(R,R)-THCCAS# 138090-06-9 |
- Salmefamol
Catalog No.:BCC1919
CAS No.:18910-65-1
- Guanfacine hydrochloride
Catalog No.:BCC1609
CAS No.:29110-48-3
- Doxazosin Mesylate
Catalog No.:BCC1257
CAS No.:77883-43-3
- Medetomidine
Catalog No.:BCC1736
CAS No.:86347-14-0
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 138090-06-9 | SDF | Download SDF |
| PubChem ID | 446849 | Appearance | Powder |
| Formula | C22H24O2 | M.Wt | 320.42 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble to 100 mM in DMSO and to 100 mM in ethanol | ||
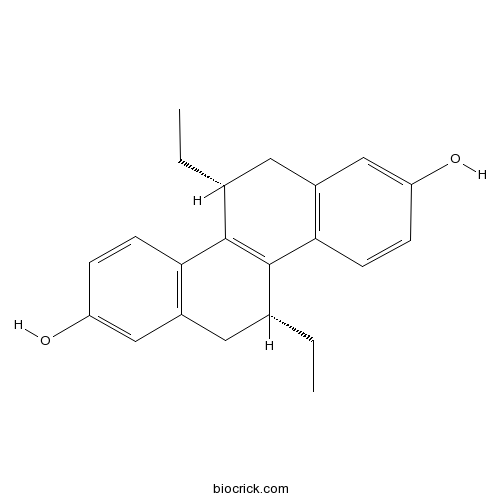
| Chemical Name | (5R,11R)-5,11-diethyl-5,6,11,12-tetrahydrochrysene-2,8-diol | ||
| SMILES | CCC1CC2=C(C=CC(=C2)O)C3=C1C4=C(CC3CC)C=C(C=C4)O | ||
| Standard InChIKey | MASYAWHPJCQLSW-ZIAGYGMSSA-N | ||
| Standard InChI | InChI=1S/C22H24O2/c1-3-13-9-15-11-17(23)6-8-20(15)22-14(4-2)10-16-12-18(24)5-7-19(16)21(13)22/h5-8,11-14,23-24H,3-4,9-10H2,1-2H3/t13-,14-/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Non-steroidal, selective estrogen receptor ligand; agonist at ERα receptor (Ki = 9.0 nM) and antagonist at ERβ receptor (Ki = 3.6 nM). |

(R,R)-THC Dilution Calculator

(R,R)-THC Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.1209 mL | 15.6045 mL | 31.209 mL | 62.4181 mL | 78.0226 mL |
| 5 mM | 0.6242 mL | 3.1209 mL | 6.2418 mL | 12.4836 mL | 15.6045 mL |
| 10 mM | 0.3121 mL | 1.5605 mL | 3.1209 mL | 6.2418 mL | 7.8023 mL |
| 50 mM | 0.0624 mL | 0.3121 mL | 0.6242 mL | 1.2484 mL | 1.5605 mL |
| 100 mM | 0.0312 mL | 0.156 mL | 0.3121 mL | 0.6242 mL | 0.7802 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Acetylanonamine
Catalog No.:BCN2140
CAS No.:138079-62-6
- G007-LK
Catalog No.:BCC6383
CAS No.:1380672-07-0
- LDK378 dihydrochloride
Catalog No.:BCC1694
CAS No.:1380575-43-8
- 2-(4-Hydroxy-2-oxoindolin-3-yl)acetonitrile
Catalog No.:BCN1575
CAS No.:1380540-77-1
- YM 750
Catalog No.:BCC7542
CAS No.:138046-43-2
- EHop-016
Catalog No.:BCC5022
CAS No.:1380432-32-5
- KML 29
Catalog No.:BCC6312
CAS No.:1380424-42-9
- Valeriandoid B
Catalog No.:BCN6754
CAS No.:1380399-57-4
- EPZ004777 HCl
Catalog No.:BCC4550
CAS No.:1380316-03-9
- EPZ5676
Catalog No.:BCC2215
CAS No.:1380288-87-8
- KB SRC 4
Catalog No.:BCC6253
CAS No.:1380088-03-8
- BET bromodomain inhibitor
Catalog No.:BCC6426
CAS No.:1380087-89-7
- Agomelatine
Catalog No.:BCN2165
CAS No.:138112-76-2
- 7-Methoxy-1-naphthylacetonitrile
Catalog No.:BCN2242
CAS No.:138113-08-3
- H-β-HoPhe-OH
Catalog No.:BCC3240
CAS No.:138165-77-2
- (RS)-(Tetrazol-5-yl)glycine
Catalog No.:BCC6599
CAS No.:138199-51-6
- ML 289
Catalog No.:BCC6343
CAS No.:1382481-79-9
- Stearyl glycyrrhetinate
Catalog No.:BCN8486
CAS No.:13832-70-7
- Hedycoronen A
Catalog No.:BCN7653
CAS No.:1383441-73-3
- CYM 9484
Catalog No.:BCC6238
CAS No.:1383478-94-1
- BD 1008 dihydrobromide
Catalog No.:BCC6674
CAS No.:138356-09-9
- BD 1047 dihydrobromide
Catalog No.:BCC6863
CAS No.:138356-21-5
- Alvimopan monohydrate
Catalog No.:BCC1349
CAS No.:1383577-62-5
- Boc-Arg(Tos)-OH
Catalog No.:BCC3067
CAS No.:13836-37-8
An observational postmarketing safety registry of patients in the UK, Germany, and Switzerland who have been prescribed Sativex((R)) (THC:CBD, nabiximols) oromucosal spray.[Pubmed:27956834]
Ther Clin Risk Manag. 2016 Nov 11;12:1667-1675.
The global exposure of Sativex((R)) (Delta(9)-tetrahydrocannabinol [THC]:cannabidiol [CBD], nabiximols) is estimated to be above 45,000 patient-years since it was given marketing approval for treating treatment-resistant spasticity in multiple sclerosis (MS). An observational registry to collect safety data from patients receiving THC:CBD was set up following its approval in the UK, Germany, and Switzerland, with the aim of determining its long-term safety in clinical practice. Twice a year, the Registry was opened to prescribing physicians to voluntarily report data on patients' use of THC:CBD, clinically significant adverse events (AEs), and special interest events. The Registry contains data from 941 patients with 2,213.98 patient-years of exposure. Within this cohort, 60% were reported as continuing treatment, while 83% were reported as benefiting from the treatment. Thirty-two percent of patients stopped treatment, with approximately one third citing lack of effectiveness and one quarter citing AEs. Psychiatric AEs of clinical significance were reported in 6% of the patients, 6% reported falls requiring medical attention, and suicidality was reported in 2%. Driving ability was reported to have worsened in 2% of patients, but improved in 7%. AEs were more common during the first month of treatment. The most common treatment-related AEs included dizziness (2.3%) and fatigue (1.7%). There were no signals to indicate abuse, diversion, or dependence. The long-term risk profile from the Registry is consistent with the known (labeled) safety profile of THC:CBD, and therefore supports it being a well-tolerated and beneficial medication for the treatment of MS spasticity. No evidence of new long-term safety concerns has emerged.
Nabiximols (THC/CBD oromucosal spray, Sativex(R)) in clinical practice--results of a multicenter, non-interventional study (MOVE 2) in patients with multiple sclerosis spasticity.[Pubmed:24525548]
Eur Neurol. 2014;71(5-6):271-9.
BACKGROUND: Nabiximols (Sativex(R)), a cannabinoid-based oromucosal spray, is an add-on therapy for patients with moderate to severe multiple sclerosis spasticity (MSS) resistant to other medications. The primary objective was to provide real-life observational data of clinical experience of nabiximols in contrast to formal clinical trials of effectiveness. METHODS: This was an observational, prospective, multicenter, non-interventional study with a follow-up period of 3-4 months, conducted in routine care setting in Germany. Patients with moderate to severe MSS were included at nabiximols' initiation. Structured documentation forms, questionnaires and validated instruments were used for data collection at inclusion, 1 and 3 months after inclusion. RESULTS: Overall, 335 patients were assessed of whom 276 fitted the criteria and were included in the effectiveness analysis. After 1 month, nabiximols provided relief of resistant MSS in 74.6% of patients according to specialist assessment; mean spasticity 0-10 numerical rating scale (NRS) score decreased from 6.1 +/- 1.8 to 5.2 +/- 2.0 points; in patients with NRS improvement >/=20% mean NRS score decreased by 40%. After 3 months, 55.3% of patients had continued to use nabiximols and the mean NRS score had decreased by 25% from baseline. 17% of patients reported adverse events. CONCLUSION: Real-life data confirm nabiximols as an effective and well-tolerated treatment option for resistant MSS in clinical practice.
The detection of THC, CBD and CBN in the oral fluid of Sativex(R) patients using two on-site screening tests and LC-MS/MS.[Pubmed:24699310]
Forensic Sci Int. 2014 May;238:113-9.
Sativex((R)) is an oromucosal spray used to treat spasticity in multiple sclerosis sufferers in some European countries, the United Kingdom, Canada and New Zealand. The drug has also recently been registered by the Therapeutic Goods Administration (TGA) in Australia for treatment of multiple sclerosis. Sativex((R)) contains high concentrations of Delta(9)-tetrahydrocannabinol (THC) and cannabidiol (CBD), with the former being the subject of random roadside drug tests across Australia to detect cannabis use. This pilot study aims to determine whether or not patients taking Sativex((R)) will test positive to THC using these roadside screening tests. Detectable levels of THC, CBD and cannabinol (CBN) in their oral fluid were also confirmed by liquid chromatography-tandem mass spectrometry (LC-MS/MS). The study was a double-blind, placebo controlled design. Oral fluid was tested prior to and immediately after dosing with either Sativex((R)) or placebo at intervals up to 2h after the dose. Two Sativex((R)) doses were studied. The low dose contained 5.4mg THC, the high dose 21.6mg THC. Results indicate that the primary screening test used in Australian roadside drug testing, the DrugWipe((R)) II Twin, often gave a false negative response for THC, even with high concentrations present. However, secondary screening test, Cozart((R)) DDS (used by police after a DrugWipe test gives a positive result), gave true positive results in all cases where patients were being treated with Sativex((R)). Confirmatory testing showed high concentrations of THC and CBD (>5356ng/mL THC and >3826ng/mL CBD) in the oral fluid shortly after dosing and also elevated concentrations of CBN. Levels dropped quickly but remained at detectable concentrations (>67.6ng/mL) two hours after drug administration. The average concentration ratio of THC/CBD across all positive samples was 1.10 (%RSD 19.9) reflecting the composition of the Sativex((R)) spray. In conclusion, Sativex((R)) users may test positive for THC by roadside drug testing within 2-3h of use. Confirmatory analysis can identify Sativex((R)) treatment through use of THC/CBD ratios, however, these ratios would unlikely be sufficient to differentiate non-medicinal cannabis use from Sativex((R)) use if both are taken concurrently.
THC and CBD oromucosal spray (Sativex(R)) in the management of spasticity associated with multiple sclerosis.[Pubmed:21456949]
Expert Rev Neurother. 2011 May;11(5):627-37.
People with multiple sclerosis may present with a wide range of disease symptoms during the evolution of the disease; among these, spasticity can have a marked impact on their well-being and quality of life. Symptom control, including spasticity, remains a key management strategy to improve the patient's well-being and functional status. However, available drug therapies for spasticity sometimes have limited benefit and they are often associated with poor tolerability. Sativex is a 1:1 mix of 9-delta-tetrahydrocannabinol and cannabidiol extracted from cloned Cannabis sativa chemovars, which is available as an oromucosal spray. Clinical experience with Sativex in patients with multiple sclerosis is accumulating steadily. Results from randomized, controlled trials have reported a reduction in the severity of symptoms associated with spasticity, leading to a better ability to perform daily activities and an improved perception of patients and their carers regarding functional status when Sativex was added to the current treatment regimen. Adverse events such as dizziness, diarrhea, fatigue, nausea, headache and somnolence occur quite frequently with Sativex, but they are generally of mild-to-moderate intensity and their incidence can be markedly reduced by gradual 'uptitration'. In summary, initial well-controlled studies with Sativex oromucosal spray administered as an add-on to usual therapy have produced promising results and highlight encouraging avenues for future research.
Conformational changes and coactivator recruitment by novel ligands for estrogen receptor-alpha and estrogen receptor-beta: correlations with biological character and distinct differences among SRC coactivator family members.[Pubmed:11014206]
Endocrinology. 2000 Oct;141(10):3534-45.
Ligands for the estrogen receptor (ER) that have the capacity to selectively bind to or activate the ER subtypes ERalpha or ERbeta would be useful in elucidating the biology of these two receptors and might assist in the development of estrogen pharmaceuticals with improved tissue selectivity. In this study, we examine three compounds of novel structure that act as ER subtype-selective ligands. These are a propyl pyrazole triol (PPT), which is a potent agonist on ERalpha but is inactive on ERbeta, and a pair of substituted tetrahydrochrysenes (THC), one enantiomer of which (S,S-THC) is an agonist on both ERalpha and ERbeta, the other (R,R-THC) being an agonist on ERalpha but an antagonist on ERbeta. To investigate the molecular mechanisms underlying the ER subtype-selective actions of these compounds, we have determined the conformational changes induced in ERalpha and ERbeta by these ligands using protease digestion sensitivity, and we have tested the ability of these ligands to promote the recruitment of representatives of the three SRC/p160 coactivator protein family members (SRC-1, GRIP-1, ACTR, respectively) to ERalpha and ERbeta using yeast two-hybrid and glutathione-S-transferase (GST) pull-down assays. We find that the ligand-ER protease digestion pattern is distinctly different for stimulatory and inhibitory ligands, and that this assay, as well as coactivator recruitment, are excellent indicators of their agonist/antagonist character. Interestingly however, compared with estradiol, the novel agonist ligands show some quantitative differences in their ability to recruit SRC-1, -2, and -3. This implies that while generally similar to estradiol, these ligands induce ER conformations that differ somewhat from that induced by estradiol, differences that are illustrative of the nature of their biological character. The application of methods to characterize the conformations induced in ER subtypes by novel ligands, as done in this study, enables a greater understanding of how ligand-receptor conformations relate to estrogen agonist or antagonist behavior.
Novel ligands that function as selective estrogens or antiestrogens for estrogen receptor-alpha or estrogen receptor-beta.[Pubmed:9927308]
Endocrinology. 1999 Feb;140(2):800-4.
We report on the identification of novel, nonsteroidal ligands that show pronounced subtype-selective differences in ligand binding and transcriptional potency or efficacy for the two estrogen receptor (ER) subtypes, ER alpha and ER beta. An aryl-substituted pyrazole is an ER alpha potency-selective agonist, showing higher binding affinity for ER alpha and 120-fold higher potency in stimulation of ER alpha vs. ER beta in transactivation assays in cells. A tetrahydrochrysene (THC) has a 4-fold preferential binding affinity for ER beta; it is an agonist on ER alpha, but a complete antagonist on ER beta. Intriguingly, the antagonist activity of THC is associated with the R,R-enantiomer (R,R-THC). The S,S-enantiomer (S,S-THC) is an agonist on both ER alpha and ER beta but has a 20-fold lower affinity for ER beta than R,R-THC. This difference in binding affinity accounts for the full ER beta antagonist activity of the THC racemate (a 1:1 mixture of R,R-THC and S,S-THC). These compounds should be useful in probing the conformational changes in these two ERs that are evoked by agonists and antagonists, and in evaluating the distinct roles that ER beta and ER alpha may play in the diverse target tissues in which estrogens act.


