofuran G pentaacetate
Catalog No.:BCN6518
CAS No.:99217-75-1
- Sertaconazole
Catalog No.:BCC9146
CAS No.:99592-32-2
- Sertaconazole nitrate
Catalog No.:BCC4716
CAS No.:99592-39-9
- Ondansetron HCl
Catalog No.:BCC2493
CAS No.:99614-01-4
- Ondansetron
Catalog No.:BCC5043
CAS No.:99614-02-5
- Leucanthogenin
Catalog No.:BCN7932
CAS No.:99615-00-6
- Isothymonin
Catalog No.:BCN3393
CAS No.:99615-01-7
- Kazinol B
Catalog No.:BCN4538
CAS No.:99624-27-8
- Kazinol A
Catalog No.:BCN3388
CAS No.:99624-28-9
- ent-3beta,18-Dihydroxylabda-8(17),13E-dien-15-oic acid
Catalog No.:BCN7669
CAS No.:99624-39-2
- Uncinatone
Catalog No.:BCN4547
CAS No.:99624-92-7
- Ro 19-4603
Catalog No.:BCC7228
CAS No.:99632-94-7
- 14-Benzoylneoline
Catalog No.:BCN6493
CAS No.:99633-05-3
Relationship of neuropeptide FF receptors with pubertal maturation of gilts.[Pubmed:28339619]
Biol Reprod. 2017 Mar 1;96(3):617-634.
Mechanisms governing the timing of puberty in pigs are poorly understood. A genome-wide association study for age at first estrus in pigs identified candidate genes including Neuropeptide FF receptor 2 (NPFFR2), which is a putative receptor for RFamide-related peptides (RFRP). RFRP has been shown to negatively regulate secretion of reproductive hormones from hypothalamic and pituitary tissue of pigs in culture. Here, the porcine NPFFR2 gene was further screened and four potentially functional variants were identified to be associated with age at first estrus in pigs (1,288 gilts). The RFRP neurons in the porcine hypothalamus were localized in the paraventricular and dorsomedial nuclei with RFRP fibers in the lateral hypothalamic area. There were marked changes in expression of NPFF receptors in the anterior pituitary gland and hypothalamus of gilts beginning with the peripubertal period. The hypothesis that NPFF receptor function is related to secretion of luteinizing hormone (LH) in gilts was tested with various NPFF receptor ligands. The NPFF receptor antagonist RF9 stimulated a pulse-like release of LH in prepubertal gilts. The putative NPFF receptor agonist RFRP3 modestly suppressed LH pulses in ovariectomized (OVX) prepubertal gilts. A porcine-specific RFRP2 failed to have an effect on LH secretion in OVX prepubertal gilts despite its high degree of homology to avian gonadotropin-inhibitory hormone. Results indicate that an RFRP system is present in the pig and that NPFFR2 is important for pubertal onset in gilts. It is not clear if this regulation involves major control of LH secretion or another unknown mechanism.
Structure-Based Optimization of Multifunctional Agonists for Opioid and Neuropeptide FF Receptors with Potent Nontolerance Forming Analgesic Activities.[Pubmed:27798836]
J Med Chem. 2016 Nov 23;59(22):10198-10208.
The opioid and Neuropeptide FF pharmacophore-containing chimeric peptide 0 (BN-9) was recently developed and produced potent nontolerance forming analgesia. In this study, 11 analogues of 0 were designed and synthesized. An in vitro cAMP assay demonstrated that these analogues behaved as multifunctional agonists at both opioid and NPFF receptors. In mouse tail-flick test, most of the analogues produced potent nontolerance forming antinociception. Notably, 11 (DN-9) was 33-fold more potent than 0 at analgesic effects, which was mediated by mu- and kappa-opioid receptors. In addition, 11 also produced powerful analgesic effects in the formalin pain and CFA-induced chronic inflammatory pain models. Strikingly, following its repeated administration for 6 days, 11 did not produce antinociceptive tolerance in the tail-flick test and CFA-induced pain model. The present work indicates that it is reasonable to design multifunctional peptide ligands for opioid and NPFF receptors in a single molecule producing effective nontolerance forming antinociception.
RF313, an orally bioavailable neuropeptide FF receptor antagonist, opposes effects of RF-amide-related peptide-3 and opioid-induced hyperalgesia in rodents.[Pubmed:28288815]
Neuropharmacology. 2017 May 15;118:188-198.
Although opiates represent the most effective analgesics, their use in chronic treatments is associated with numerous side effects including the development of pain hypersensitivity and analgesic tolerance. We recently identified a novel orally active Neuropeptide FF (NPFF) receptor antagonist, RF313, which efficiently prevents the development of fentanyl-induced hyperalgesia in rats. In this study, we investigated the properties of this compound into more details. We show that RF313 exhibited a pronounced selectivity for NPFF receptors, antagonist activity at NPFF1 receptor (NPFF1R) subtype both in vitro and in vivo and no major side effects when administered in mice up to 30 mg/kg. When co-administered with opiates in rats and mice, it improved their analgesic efficacy and prevented the development of long lasting opioid-induced hyperalgesia. Moreover, and in marked contrast with the dipeptidic NPFF receptor antagonist RF9, RF313 displayed negligible affinity and no agonist activity (up to 100 muM) toward the kisspeptin receptor. Finally, in male hamster, RF313 had no effect when administered alone but fully blocked the increase in LH induced by RFRP-3, while RF9 per se induced a significant increase in LH levels which is consistent with its ability to activate kisspeptin receptors. Altogether, our data indicate that RF313 represents an interesting compound for the development of therapeutic tools aiming at improving analgesic action of opiates and reducing adverse side effects associated with their chronic administration. Moreover, its lack of agonist activity at the kisspeptin receptor indicates that RF313 might be considered a better pharmacological tool, when compared to RF9, to examine the regulatory roles of RF-amide-related peptides and NPFF1R in reproduction.
Central neuropeptide FF reduces feed consumption and affects hypothalamic chemistry in chicks.[Pubmed:17936900]
Neuropeptides. 2007 Dec;41(6):433-9.
Information on the physiological functions of Neuropeptide FF; NPFF, a morphine modulating octapeptide in avians is lacking. Thus, we designed a study to investigate the effects of central NPFF with particular emphasis on appetite-related processes. Cobb-500 chicks were intracerebroventricularly (ICV) injected with 0, 4.16, 8.32 or 16.6nmol NPFF, and feed and water intake were quantified. Feed intake was linearly decreased as NPFF dose increased, and this effect decayed over time and was not significant by 120min post-injection. Water intake was not affected by ICV NPFF. In a second exp, we observed that naloxone completely reversed the NPFF-induced decrease in feed intake. The amount of time a visible marker took to travel through the total length of the alimentary canal linearly increased as NPFF dose increased. We measured neuronal activation in the lateral hypothalamus (LH), paraventricular nucleus (PVN) dorsomedial nucleus (DMN) and ventromedial hypothalamus (VMN) of the hypothalamus, and nucleus dorsomedialis posterior thalami (DMP) of the thalamus. The DMN, DMP, PVN and VMH were all activated by ICV NPFF while the LH was not affected. Finally, we determined that the anorexigenic effect of ICV NPFF is primarily behavior specific, since behaviors unrelated to ingestion were not increased the same duration of time as was consumatory pecking. We conclude that NPFF causes anorexigenic effects in chicks that are primarily behavior specific.
Functional differences between NPFF1 and NPFF2 receptor coupling: high intrinsic activities of RFamide-related peptides on stimulation of [35S]GTPgammaS binding.[Pubmed:17011599]
Neuropharmacology. 2007 Feb;52(2):376-86.
By using an optimized [(35)S]GTPgammaS binding assay, the functional activities (potency and efficacy) of peptides belonging to three members of the RFamide family; Neuropeptide FF (NPFF), prolactin-releasing peptide (PrRP) and 26RFamide, were investigated on NPFF(1) and NPFF(2) receptors stably expressed in Chinese Hamster Ovary (CHO) cells. Despite their large differences in affinity and selectivity, all analogues tested behaved as agonists toward NPFF(1) and NPFF(2) receptors. High NaCl concentration in the assay strongly increased the efficacy toward NPFF(2) receptors and augmented differences among agonists. In low sodium conditions, whereas the potencies of agonists correlated with their affinities for NPFF(1) receptors, NPFF(2) receptors exhibited an extraordinary activity since all compounds tested displayed EC(50) values of GTPgammaS binding lower than their K(I) values. Comparisons of functional values between NPFF(1) and NPFF(2) receptors revealed unexpected potent selective NPFF(2) agonists especially for the PLRFamide and the VGRFamide sequences. By using blocker peptides, we also show that Galpha(i3) and Galpha(s) are the main transducers of NPFF(1) receptors while NPFF(2) are probably coupled with Galpha(i2), Galpha(i3), Galpha(o) and Galpha(s) proteins. Our data indicate that NPPF(1) and NPFF(2) receptors are differently coupled to G proteins in CHO cells.


