2-Picenecarboxylic acidCAS# 118172-80-8 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 118172-80-8 | SDF | Download SDF |
| PubChem ID | 23757062 | Appearance | Powder |
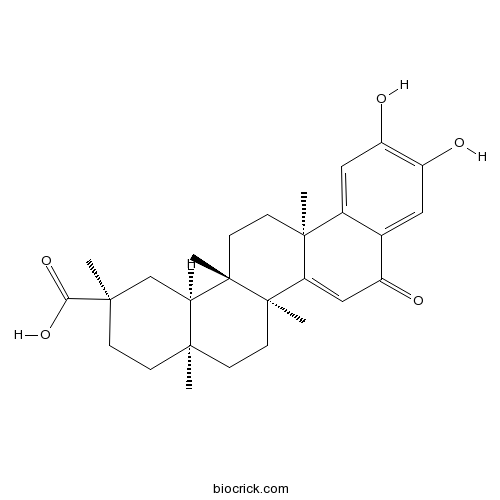
| Formula | C28H36O5 | M.Wt | 452.6 |
| Type of Compound | Triterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
| Chemical Name | (2R,4aS,6aR,6aS,14aS,14bR)-10,11-dihydroxy-2,4a,6a,6a,14a-pentamethyl-8-oxo-1,3,4,5,6,13,14,14b-octahydropicene-2-carboxylic acid | ||
| SMILES | CC12CCC(CC1C3(CCC4(C5=CC(=C(C=C5C(=O)C=C4C3(CC2)C)O)O)C)C)(C)C(=O)O | ||
| Standard InChIKey | MEUCDRGPRSOAHE-BRUCSKOJSA-N | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

2-Picenecarboxylic acid Dilution Calculator

2-Picenecarboxylic acid Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.2095 mL | 11.0473 mL | 22.0946 mL | 44.1891 mL | 55.2364 mL |
| 5 mM | 0.4419 mL | 2.2095 mL | 4.4189 mL | 8.8378 mL | 11.0473 mL |
| 10 mM | 0.2209 mL | 1.1047 mL | 2.2095 mL | 4.4189 mL | 5.5236 mL |
| 50 mM | 0.0442 mL | 0.2209 mL | 0.4419 mL | 0.8838 mL | 1.1047 mL |
| 100 mM | 0.0221 mL | 0.1105 mL | 0.2209 mL | 0.4419 mL | 0.5524 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- 6-Oxo-23-norpristimerol
Catalog No.:BCN8054
CAS No.:118172-79-5
- 6''-O-Acetylastragalin
Catalog No.:BCN6058
CAS No.:118169-27-0
- Volvaltrate B
Catalog No.:BCN6736
CAS No.:1181224-13-4
- [Phe8Ψ(CH-NH)-Arg9]-Bradykinin
Catalog No.:BCC5995
CAS No.:118122-39-7
- Schisanwilsonin I
Catalog No.:BCN5548
CAS No.:1181216-84-1
- Schisanwilsonin H
Catalog No.:BCN3315
CAS No.:1181216-83-0
- Karounidiol
Catalog No.:BCN2704
CAS No.:118117-31-0
- Cyprodime hydrochloride
Catalog No.:BCC7425
CAS No.:118111-54-9
- Prehelminthosporolactone
Catalog No.:BCN7289
CAS No.:118101-72-7
- L-365,260
Catalog No.:BCC7477
CAS No.:118101-09-0
- Zoledronic Acid
Catalog No.:BCC1067
CAS No.:118072-93-8
- Dihydroprehelminthosporol
Catalog No.:BCN7288
CAS No.:118069-95-7
- EMD638683
Catalog No.:BCC1551
CAS No.:1181770-72-8
- Soyasaponin Ab
Catalog No.:BCN2896
CAS No.:118194-13-1
- 1,4-Dicaffeoylquinic acid
Catalog No.:BCN5912
CAS No.:1182-34-9
- Isodorsmanin A
Catalog No.:BCN6460
CAS No.:118266-99-2
- Lafutidine
Catalog No.:BCC4544
CAS No.:118288-08-7
- AF-DX 384
Catalog No.:BCC7024
CAS No.:118290-26-9
- 6-O-Acetylscandoside
Catalog No.:BCN8320
CAS No.:118292-15-2
- Tazarotene
Catalog No.:BCC2540
CAS No.:118292-40-3
- Licoricesaponin A3
Catalog No.:BCN7905
CAS No.:118325-22-7
- Australine
Catalog No.:BCN2053
CAS No.:118396-02-4
- Trimethylamine oxide
Catalog No.:BCN1819
CAS No.:1184-78-7
- AG-18
Catalog No.:BCC1051
CAS No.:118409-57-7
Physiologic Medium Rewires Cellular Metabolism and Reveals Uric Acid as an Endogenous Inhibitor of UMP Synthase.[Pubmed:28388410]
Cell. 2017 Apr 6;169(2):258-272.e17.
A complex interplay of environmental factors impacts the metabolism of human cells, but neither traditional culture media nor mouse plasma mimic the metabolite composition of human plasma. Here, we developed a culture medium with polar metabolite concentrations comparable to those of human plasma (human plasma-like medium [HPLM]). Culture in HPLM, relative to that in traditional media, had widespread effects on cellular metabolism, including on the metabolome, redox state, and glucose utilization. Among the most prominent was an inhibition of de novo pyrimidine synthesis-an effect traced to uric acid, which is 10-fold higher in the blood of humans than of mice and other non-primates. We find that uric acid directly inhibits uridine monophosphate synthase (UMPS) and consequently reduces the sensitivity of cancer cells to the chemotherapeutic agent 5-fluorouracil. Thus, media that better recapitulates the composition of human plasma reveals unforeseen metabolic wiring and regulation, suggesting that HPLM should be of broad utility.
Critical Evaluation of NIR and ATR-IR Spectroscopic Quantifications of Rosmarinic Acid in Rosmarini folium Supported by Quantum Chemical Calculations.[Pubmed:28388786]
Planta Med. 2017 Aug;83(12-13):1076-1084.
The present study evaluates the analytical performance of near infrared as well as attenuated total reflection infrared spectroscopy for the determination of the rosmarinic acid content in Rosmarini folium. Therefore, the recorded near infrared and attenuated total reflection infrared spectra of 42 milled Rosmarini folium samples were correlated with reference data (range: 1.138-2.199 rosmarinic acid %) obtained by HPLC analysis. Partial least squares regression models were established as a quantitative multivariate data analysis tool. Evaluation via full cross-validation and test set validation resulted in comparable performances for both techniques: near infrared [coefficient of determination: 0.90 (test set validation); standard error of cross-validation: 0.060 rosmarinic acid %; standard error of prediction: 0.058 rosmarinic acid %] and attenuated total reflection infrared [coefficient of determination: 0.91 (test set validation); standard error of cross-validation: 0.063 rosmarinic acid %; standard error of prediction: 0.060 rosmarinic acid %]. Furthermore, quantum chemical calculations were applied to obtain a theoretical infrared spectrum of rosmarinic acid. Good agreement to the spectrum of pure rosmarinic acid was achieved in the lower wavenumber region, whereas the higher wavenumber region showed less compliance. The knowledge of the vibrational modes of rosmarinic acid was used for the association with the high values of the regression coefficient plots of the established partial least squares regression models.
Application of UV-irradiated Fe(III)-nitrilotriacetic acid (UV-Fe(III)NTA) and UV-NTA-Fenton systems to degrade model and natural occurring naphthenic acids.[Pubmed:28388447]
Chemosphere. 2017 Jul;179:359-366.
Naphthenic acids (NAs) are a highly complex mixture of organic compounds naturally present in bitumen and identified as the primary toxic constituent of oil sands process-affected water (OSPW). This work investigated the degradation of cyclohexanoic acid (CHA), a model NA compound, and natural occurring NAs during the UV photolysis of Fe(III)-nitrilotriacetic acid (UV-Fe(III)NTA) and UV-NTA-Fenton processes. The results indicated that in the UV-Fe(III)NTA process at pH 8, the CHA removal increased with increasing NTA dose (0.18, 0.36 and 0.72 mM), while it was independent of the Fe(III) dose (0.09, 0.18 and 0.36 mM). Moreover, the three Fe concentrations had no influence on the photolysis of the Fe(III)NTA complex. The main responsible species for the CHA degradation was hydroxyl radical (OH), and the role of dissolved O2 in the OH generation was found to be negligible. Real OSPW was treated with the UV-Fe(III)NTA and UV-NTA-Fenton advanced oxidation processes (AOPs). The removals of classical NAs (O2-NAs), oxidized NAs with one additional oxygen atom (O3-NAs) and with two additional oxygen atoms (O4-NAs) were 44.5%, 21.3%, and 25.2% in the UV-Fe(III)NTA process, respectively, and 98.4%, 86.0%, and 81.0% in the UV-NTA-Fenton process, respectively. There was no influence of O2 on the NA removal in these two processes. The results also confirmed the high reactivity of the O2-NA species with more carbons and increasing number of rings or double bond equivalents. This work opens a new window for the possible treatment of OSPW at natural pH using these AOPs.


