Cinchonain IIbCAS# 85022-68-0 |
- Cinchonain IIa
Catalog No.:BCN7716
CAS No.:85081-23-8
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 85022-68-0 | SDF | Download SDF |
| PubChem ID | 21676385 | Appearance | Powder |
| Formula | C39H32O15 | M.Wt | 740.66 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
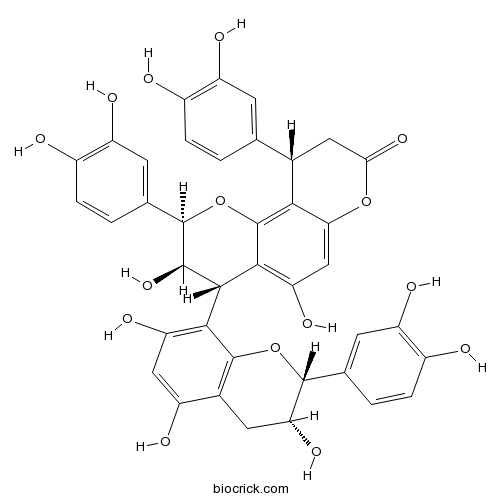
| Chemical Name | (2R,3R,4S,10S)-2,10-bis(3,4-dihydroxyphenyl)-4-[(2R,3R)-2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-3,4-dihydro-2H-chromen-8-yl]-3,5-dihydroxy-3,4,9,10-tetrahydro-2H-pyrano[2,3-h]chromen-8-one | ||
| SMILES | C1C(C(OC2=C1C(=CC(=C2C3C(C(OC4=C5C(CC(=O)OC5=CC(=C34)O)C6=CC(=C(C=C6)O)O)C7=CC(=C(C=C7)O)O)O)O)O)C8=CC(=C(C=C8)O)O)O | ||
| Standard InChIKey | NWZBNZUABGSPSN-FUOMFPIFSA-N | ||
| Standard InChI | InChI=1S/C39H32O15/c40-19-4-1-14(7-23(19)44)17-11-30(50)52-29-13-27(48)33-34(35(51)37(54-39(33)31(17)29)16-3-6-21(42)25(46)9-16)32-26(47)12-22(43)18-10-28(49)36(53-38(18)32)15-2-5-20(41)24(45)8-15/h1-9,12-13,17,28,34-37,40-49,51H,10-11H2/t17-,28+,34-,35+,36+,37+/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Cinchonain IIb shows high radical scavenging activity and reducing power. |

Cinchonain IIb Dilution Calculator

Cinchonain IIb Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.3501 mL | 6.7507 mL | 13.5015 mL | 27.0029 mL | 33.7537 mL |
| 5 mM | 0.27 mL | 1.3501 mL | 2.7003 mL | 5.4006 mL | 6.7507 mL |
| 10 mM | 0.135 mL | 0.6751 mL | 1.3501 mL | 2.7003 mL | 3.3754 mL |
| 50 mM | 0.027 mL | 0.135 mL | 0.27 mL | 0.5401 mL | 0.6751 mL |
| 100 mM | 0.0135 mL | 0.0675 mL | 0.135 mL | 0.27 mL | 0.3375 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Shikonofuran A
Catalog No.:BCN2826
CAS No.:85022-66-8
- ADL5859 HCl
Catalog No.:BCC1265
CAS No.:850173-95-4
- Afatinib dimaleate
Catalog No.:BCC1330
CAS No.:850140-73-7
- Sulfatinib
Catalog No.:BCC8811
CAS No.:1308672-74-3
- 5-Methoxysuberenone
Catalog No.:BCN3638
CAS No.:85011-58-1
- Methyl 1,6-dihydroxy-3-methylxanthone-8-carboxylate
Catalog No.:BCN7473
CAS No.:85003-85-6
- Altrenogest
Catalog No.:BCC4479
CAS No.:850-52-2
- Sudan III
Catalog No.:BCN6962
CAS No.:85-86-9
- Sudan IV;Solvent Red 24
Catalog No.:BCN8379
CAS No.:85-83-6
- 2-Benzoylbenzoic acid
Catalog No.:BCC8561
CAS No.:85-52-9
- Maohuoside B
Catalog No.:BCN8084
CAS No.:849834-04-4
- ARP 101
Catalog No.:BCC2371
CAS No.:849773-63-3
- Rosin
Catalog No.:BCN5969
CAS No.:85026-55-7
- Fmoc-Arg(Tos)-ol
Catalog No.:BCC2588
CAS No.:850330-29-9
- Norviburtinal
Catalog No.:BCN4399
CAS No.:85051-41-8
- Apigenin 5-O-neohesperidoside
Catalog No.:BCN6840
CAS No.:850630-40-9
- Alogliptin (SYR-322)
Catalog No.:BCC2113
CAS No.:850649-61-5
- Alogliptin Benzoate
Catalog No.:BCC1341
CAS No.:850649-62-6
- GSK269962A
Catalog No.:BCC5178
CAS No.:850664-21-0
- BX517(PDK1 inhibitor2)
Catalog No.:BCC6391
CAS No.:850717-64-5
- NBI-98782
Catalog No.:BCC4277
CAS No.:85081-18-1
- Cinchonain IIa
Catalog No.:BCN7716
CAS No.:85081-23-8
- Danoprevir (RG7227)
Catalog No.:BCC2106
CAS No.:850876-88-9
- Amuvatinib (MP-470, HPK 56)
Catalog No.:BCC2258
CAS No.:850879-09-3
Analysis of Antioxidant Polyphenols in Loquat Leaves using HPLC-based Activity Profiling.[Pubmed:30428201]
Nat Prod Commun. 2017 Feb;12(2):163-166.
Leaves of Eriobotryajaponica (loquat) have been used in Traditional Chinese Medicine with beneficial effects in numerous diseases. Extracts from loquat leaves are rich in antioxidants, containing among others: triterpenes, sesquiterpenes, flavonoids, tannins, and megastigmane glycosides. However, there is no conclusive study revealing which of these compounds are the main bioactive principles. The goal of this study was to pinpoint compounds responsible for strong antioxidant activity. Eriobotryaefolium was extracted and fractionated between solvents of increasing polarity. All extracts and fractions were screened for total polyphenols and tannins, and antioxidant activity was checked by DPPH, phosphomolybdenum and linoleic acid tests. The ethyl acetate fraction demonstrated the highest antioxidant activity and contained the largest amount of polyphenols. Applying HPLC-based activity profiling to localize antioxidants revealed that Cinchonain IIb, as well as flavonoid glycosides such as hyperoside, isoquercitrin, kaempferol glycosides, quercetin-rhamnoside, as well as two tentatively identified protocatechuic acid derivatives are the main substances responsible for the strong antioxidant activity of the ethyl acetate fraction.


