Ro 90-7501CAS# 293762-45-5 |
- BS-181
Catalog No.:BCC1439
CAS No.:1092443-52-1
- WHI-P180 hydrochloride
Catalog No.:BCC4243
CAS No.:153437-55-9
- SNS-032 (BMS-387032)
Catalog No.:BCC1152
CAS No.:345627-80-7
- Dinaciclib (SCH727965)
Catalog No.:BCC3765
CAS No.:779353-01-4
- RGB-286638
Catalog No.:BCC5519
CAS No.:784210-87-3
- AT7519 Hydrochloride
Catalog No.:BCC1376
CAS No.:902135-91-5
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 293762-45-5 | SDF | Download SDF |
| PubChem ID | 824226 | Appearance | Powder |
| Formula | C20H16N6 | M.Wt | 340.39 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 10 mM in 1eq. HCl and to 100 mM in DMSO | ||
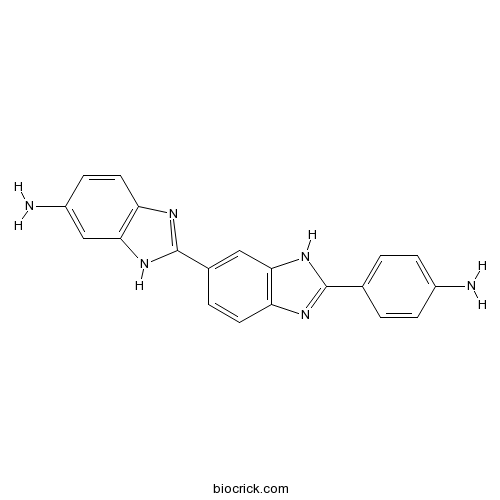
| Chemical Name | 2-[2-(4-aminophenyl)-3H-benzimidazol-5-yl]-3H-benzimidazol-5-amine | ||
| SMILES | C1=CC(=CC=C1C2=NC3=C(N2)C=C(C=C3)C4=NC5=C(N4)C=C(C=C5)N)N | ||
| Standard InChIKey | PAGZCEHLFCJSPV-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H16N6/c21-13-4-1-11(2-5-13)19-23-15-7-3-12(9-17(15)25-19)20-24-16-8-6-14(22)10-18(16)26-20/h1-10H,21-22H2,(H,23,25)(H,24,26) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Inhibitor of amyloid β42 (Aβ42) fibril assembly; reduces Aβ42-induced toxicity (EC50 = 2 μM). |

Ro 90-7501 Dilution Calculator

Ro 90-7501 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.9378 mL | 14.689 mL | 29.3781 mL | 58.7561 mL | 73.4452 mL |
| 5 mM | 0.5876 mL | 2.9378 mL | 5.8756 mL | 11.7512 mL | 14.689 mL |
| 10 mM | 0.2938 mL | 1.4689 mL | 2.9378 mL | 5.8756 mL | 7.3445 mL |
| 50 mM | 0.0588 mL | 0.2938 mL | 0.5876 mL | 1.1751 mL | 1.4689 mL |
| 100 mM | 0.0294 mL | 0.1469 mL | 0.2938 mL | 0.5876 mL | 0.7345 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Thevetiaflavone
Catalog No.:BCN4024
CAS No.:29376-68-9
- T0901317
Catalog No.:BCC1178
CAS No.:293754-55-9
- SR3335
Catalog No.:BCC1964
CAS No.:293753-05-6
- H-Phg-OH
Catalog No.:BCC3310
CAS No.:2935-35-5
- Olean-12-ene-3,11-dione
Catalog No.:BCN5195
CAS No.:2935-32-2
- Ciclopirox
Catalog No.:BCC4899
CAS No.:29342-05-0
- Tetrahydropalmatine
Catalog No.:BCN6310
CAS No.:2934-97-6
- Licarbazepine
Catalog No.:BCC7794
CAS No.:29331-92-8
- Genipin-1-O-gentiobioside
Catalog No.:BCN5349
CAS No.:29307-60-6
- Deoxyelephantopin
Catalog No.:BCN4655
CAS No.:29307-03-7
- L-685,458
Catalog No.:BCC2344
CAS No.:292632-98-5
- 25,26-Dihydroxyvitamin D3
Catalog No.:BCC4201
CAS No.:29261-12-9
- Anhydrosecoisolariciresinol
Catalog No.:BCN7521
CAS No.:29388-33-8
- Secoisolariciresinol
Catalog No.:BCN5196
CAS No.:29388-59-8
- Cyclen
Catalog No.:BCN8441
CAS No.:294-90-6
- 7-Nitroindazole
Catalog No.:BCC6713
CAS No.:2942-42-9
- 4',7-Di-O-methylnaringenin
Catalog No.:BCN5197
CAS No.:29424-96-2
- Pseudolycorine
Catalog No.:BCN5371
CAS No.:29429-03-6
- Sophorabioside
Catalog No.:BCN7838
CAS No.:2945-88-2
- Ticarcillin sodium
Catalog No.:BCC4737
CAS No.:29457-07-6
- L 006235
Catalog No.:BCC2361
CAS No.:294623-49-7
- Ganoderic acid Z
Catalog No.:BCN2440
CAS No.:294674-09-2
- Narciclasine
Catalog No.:BCN4732
CAS No.:29477-83-6
- Cypellocarpin C
Catalog No.:BCN7556
CAS No.:294856-66-9
RO 90-7501 enhances TLR3 and RLR agonist induced antiviral response.[Pubmed:23056170]
PLoS One. 2012;7(10):e42583.
Recognition of virus infection by innate pattern recognition receptors (PRRs), including membrane-associated toll-like receptors (TLR) and cytoplasmic RIG-I-like receptors (RLR), activates cascades of signal transduction pathways leading to production of type I interferons (IFN) and proinflammatory cytokines that orchestrate the elimination of the viruses. Although it has been demonstrated that PRR-mediated innate immunity plays an essential role in defending virus from infection, it also occasionally results in overwhelming production of proinflammatory cytokines that cause severe inflammation, blood vessel leakage and tissue damage. In our efforts to identify small molecules that selectively enhance PRR-mediated antiviral, but not the detrimental inflammatory response, we discovered a compound, Ro 90-7501 ('2'-(4-Aminophenyl)-[2,5'-bi-1H-benzimidazol]-5-amine), that significantly promoted both TLR3 and RLR ligand-induced IFN-beta gene expression and antiviral response, most likely via selective activation of p38 mitogen-activated protein kinase (MAPK) pathway. Our results thus imply that pharmacological modulation of PRR signal transduction pathways in favor of the induction of a beneficial antiviral response can be a novel therapeutic strategy.
Ro 90-7501 inhibits PP5 through a novel, TPR-dependent mechanism.[Pubmed:27840051]
Biochem Biophys Res Commun. 2017 Jan 8;482(2):215-220.
Protein phosphatase 5 (PP5) is a serine/threonine phosphatase that belongs to the PPP family phosphatases. PP5 and the other phosphatases of the PPP family share significantly similar catalytic domain structure. Due to this structural similarity, natural competitive inhibitors such as okadaic acid and cantharidin exhibit broad specificity over the PPP family phosphatases. In this study, we report the identification of three PP5 inhibitors, Ro 90-7501, aurothioglucose, and N-oleoyldopamine, along with a novel inhibitory mechanism of Ro 90-7501. Unlike other inhibitors binding to the phosphatase domain, Ro 90-7501 inhibited PP5 in a TPR-dependent manner. This TPR-dependent PP5 inhibition shown by Ro 90-7501 is a unique and novel inhibitory mechanism, which might be a useful tool for studies of PP5 on both regulatory mechanism and drug discovery.
Self-assembly of beta-amyloid 42 is retarded by small molecular ligands at the stage of structural intermediates.[Pubmed:10940228]
J Struct Biol. 2000 Jun;130(2-3):232-46.
Assemblyof the amyloid-beta peptide (Abeta) into fibrils and its deposition in distinct brain areas is considered responsible for the pathogenesis of Alzheimer's disease (AD). Thus, inhibition of fibril assembly is a potential strategy for therapeutic intervention. Electron cryomicroscopy was used to monitor the initial, native assembly structure of Abeta42. In addition to the known fibrillar intermediates, a nonfibrillar, polymeric sheet-like structure was identified. A temporary sequence of supramolecular structures was revealed with (i) polymeric Abeta42 sheets during the onset of assembly, inversely related to the appearance of (ii) fibril intermediates, which again are time-dependently replaced by (iii) mature fibrils. A cell-based primary screening assay was used to identify compounds that decrease Abeta42-induced toxicity. Hit compounds were further assayed for binding to Abeta42, radical scavenger activity, and their influence on the assembly structure of Abeta42. One compound, Ro 90-7501, was found to efficiently retard mature fibril formation, while extended polymeric Abeta42 sheets and fibrillar intermediates are accumulated. Ro 90-7501 may serve as a prototypic inhibitor for Abeta42 fibril formation and as a tool for studying the molecular mechanism of fibril assembly.


