Bestatin trifluoroacetateCAS# 223763-80-2 |
- 3,3'-Diindolylmethane
Catalog No.:BCC1306
CAS No.:1968-05-4
- BAM7
Catalog No.:BCC1397
CAS No.:331244-89-4
- Capsaicin
Catalog No.:BCN1016
CAS No.:404-86-4
- Betulinic acid
Catalog No.:BCN5524
CAS No.:472-15-1
- Brassinolide
Catalog No.:BCC1438
CAS No.:72962-43-7
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 223763-80-2 | SDF | Download SDF |
| PubChem ID | 78358331 | Appearance | Powder |
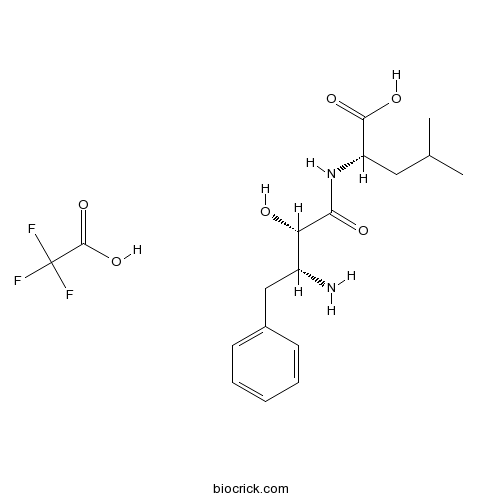
| Formula | C18H25F3N2O6 | M.Wt | 422.4 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Synonyms | Ubenimex trifluoroacetate | ||
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (2S)-2-[[(2S,3R)-3-amino-2-hydroxy-4-phenylbutanoyl]amino]-4-methylpentanoic acid;2,2,2-trifluoroacetic acid | ||
| SMILES | CC(C)CC(C(=O)O)NC(=O)C(C(CC1=CC=CC=C1)N)O.C(=O)(C(F)(F)F)O | ||
| Standard InChIKey | UOALAMWBTXFYPB-UDYGKFQRSA-N | ||
| Standard InChI | InChI=1S/C16H24N2O4.C2HF3O2/c1-10(2)8-13(16(21)22)18-15(20)14(19)12(17)9-11-6-4-3-5-7-11;3-2(4,5)1(6)7/h3-7,10,12-14,19H,8-9,17H2,1-2H3,(H,18,20)(H,21,22);(H,6,7)/t12-,13+,14+;/m1./s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Bestatin trifluoroacetate is an inhibitor of CD13 (Aminopeptidase N)/APN and leukotriene A4 hydrolase, used for cancer treatment.In Vitro:Bestatin enhances ATRA-induced differentiation and inhibits ATRA-driven phosphorylation of p38 MAPK in ATRA-sensitive APL NB4 cells. Bestatin can not reverse the differentiation block in ATRA-resistant APL MR2 cells. CD13 ligation with anti-CD13 antibody WM-15 results in phosphorylation of p38 MAPK, reduces the inhibition of Bestatin on the phosphorylation of p38 MAPK, and completely abolishes the enhancement of Bestatin on ATRA-inducing differentiation in NB4 cells[2]. Bestatin (600 μM)-treated cells progress slower through the cell cycle due to decreased rate of cell growth and the frequency of cell division. Bestatin inhibits the frequency of mitosis and the inherent multinuclearity in D. discoideum, and is not cytotoxic to D. discoideum cells at 0-600 μM. Bestatin inhibits aminopeptidase activity in lysates of PsaA-GFP- and GFP-expressing cells by 69.39% ± 10.5% and 39.93% ± 18.7% of control, respectively[4].In Vivo:Bestatin (20 μM) significantly reduces CD13 expression in diabetic mice and results a significant inhibition of MMP-9 specific gelationolytic band densities compared to diabetic vehicle-treated mice. Bestatin treatment significantly inhibits the expression of VEGF and heparanase in diabetic mice. Intravitreal bestatin treatment significantly downregulates the expression of both HIF-1α and VEGF in diabetic mice retinas. Furthermore, the upregulated expression of heparanase in diabetic mice retinas is significantly inhibited by intravitreal bestatin treatment[1]. Bestatin (10, 1, and 0.1mg/kg, i.p.) treatment before the antigen-potentiated humoral response to SRBC results in an increased number of splenocytes producing hemolytic anti-SRBC antibodies (PFC) and the 2-ME-resistant serum hemagglutinin titer (at a dose of 0.1 mg/kg). Bestatin (1 and 0.1 mg/kg) administered to mice five times on alternate days after cyclophosphamide injection does not change the suppressive effect of the drug regarding the number of PFC, and even causes the further decrease of the total anti-SRBC hemagglutinins at dose of 1 mg/kg on day 7 after antigen stimulation[3]. References: | |||||

Bestatin trifluoroacetate Dilution Calculator

Bestatin trifluoroacetate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.3674 mL | 11.8371 mL | 23.6742 mL | 47.3485 mL | 59.1856 mL |
| 5 mM | 0.4735 mL | 2.3674 mL | 4.7348 mL | 9.4697 mL | 11.8371 mL |
| 10 mM | 0.2367 mL | 1.1837 mL | 2.3674 mL | 4.7348 mL | 5.9186 mL |
| 50 mM | 0.0473 mL | 0.2367 mL | 0.4735 mL | 0.947 mL | 1.1837 mL |
| 100 mM | 0.0237 mL | 0.1184 mL | 0.2367 mL | 0.4735 mL | 0.5919 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Bestatin trifluoroacetate (Ubenimex ) is a competitive aminopeptidase inhibitor, which is studied for use in the treatment of acute myelocytic leukemia.
- NPE-caged-HPTS
Catalog No.:BCC5950
CAS No.:223759-19-1
- Sodium Monensin
Catalog No.:BCC5319
CAS No.:22373-78-0
- 4-Methoxysalicylic acid
Catalog No.:BCN7783
CAS No.:2237-36-7
- Eupatilin
Catalog No.:BCN2336
CAS No.:22368-21-4
- Mirabegron (YM178)
Catalog No.:BCC3814
CAS No.:223673-61-8
- Collagen proline hydroxylase inhibitor
Catalog No.:BCC1494
CAS No.:223666-07-7
- Collagen proline hydroxylase inhibitor-1
Catalog No.:BCC1495
CAS No.:223663-32-9
- K-115 free base
Catalog No.:BCC5501
CAS No.:223645-67-8
- Tiadinil
Catalog No.:BCC8070
CAS No.:223580-51-6
- CPA inhibitor
Catalog No.:BCC1500
CAS No.:223532-02-3
- YM 58483
Catalog No.:BCC7817
CAS No.:223499-30-7
- ONO 4817
Catalog No.:BCC2375
CAS No.:223472-31-9
- Pedalitin
Catalog No.:BCN3954
CAS No.:22384-63-0
- Serratenediol
Catalog No.:BCN5061
CAS No.:2239-24-9
- 3,8-Di-O-methylellagic acid
Catalog No.:BCN5062
CAS No.:2239-88-5
- Aristola-1(10),8-dien-2-one
Catalog No.:BCN7608
CAS No.:22391-34-0
- Cyclobuxine D
Catalog No.:BCC9221
CAS No.:2241-90-9
- 3-Epiturraeanthin
Catalog No.:BCN5063
CAS No.:22415-24-3
- Siramesine hydrochloride
Catalog No.:BCC5134
CAS No.:224177-60-0
- Incensole
Catalog No.:BCN3831
CAS No.:22419-74-5
- 2,2'-Anhydro-5-methyluridine
Catalog No.:BCC8486
CAS No.:22423-26-3
- Bayogenin methyl ester
Catalog No.:BCN3722
CAS No.:22425-81-6
- Ginsenoside Rg1
Catalog No.:BCN1066
CAS No.:22427-39-0
- Retapamulin
Catalog No.:BCC4837
CAS No.:224452-66-8
Effect of protease inhibitors on pulmonary bioavailability of therapeutic proteins and peptides in the rat.[Pubmed:25460544]
Eur J Pharm Sci. 2015 Feb 20;68:1-10.
The objective of the present study was to evaluate the effect of protease inhibitors on the pulmonary absorption of therapeutic peptides and proteins with varying molecular weights. Dry powder formulations of leuprolide (1.2 kD), salmon calcitonin (3.4 kD), human insulin (5.8 kD), human leptin (16.0 kD), and human chorionic gonadotropin (HCG) (36.5 kD) were prepared with or without protease inhibitors; aprotinin and bestatin. The formulations were administered intrapulmonary to anesthetized rats. The pharmacokinetics of these proteins were assessed by measuring serum drug concentrations. In addition, in vitro stability of these proteins in rat lung homogenate was assessed using the trifluoroacetic acid method. Bioavailability of leuprolide following pulmonary administration was 75% higher compared to subcutaneously administered leuprolide. Protease inhibitors had little or no effect on the pulmonary bioavailability of leuprolide. However, protease inhibitors (1 mg/kg) increased the bioavailability of calcitonin by more than 50%. Similarly, the bioavailabilities of leptin and HCG in the presence of bestatin were increased by 1.9 and 2.1-fold, respectively. Leuprolide was stable both in the lung cytosol and subcellular pellets with about 10% degradation at the end of the study period (4h). In contrast, calcitonin, insulin, leptin and HCG were significantly degraded in the lung cytosol and subcellular pellets. Presence of protease inhibitors in formulation could improve the stability of protein drugs. The results of this study demonstrate that the pulmonary absorption of proteins may be enhanced by the selection of optimal concentration and type of protease inhibitor.
Effect of protease inhibitors on degradation of recombinant human epidermal growth factor in skin tissue.[Pubmed:18975209]
Arch Pharm Res. 1997 Feb;20(1):34-8.
Recombinant human epidermal growth factor (rhEGF), a polypeptide of 53 amino acid residues, is subject to degradation by numerous enzymes, especially proteases, when it is applied on the skin for the treatment of open wound. Amastatin, aprotinin, bestatin, EDTA, EGTA, gabexate, gentamicin, leupeptin, and TPCK were investigated for the possible protease inhibitors, which may use to protect rhEGF from degradation by the enzymes in the skin. Skin homogenates containing protease inhibitors and rhEGF were incubated at 37 degrees C for 30 minutes. After the reaction was stopped with trifluoroacetic acid, the amount of rhEGF remaining in the sample was determined with an HPLC method. The percentages of rhEGF degraded, at the skin/PBS ratio of 0.25, in the mouse, rat, and human skin homogenate were 85%, 70%, and 46%, respectively. The degree of degradation of rhEGF in the cytosolic fraction was higher than that in the membrane fraction and these enzyme reactions were completed in 30 minutes. Bestatin, EGTA, and TPCK showed significant inhibitory effects on the degradation of rhEGF in the two fractions (p<0.05), while the other protease inhibitors had no significant inhibitory effects or, even resulted in deleterious effects. Therefore, the formulation containing one or several inhibitors among these effective inhibitors would be a promising topical preparation of rhEGF for the treatment of open wound.


