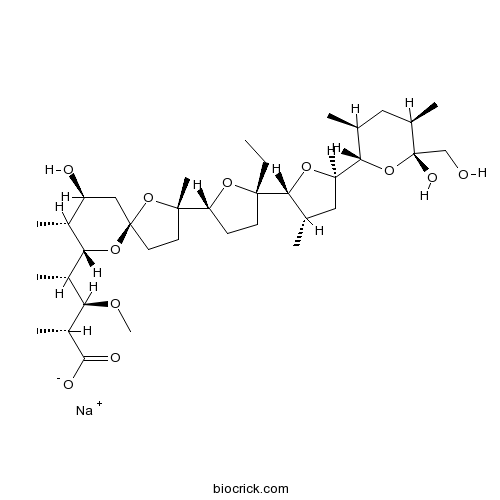
Sodium MonensinSodium ionophore; antibacterial agent CAS# 22373-78-0 |
- S0859
Catalog No.:BCC1914
CAS No.:1019331-10-2
- Nav1.7 inhibitor
Catalog No.:BCC4191
CAS No.:1355631-24-1
- Riluzole
Catalog No.:BCC3849
CAS No.:1744-22-5
- Flecainide acetate
Catalog No.:BCC1578
CAS No.:54143-56-5
- Dibucaine (Cinchocaine) HCl
Catalog No.:BCC3760
CAS No.:61-12-1
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 22373-78-0 | SDF | Download SDF |
| PubChem ID | 23667299 | Appearance | Powder |
| Formula | C36H61NaO11 | M.Wt | 692.85 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Ethanol : 25 mg/mL (36.08 mM; Need ultrasonic) DMSO : < 1 mg/mL (insoluble or slightly soluble) H2O : < 0.1 mg/mL (insoluble) | ||
| Chemical Name | sodium;(2S,3R,4S)-4-[(2S,5R,7S,8R,9S)-2-[(2R,5S)-5-ethyl-5-[(2R,3S,5R)-5-[(2S,3S,5R,6R)-6-hydroxy-6-(hydroxymethyl)-3,5-dimethyloxan-2-yl]-3-methyloxolan-2-yl]oxolan-2-yl]-7-hydroxy-2,8-dimethyl-1,10-dioxaspiro[4.5]decan-9-yl]-3-methoxy-2-methylpentanoate | ||
| SMILES | CCC1(CCC(O1)C2(CCC3(O2)CC(C(C(O3)C(C)C(C(C)C(=O)[O-])OC)C)O)C)C4C(CC(O4)C5C(CC(C(O5)(CO)O)C)C)C.[Na+] | ||
| Standard InChIKey | XOIQMTLWECTKJL-FBZUZRIGSA-M | ||
| Standard InChI | InChI=1S/C36H62O11.Na/c1-10-34(31-20(3)16-26(43-31)28-19(2)15-21(4)36(41,18-37)46-28)12-11-27(44-34)33(8)13-14-35(47-33)17-25(38)22(5)30(45-35)23(6)29(42-9)24(7)32(39)40;/h19-31,37-38,41H,10-18H2,1-9H3,(H,39,40);/q;+1/p-1/t19-,20-,21+,22+,23-,24-,25-,26+,27+,28-,29+,30-,31+,33-,34-,35+,36-;/m0./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Sodium ionophore. Neutralizes acidic intracellular compartments. Disrupts Golgi apparatus structure and inhibits vesicular transport in eukaryotic cells. Induces apoptosis in prostate cancer cells. Antiprotozoal, antibacterial and antifungal agent. |

Sodium Monensin Dilution Calculator

Sodium Monensin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.4433 mL | 7.2166 mL | 14.4331 mL | 28.8663 mL | 36.0828 mL |
| 5 mM | 0.2887 mL | 1.4433 mL | 2.8866 mL | 5.7733 mL | 7.2166 mL |
| 10 mM | 0.1443 mL | 0.7217 mL | 1.4433 mL | 2.8866 mL | 3.6083 mL |
| 50 mM | 0.0289 mL | 0.1443 mL | 0.2887 mL | 0.5773 mL | 0.7217 mL |
| 100 mM | 0.0144 mL | 0.0722 mL | 0.1443 mL | 0.2887 mL | 0.3608 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Sodium Monensin, isolated from Streptomyces cinnamonensis, is a well-known representative of naturally polyether ionophore antibiotics.
- 4-Methoxysalicylic acid
Catalog No.:BCN7783
CAS No.:2237-36-7
- Eupatilin
Catalog No.:BCN2336
CAS No.:22368-21-4
- Mirabegron (YM178)
Catalog No.:BCC3814
CAS No.:223673-61-8
- Collagen proline hydroxylase inhibitor
Catalog No.:BCC1494
CAS No.:223666-07-7
- Collagen proline hydroxylase inhibitor-1
Catalog No.:BCC1495
CAS No.:223663-32-9
- K-115 free base
Catalog No.:BCC5501
CAS No.:223645-67-8
- Tiadinil
Catalog No.:BCC8070
CAS No.:223580-51-6
- CPA inhibitor
Catalog No.:BCC1500
CAS No.:223532-02-3
- YM 58483
Catalog No.:BCC7817
CAS No.:223499-30-7
- ONO 4817
Catalog No.:BCC2375
CAS No.:223472-31-9
- GLP-2 (human)
Catalog No.:BCC5891
CAS No.:223460-79-5
- FG2216
Catalog No.:BCC6402
CAS No.:223387-75-5
- NPE-caged-HPTS
Catalog No.:BCC5950
CAS No.:223759-19-1
- Bestatin trifluoroacetate
Catalog No.:BCC3909
CAS No.:223763-80-2
- Pedalitin
Catalog No.:BCN3954
CAS No.:22384-63-0
- Serratenediol
Catalog No.:BCN5061
CAS No.:2239-24-9
- 3,8-Di-O-methylellagic acid
Catalog No.:BCN5062
CAS No.:2239-88-5
- Aristola-1(10),8-dien-2-one
Catalog No.:BCN7608
CAS No.:22391-34-0
- Cyclobuxine D
Catalog No.:BCC9221
CAS No.:2241-90-9
- 3-Epiturraeanthin
Catalog No.:BCN5063
CAS No.:22415-24-3
- Siramesine hydrochloride
Catalog No.:BCC5134
CAS No.:224177-60-0
- Incensole
Catalog No.:BCN3831
CAS No.:22419-74-5
- 2,2'-Anhydro-5-methyluridine
Catalog No.:BCC8486
CAS No.:22423-26-3
- Bayogenin methyl ester
Catalog No.:BCN3722
CAS No.:22425-81-6
Meta-analysis of the effects of laidlomycin propionate, fed alone or in combination with chlortetracycline, compared with monensin sodium, fed alone or in combination with tylosin, on growth performance, health, and carcass outcomes in finishing steers in North America.[Pubmed:27136025]
J Anim Sci. 2016 Apr;94(4):1662-76.
The objective of this research was to use data from multiple studies to comprehensively quantify the effects of feeding 1) laidlomycin propionate (LP), alone and/or in combination with chlortetracycline, compared with 2) monensin sodium (MS), alone and/or in combination with tylosin, at commercially approved dosages, on ADG, DMI, feed efficiency (FE), mortality, and carcass characteristics (HCW and liver abscesses). A secondary objective was to explore potential sources of heterogeneity among the comparative effectiveness studies. A systematic review of peer-reviewed literature and industry reports was used to identify studies that included direct comparisons of these treatments in finishing steers in North America. Random-effects meta-analysis models of performance, carcass, and health-related outcomes were fitted with extracted data, consisting of a total of 17 data sets comprising a total of 135 pens and 13,603 steers. Results showed that pens of steers fed LP had increased ADG (live and carcass adjusted), DMI, and HCW compared with those fed monensin ( < 0.05). However, liver abscesses were more common in LP-fed cattle than in MS-fed cattle ( < 0.05), presumably because of differences in the concurrently fed antimicrobials. No significant effects ( > 0.05) were identified for FE or for health-related outcomes (overall and cause-specific mortality). There was a substantial amount of heterogeneity in outcomes among studies, and when pen size and type of production setting were included in mixed-effects meta-regression models, they accounted for only a small proportion of the between-study heterogeneity found in the meta-analysis models. Therefore, caution should be exercised when interpreting summary estimates in the presence of substantial heterogeneity. However, these results provide comprehensive information on the comparative effects of different ionophores across multiple studies and multiple years, states, and production settings. These unique results can enable quantitative and informed decisions by potential end users of these feed additives that are widely used in the U.S. beef industry for reducing the costs of beef production through enhanced cattle performance.
Pelleting in Associated with Sodium Monensin Increases the Conjugated Linoleic Acids Concentration in the Milk of Dairy Cows Fed Canola Seeds.[Pubmed:26104517]
Asian-Australas J Anim Sci. 2015 Aug;28(8):1095-104.
To evaluate the effects of the pelleting and the addition of Sodium Monensin on production, the chemical and lipid composition of milk and butter physical characteristics, 4 Holstein dairy cows (135 days of lactation) with an average milk production of 14.7 kg/d, were supplemented with a concentrate containing ground canola seeds. The cows were assigned to a 4x4 Latin square design with a 2x2 factorial arrangement of treatments: i) ground maize, soybean meal, mineral and vitamin supplements, and ground canola seeds (CG); ii) CG concentrate with 31.5 mg of monensin added per kg of dry matter (DM); iii) CG pelleted concentrate; iv) CG concentrate with monensin addition pelleted. There was no difference in milk production and composition. The addition of monensin increased milk concentration of polyunsaturated fatty acids (PUFA), the PUFA/saturated fatty acids (SFA) ratio, and omega 6. The pelleting increased the concentration of monounsaturated fatty acids, the PUFA/SFA ratio, and the omega 6/omega 3 ratio, but decreased the concentration of SFA. The association between pelleting and the addition of monensin increased the concentration of conjugated linoleic acids by 46.9%. The physical characteristics of butter were not affected by the evaluated diets. We concluded that the concentrate with 31.5 mg of monensin added per kg DM basis combined with the pelleting improves the lipid composition of milk from Holstein cows that are on pasture and supplemented with ground canola seeds, without changing the production, milk composition, and spreadability of butter.
Monensin A acid complexes as a model of electrogenic transport of sodium cation.[Pubmed:22564680]
Biochim Biophys Acta. 2012 Sep;1818(9):2108-19.
New Monensin A acid complexes with water molecule, sodium chloride and sodium perchlorate were obtained and studied by X-ray and (1)H, (13)C NMR and FT-IR methods as well as ab initio calculations. The crystal structure of the complexes indicates the complexation of the water molecule and Na(+) cation in the pseudo-cycle conformation of the Monensin acid molecule stabilised by intramolecular hydrogen bonds. Important for stabilisation of this structure is also the intermolecular hydrogen bonds with water molecule or the coordination bonds with Na(+) cation. It is demonstrated that the counterions forming intermolecular hydrogen bonds with OH groups influence the strength of the intramolecular hydrogen bonds, but they have no influence on the formation of pseudo-cyclic structure. Spectroscopic studies of the complexes in dichloromethane solution have shown that the pseudo-cyclic structure of the compounds is conserved. As follows from the ab initio calculations, the interactions between the Na(+) cation and the electronegative oxygen atoms of Monensin acid totally change the molecular electrostatic potential around the supramolecular Monensin acid-Na(+) cationic complex relative to that of the neutral Monensin acid molecule.
FT-IR, 1H, 13C NMR, ESI-MS and semiempirical investigation of the structures of Monensin phenyl urethane complexes with the sodium cation.[Pubmed:23578536]
Spectrochim Acta A Mol Biomol Spectrosc. 2013 Jun;110:285-90.
In this paper three forms of phenyl urethane of Monensin i.e. its acid form (H-MU) and its 1:1 complex with NaClO4 (H-MU-Na) and its sodium salt (Na-MU) were obtained and their structures were studied by FT-IR, (1)H and (13)C NMR, ESI MS and PM5 methods. The FT-IR data of Na-MU complexes demonstrate that the C=O urethane group is not engaged in the complexation of the sodium cation. However spectroscopic studies of H-MU-Na complex show that the structure in which this C=O urethane groups participate in the complexation is also present, but it is in the minority. The PM5 semiempirical calculations allow visualisation of all structures and determination of the hydrogen bond parameters.
Structure and antimicrobial properties of monensin A and its derivatives: summary of the achievements.[Pubmed:23509771]
Biomed Res Int. 2013;2013:742149.
In this paper structural and microbiological studies on the ionophorous antibiotic monensin A and its derivatives have been collected. Monensin A is an ionophore which selectively complexes and transports sodium cation across lipid membranes, and therefore it shows a variety of biological properties. This antibiotic is commonly used as coccidiostat and nonhormonal growth promoter. The paper focuses on both the latest and earlier achievements concerning monensin A antimicrobial activity. The activities of monensin derivatives, including modifications of hydroxyl groups and carboxyl group, are also presented.
Monensin is a potent inducer of oxidative stress and inhibitor of androgen signaling leading to apoptosis in prostate cancer cells.[Pubmed:21159605]
Mol Cancer Ther. 2010 Dec;9(12):3175-85.
Current treatment options for advanced and hormone refractory prostate cancer are limited and responses to commonly used androgen pathway inhibitors are often unsatisfactory. Our recent results indicated that sodium ionophore monensin is one of the most potent and cancer-specific inhibitors in a systematic sensitivity testing of most known drugs and drug-like molecules in a panel of prostate cancer cell models. Because monensin has been extensively used in veterinary applications to build muscle mass in cattle, the link to prostate cancer and androgen signaling was particularly interesting. Here, we showed that monensin effects at nanomolar concentrations are linked to induction of apoptosis and potent reduction of androgen receptor mRNA and protein in prostate cancer cells. Monensin also elevated intracellular oxidative stress in prostate cancer cells as evidenced by increased generation of intracellular reactive oxygen species and by induction of a transcriptional profile characteristic of an oxidative stress response. Importantly, the antiproliferative effects of monensin were potentiated by combinatorial treatment with the antiandrogens and antagonized by antioxidant vitamin C. Taken together, our results suggest monensin as a potential well-tolerated, in vivo compatible drug with strong proapoptotic effects in prostate cancer cells, and synergistic effects with antiandrogens. Moreover, our data suggest a general strategy by which the effects of antiandrogens could be enhanced by combinatorial administration with agents that increase oxidative stress in prostate cancer cells.
Alteration of intracellular traffic by monensin; mechanism, specificity and relationship to toxicity.[Pubmed:2160275]
Biochim Biophys Acta. 1990 May 7;1031(2):225-46.
Monensin, a monovalent ion-selective ionophore, facilitates the transmembrane exchange of principally sodium ions for protons. The outer surface of the ionophore-ion complex is composed largely of nonpolar hydrocarbon, which imparts a high solubility to the complexes in nonpolar solvents. In biological systems, these complexes are freely soluble in the lipid components of membranes and, presumably, diffuse or shuttle through the membranes from one aqueous membrane interface to the other. The net effect for monensin is a trans-membrane exchange of sodium ions for protons. However, the interaction of an ionophore with biological membranes, and its ionophoric expression, is highly dependent on the biochemical configuration of the membrane itself. One apparent consequence of this exchange is the neutralization of acidic intracellular compartments such as the trans Golgi apparatus cisternae and associated elements, lysosomes, and certain endosomes. This is accompanied by a disruption of trans Golgi apparatus cisternae and of lysosome and acidic endosome function. At the same time, Golgi apparatus cisternae appear to swell, presumably due to osmotic uptake of water resulting from the inward movement of ions. Monensin effects on Golgi apparatus are observed in cells from a wide range of plant and animal species. The action of monensin is most often exerted on the trans half of the stacked cisternae, often near the point of exit of secretory vesicles at the trans face of the stacked cisternae, or, especially at low monensin concentrations or short exposure times, near the middle of the stacked cisternae. The effects of monensin are quite rapid in both animal and plant cells; i.e., changes in Golgi apparatus may be observed after only 2-5 min of exposure. It is implicit in these observations that the uptake of osmotically active cations is accompanied by a concomitant efflux of H+ and that a net influx of protons would be required to sustain the ionic exchange long enough to account for the swelling of cisternae observed in electron micrographs. In the Golgi apparatus, late processing events such as terminal glycosylation and proteolytic cleavages are most susceptible to inhibition by monensin. Yet, many incompletely processed molecules may still be secreted via yet poorly understood mechanisms that appear to bypass the Golgi apparatus. In endocytosis, monensin does not prevent internalization. However, intracellular degradation of internalized ligands may be prevented.(ABSTRACT TRUNCATED AT 400 WORDS)


