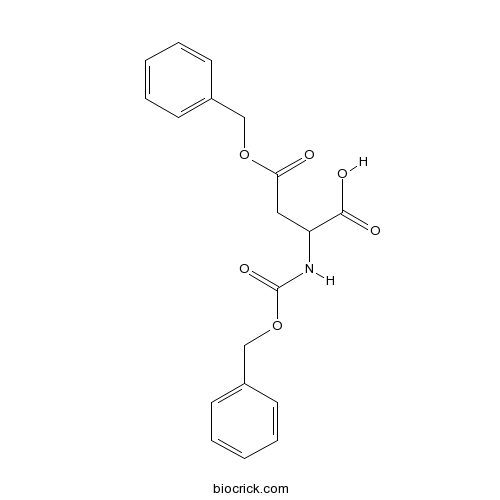
Z-Asp(OBzl)-OHCAS# 3479-47-8 |
- Fulvestrant
Catalog No.:BCC1081
CAS No.:129453-61-8
- Bazedoxifene
Catalog No.:BCC1411
CAS No.:198481-32-2
- Bazedoxifene acetate
Catalog No.:BCC1412
CAS No.:198481-33-3
- (E)-2-Decenoic acid
Catalog No.:BCC1292
CAS No.:334-49-6
- Toremifene
Catalog No.:BCC2010
CAS No.:89778-26-7
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 3479-47-8 | SDF | Download SDF |
| PubChem ID | 273366 | Appearance | Powder |
| Formula | C19H19NO6 | M.Wt | 357.4 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | 4-oxo-4-phenylmethoxy-2-(phenylmethoxycarbonylamino)butanoic acid | ||
| SMILES | C1=CC=C(C=C1)COC(=O)CC(C(=O)O)NC(=O)OCC2=CC=CC=C2 | ||
| Standard InChIKey | VUKCNAATVIWRTF-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C19H19NO6/c21-17(25-12-14-7-3-1-4-8-14)11-16(18(22)23)20-19(24)26-13-15-9-5-2-6-10-15/h1-10,16H,11-13H2,(H,20,24)(H,22,23) | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Z-Asp(OBzl)-OH Dilution Calculator

Z-Asp(OBzl)-OH Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.798 mL | 13.9899 mL | 27.9799 mL | 55.9597 mL | 69.9496 mL |
| 5 mM | 0.5596 mL | 2.798 mL | 5.596 mL | 11.1919 mL | 13.9899 mL |
| 10 mM | 0.2798 mL | 1.399 mL | 2.798 mL | 5.596 mL | 6.995 mL |
| 50 mM | 0.056 mL | 0.2798 mL | 0.5596 mL | 1.1192 mL | 1.399 mL |
| 100 mM | 0.028 mL | 0.1399 mL | 0.2798 mL | 0.5596 mL | 0.6995 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Z-Asp(OBzl)-OH
- Sodium usnate
Catalog No.:BCN8376
CAS No.:34769-44-3
- ITX3
Catalog No.:BCC6066
CAS No.:347323-96-0
- Ferrostatin-1 (Fer-1)
Catalog No.:BCC2323
CAS No.:347174-05-4
- Incensole acetate
Catalog No.:BCN3830
CAS No.:34701-53-6
- 1-Indanamine
Catalog No.:BCN2246
CAS No.:34698-41-4
- TCS 2510
Catalog No.:BCC7853
CAS No.:346673-06-1
- Dehydrodeguelin
Catalog No.:BCN4778
CAS No.:3466-23-7
- Cyperotundone
Catalog No.:BCN8339
CAS No.:3466-15-7
- Amoxicillin Sodium
Catalog No.:BCC4947
CAS No.:34642-77-8
- 1-Methoxycarbonyl-beta-carboline
Catalog No.:BCN5282
CAS No.:3464-66-2
- 2-Benzoyloxy-3-hydroxynortropane
Catalog No.:BCN1872
CAS No.:34622-25-8
- Maltohexaose
Catalog No.:BCN6710
CAS No.:34620-77-4
- H-D-Met-OH
Catalog No.:BCC2997
CAS No.:348-67-4
- H-Tyr(Bzl)-OMe.HCl
Catalog No.:BCC3132
CAS No.:34805-17-9
- Boc-Met(O)-OH
Catalog No.:BCC3425
CAS No.:34805-21-5
- BAY 57-1293
Catalog No.:BCC4050
CAS No.:348086-71-5
- 8-Acetonyldihydroavicine
Catalog No.:BCN3303
CAS No.:348098-59-9
- Marilactone
Catalog No.:BCN7363
CAS No.:34818-17-2
- 4'-Demethyleucomin
Catalog No.:BCN5283
CAS No.:34818-83-2
- DL-Dithiothreitol
Catalog No.:BCC7586
CAS No.:3483-12-3
- Bz-Tyr-Oet
Catalog No.:BCC3122
CAS No.:3483-82-7
- Metiamide
Catalog No.:BCC1742
CAS No.:34839-70-8
- 12-Hydroxyabietic acid
Catalog No.:BCN5284
CAS No.:3484-61-5
- Methyl 3-methoxyacrylate
Catalog No.:BCN2258
CAS No.:34846-90-7
Amino acids and peptides. XVIII. Dipeptide formation during the synthesis of Z-Asp(OBzl)-OH.[Pubmed:3436707]
Int J Pept Protein Res. 1987 Nov;30(5):695-700.
During the benzyloxycarbonylation of H-Asp(OBzl)-OH by the Schotten-Bauman reaction with benzyloxycarbonyl chloride in the presence of NaHCO3 or Na2CO3, besides Z-Asp(OBzl)-OH, Z-Asp(OBzl)-Asp(OBzl)-OH was formed as side product, although the extent of the dipeptide formation differed depending on the base used (10% and 20% respectively). It was found that melting point, rotation value and Rf values upon thin-layer chromatography of Z-Asp(OBzl)-Asp(OBzl)-OH were quite similar to those of Z-Asp(OBzl)-OH.


