YM 750ACAT inhibitor CAS# 138046-43-2 |
- YM155
Catalog No.:BCC2251
CAS No.:781661-94-7
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 138046-43-2 | SDF | Download SDF |
| PubChem ID | 9911716 | Appearance | Powder |
| Formula | C31H36N2O | M.Wt | 452.63 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in DMSO and to 10 mM in ethanol | ||
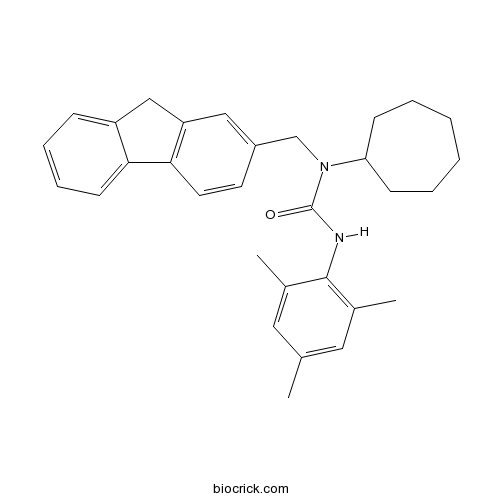
| Chemical Name | 1-cycloheptyl-1-(9H-fluoren-2-ylmethyl)-3-(2,4,6-trimethylphenyl)urea | ||
| SMILES | CC1=CC(=C(C(=C1)C)NC(=O)N(CC2=CC3=C(C=C2)C4=CC=CC=C4C3)C5CCCCCC5)C | ||
| Standard InChIKey | FMLJREWZCZHGGW-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C31H36N2O/c1-21-16-22(2)30(23(3)17-21)32-31(34)33(27-11-6-4-5-7-12-27)20-24-14-15-29-26(18-24)19-25-10-8-9-13-28(25)29/h8-10,13-18,27H,4-7,11-12,19-20H2,1-3H3,(H,32,34) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Acyl-CoA:cholesterol acyltransferase (ACAT) inhibitor (IC50 = 0.18 μM). Exhibits hypocholesterolaemic and antiatherosclerotic activity in vivo. |

YM 750 Dilution Calculator

YM 750 Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.2093 mL | 11.0466 mL | 22.0931 mL | 44.1862 mL | 55.2328 mL |
| 5 mM | 0.4419 mL | 2.2093 mL | 4.4186 mL | 8.8372 mL | 11.0466 mL |
| 10 mM | 0.2209 mL | 1.1047 mL | 2.2093 mL | 4.4186 mL | 5.5233 mL |
| 50 mM | 0.0442 mL | 0.2209 mL | 0.4419 mL | 0.8837 mL | 1.1047 mL |
| 100 mM | 0.0221 mL | 0.1105 mL | 0.2209 mL | 0.4419 mL | 0.5523 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- EHop-016
Catalog No.:BCC5022
CAS No.:1380432-32-5
- KML 29
Catalog No.:BCC6312
CAS No.:1380424-42-9
- Valeriandoid B
Catalog No.:BCN6754
CAS No.:1380399-57-4
- EPZ004777 HCl
Catalog No.:BCC4550
CAS No.:1380316-03-9
- EPZ5676
Catalog No.:BCC2215
CAS No.:1380288-87-8
- KB SRC 4
Catalog No.:BCC6253
CAS No.:1380088-03-8
- BET bromodomain inhibitor
Catalog No.:BCC6426
CAS No.:1380087-89-7
- Decorticasine
Catalog No.:BCN2006
CAS No.:1380-03-6
- Limonene
Catalog No.:BCN3797
CAS No.:138-86-3
- Shikimic acid
Catalog No.:BCN6200
CAS No.:138-59-0
- Picrocrocine
Catalog No.:BCC8232
CAS No.:138-55-6
- D-(-)-Salicin
Catalog No.:BCN6298
CAS No.:138-52-3
- 2-(4-Hydroxy-2-oxoindolin-3-yl)acetonitrile
Catalog No.:BCN1575
CAS No.:1380540-77-1
- LDK378 dihydrochloride
Catalog No.:BCC1694
CAS No.:1380575-43-8
- G007-LK
Catalog No.:BCC6383
CAS No.:1380672-07-0
- Acetylanonamine
Catalog No.:BCN2140
CAS No.:138079-62-6
- (R,R)-THC
Catalog No.:BCC7224
CAS No.:138090-06-9
- Agomelatine
Catalog No.:BCN2165
CAS No.:138112-76-2
- 7-Methoxy-1-naphthylacetonitrile
Catalog No.:BCN2242
CAS No.:138113-08-3
- H-β-HoPhe-OH
Catalog No.:BCC3240
CAS No.:138165-77-2
- (RS)-(Tetrazol-5-yl)glycine
Catalog No.:BCC6599
CAS No.:138199-51-6
- ML 289
Catalog No.:BCC6343
CAS No.:1382481-79-9
- Stearyl glycyrrhetinate
Catalog No.:BCN8486
CAS No.:13832-70-7
- Hedycoronen A
Catalog No.:BCN7653
CAS No.:1383441-73-3
Effects of an anti-oxidative ACAT inhibitor on apoptosis/necrosis and cholesterol accumulation under oxidative stress in THP-1 cell-derived foam cells.[Pubmed:18037448]
Life Sci. 2008 Jan 2;82(1-2):79-84.
THP-1 cell-derived foam cells were exposed to oxidative stress through combined treatment with acetylated LDL (acLDL) and copper ions (Cu2+). The foam cells showed caspase-dependent apoptotic changes on exposure to oxidative stress for 6 h, and necrotic changes with the leakage of LDH after 24 h. KY-455, an anti-oxidative ACAT inhibitor, and ascorbic acid (VC) but not YM-750, an ACAT inhibitor, prevented apoptotic and necrotic changes. These preventive effects of KY-455 and VC were accompanied by the inhibition of lipid peroxidation in culture medium containing acLDL and Cu2+, suggesting the involvement of oxidized acLDL in apoptosis and necrosis. Foam cells accumulated esterified cholesterol (EC) for 24 h in the presence of acLDL without Cu2+, which was suppressed by KY-455 and YM-750. Foam cells showed necrotic changes and died in the presence of acLDL and Cu2+. KY-455 but not YM-750 prevented cell death and reduced the amount of EC accumulated. The foam cells treated with VC further accumulated EC without necrotic changes for 24 h even in the presence of acLDL and Cu2+. YM-750 as well as KY-455 inhibited lipid accumulation when co-incubated with VC in foam cells exposed to oxidative stress. It is concluded that an anti-oxidative ACAT inhibitor or the combination of an antioxidant and an ACAT inhibitor protects foam cells from oxidative stress and effectively reduces cholesterol levels, which would be a promising approach in anti-atherosclerotic therapy.
Bioavailable acyl-CoA: cholesterol acyltransferase inhibitor with anti-peroxidative activity: synthesis and biological activity of novel indolinyl amide and urea derivatives.[Pubmed:10866142]
Chem Pharm Bull (Tokyo). 2000 Jun;48(6):817-27.
We synthesized a series of indoline derivatives with an amide or urea moiety and examined their inhibitory effects on acyl-CoA:cholesterol acyltransferase (ACAT) activity, lipid-peroxidation and serum cholesterol levels in experimental animals. Among the derivatives synthesized, a series of N-(1-alkyl-4,6-dimethylindolin-7-yl)-2,2-dimethylpropanamides++ + potently inhibited rabbit intestinal ACAT activity and lipid-peroxidation of rat brain homogenate. The effect on ACAT activity was related to the length of the alkyl chain at the 1-position of indoline. N-(4,6-Dimethyl-1-octylindolindolin-7-yl)-2,2-dimethylpropanami de hydrochloride (55) showed inhibitory effects on intestinal and hepatic ACAT activity slightly weaker than those of YM-750, and an inhibitory effect on low density lipoprotein (LDL)-peroxidation similar to that of probucol. Compound 55 also reduced serum cholesterol at 10 mg/kg/d in hyperlipidemic rats and 20 mg/kg/d in normolipidemic hamsters. The plasma concentration of 55 reached 716 ng/ml in dogs (10 mg/kg, p.o.), which is an effective concentration against hepatic ACAT activity and LDL-peroxidation. In conclusion, compound 55 is a novel bioavailable ACAT inhibitor with anti-peroxidative activity and is thus a promising anti-atherosclerotic and anti-hyperlipidemic drug. Indoline proved to be a useful pharmacophore for molecular design of new anti-peroxidative drugs.
N-[2-[N'-pentyl-(6,6-dimethyl-2,4-heptadiynyl)amino]ethyl]- (2-methyl-1-naphthylthio)acetamide (FY-087). A new acyl coenzyme a:cholesterol acyltransferase (ACAT) inhibitor of diet-induced atherosclerosis formation in mice.[Pubmed:7887979]
Biochem Pharmacol. 1995 Mar 1;49(5):643-51.
FY-087 (N-[2-[N'-pentyl-(6,6-dimethyl-2,4-heptadiynyl)amino]ethyl]- (2-methyl-1-naphthylthio)acetamide) was found to be a competitive inhibitor of human microsomal acyl coenzyme A:cholesterol acyltransferase (ACAT) with an IC50 value of 0.11 microM. Under our assay conditions, other ACAT inhibitors tested, specifically YM-750, E-5324, and melinamide, all of which are now in phase I clinical trials or in clinical use in Japan, inhibited this enzyme with IC50 values of 0.18, 0.14, and 3.2 microM, respectively. FY-087 also inhibited ACAT in acetyl-low density lipoprotein loaded human macrophages (THP-1 cells) with an IC50 of 0.17 microM. Following the oral administration of FY-087 (30 mg/kg) to rats, the plasma concentration of FY-087 reached 0.42 microgram/mL after 2 hr. This concentration of FY-087 was enough to inhibit blood vessel ACAT activity. Cholesterol-lowering and anti-atherogenic effects of FY-087 were examined using C57BL/6J mice fed an atherogenic diet. In this mouse model, treatment with FY-087 (28 mg/kg) inhibited the increase in plasma cholesterol levels by about 20% and decreased the hepatic accumulation of free and esterified cholesterol by 61 and 67%, respectively. FY-087 also significantly inhibited the atherogenic diet-induced increase in the fatty-streak lesion area of the proximal aorta by 57% in C57BL/6J mice. These results indicate that FY-087 is not only a therapeutically bioavailable ACAT inhibitor that lowers plasma cholesterol levels, but also an effective anti-atherogenic agent in mice fed an atherogenic diet.


