AjugolCAS# 52949-83-4 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 52949-83-4 | SDF | Download SDF |
| PubChem ID | 6325127 | Appearance | White powder |
| Formula | C15H24O9 | M.Wt | 348.4 |
| Type of Compound | Iridoids | Storage | Desiccate at -20°C |
| Solubility | DMSO : ≥ 3.7 mg/mL (10.62 mM) *"≥" means soluble, but saturation unknown. | ||
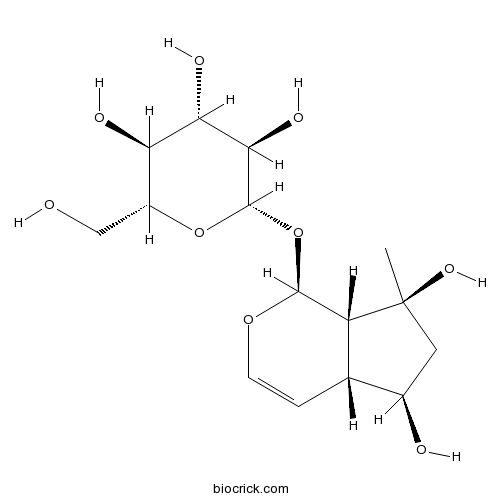
| Chemical Name | (2S,3R,4S,5S,6R)-2-[[(1S,4aR,5R,7S,7aS)-5,7-dihydroxy-7-methyl-4a,5,6,7a-tetrahydro-1H-cyclopenta[c]pyran-1-yl]oxy]-6-(hydroxymethyl)oxane-3,4,5-triol | ||
| SMILES | CC1(CC(C2C1C(OC=C2)OC3C(C(C(C(O3)CO)O)O)O)O)O | ||
| Standard InChIKey | VELYAQRXBJLJAK-XKKWFBPMSA-N | ||
| Standard InChI | InChI=1S/C15H24O9/c1-15(21)4-7(17)6-2-3-22-13(9(6)15)24-14-12(20)11(19)10(18)8(5-16)23-14/h2-3,6-14,16-21H,4-5H2,1H3/t6-,7+,8+,9+,10+,11-,12+,13-,14-,15-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | Ajugol shows some trypanocidal potential against Trypanosoma brucei rhodesiense (IC50 values 29.3–73.0 ug/ml). |
| Targets | Antifection |
| In vitro | Anti-protozoal and plasmodial FabI enzyme inhibiting metabolites of Scrophularia lepidota roots.[Pubmed: 15680992 ]Phytochemistry, 2005, 66(3):355-62.The ethanolic root extract of Scrophularia lepidota, an endemic plant of the Turkish flora, has been investigated for its anti-protozoal and inhibitory effect towards plasmodial enoyl-ACP reductase (FabI), a key enzyme of fatty acid biosynthesis in Plasmodium falciparum. |
| Structure Identification | Volume 23, Issue 11, 1982, Pages 1215-1216Structural revision of ajugol and myoporoside[Reference: WebLink]
|

Ajugol Dilution Calculator

Ajugol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.8703 mL | 14.3513 mL | 28.7026 mL | 57.4053 mL | 71.7566 mL |
| 5 mM | 0.5741 mL | 2.8703 mL | 5.7405 mL | 11.4811 mL | 14.3513 mL |
| 10 mM | 0.287 mL | 1.4351 mL | 2.8703 mL | 5.7405 mL | 7.1757 mL |
| 50 mM | 0.0574 mL | 0.287 mL | 0.5741 mL | 1.1481 mL | 1.4351 mL |
| 100 mM | 0.0287 mL | 0.1435 mL | 0.287 mL | 0.5741 mL | 0.7176 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
Ajugol is an iridiod glucoside.
References:
[1]. Elusiyan CA, et al. Distribution of iridiod glucosides and anti-oxidant compounds in Spathodea campanulata parts. Afr J Tradit Complement Altern Med. 2011;8(1):27-33.
- Genz-644282
Catalog No.:BCC1592
CAS No.:529488-28-6
- N-(2,6-Diphenylmethyl)-1-piperazine acetylamine
Catalog No.:BCC9052
CAS No.:5294-61-1
- Nanaomycin A
Catalog No.:BCC3611
CAS No.:52934-83-5
- Neosophoramine
Catalog No.:BCN5690
CAS No.:52932-74-8
- Dammaradienyl acetate
Catalog No.:BCN5689
CAS No.:52914-31-5
- PRIMA-1MET
Catalog No.:BCC2414
CAS No.:5291-32-7
- Motilin (human, porcine)
Catalog No.:BCC5894
CAS No.:52906-92-0
- 6,7-Dihydroxy-4-Methylcoumarin
Catalog No.:BCC8287
CAS No.:529-84-0
- Euxanthone
Catalog No.:BCN5694
CAS No.:529-61-3
- Genistin
Catalog No.:BCN2396
CAS No.:529-59-9
- Prunin
Catalog No.:BCN5693
CAS No.:529-55-5
- Scutellarein
Catalog No.:BCN5380
CAS No.:529-53-3
- Acetylisocupressic acid
Catalog No.:BCN5695
CAS No.:52992-82-2
- Prednisone
Catalog No.:BCC4957
CAS No.:53-03-2
- Estrone
Catalog No.:BCN2201
CAS No.:53-16-7
- Mitotane (Lsodren)
Catalog No.:BCC3815
CAS No.:53-19-0
- Cocaine hydrochloride
Catalog No.:BCC5943
CAS No.:53-21-4
- Methylprednisolone acetate
Catalog No.:BCC9043
CAS No.:53-36-1
- Oxandrolone
Catalog No.:BCC5242
CAS No.:53-39-4
- Dehydroepiandrosterone
Catalog No.:BCN2202
CAS No.:53-43-0
- Indomethacin
Catalog No.:BCC3794
CAS No.:53-86-1
- L-Picein
Catalog No.:BCC8336
CAS No.:530-14-3
- Deoxyvasicinone
Catalog No.:BCN5697
CAS No.:530-53-0
- 2,6-Dimethoxy-1,4-benzoquinone
Catalog No.:BCN5698
CAS No.:530-55-2
Anti-protozoal and plasmodial FabI enzyme inhibiting metabolites of Scrophularia lepidota roots.[Pubmed:15680992]
Phytochemistry. 2005 Feb;66(3):355-62.
The ethanolic root extract of Scrophularia lepidota, an endemic plant of the Turkish flora, has been investigated for its anti-protozoal and inhibitory effect towards plasmodial enoyl-ACP reductase (FabI), a key enzyme of fatty acid biosynthesis in Plasmodium falciparum. Chromatographic separation of the extract yielded 10 iridoids (1-10), two of which are new, and a known phenylethanoid glycoside (11). The structures of the new compounds were determined as 3,4-dihydro-methylcatalpol (8) and 6-O-[4''-O-trans-(3,4-dimethoxycinnamoyl)-alpha-L-rhamnopyranosyl]aucubin (scrolepidoside, 9) by spectroscopic means. The remaining metabolites were characterized as catalpol (1), 6-O-methylcatalpol (2), aucubin (3), 6-O-alpha-L-rhamnopyranosyl-aucubin (sinuatol, 4), 6-O-beta-D-xylopyranosylaucubin (5), Ajugol (6), ajugoside (7), an iridoid-related aglycone (10) and angoroside C (11). Nine isolates were active against Leishmania donovani, with the new compound 9 being most potent (IC50 6.1 microg/ml). Except for 4, all pure compounds revealed some trypanocidal potential against Trypanosoma brucei rhodesiense (IC50 values 29.3-73.0 microg/ml). Only compound 10 showed moderate anti-plasmodial (IC50 40.6 microg/ml) and FabI enzyme inhibitory activity (IC50 100 microg/ml). 10 is the second natural product inhibiting the fatty acid biosynthesis of Plasmodium falciparum.


