GW791343 dihydrochlorideP2X7 allosteric modulator CAS# 1019779-04-4 |
- GW791343 HCl
Catalog No.:BCC4974
CAS No.:309712-55-8
- A-740003
Catalog No.:BCC1322
CAS No.:861393-28-4
- A 839977
Catalog No.:BCC4290
CAS No.:870061-27-1
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1019779-04-4 | SDF | Download SDF |
| PubChem ID | 71576670 | Appearance | Powder |
| Formula | C20H26Cl2F2N4O | M.Wt | 447.35 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | H2O : 100 mg/mL (223.54 mM; Need ultrasonic) DMSO : 20 mg/mL (44.71 mM; Need ultrasonic) | ||
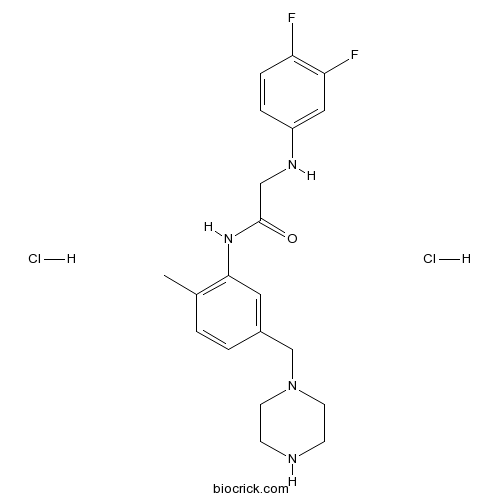
| Chemical Name | 2-(3,4-difluoroanilino)-N-[2-methyl-5-(piperazin-1-ylmethyl)phenyl]acetamide;dihydrochloride | ||
| SMILES | CC1=C(C=C(C=C1)CN2CCNCC2)NC(=O)CNC3=CC(=C(C=C3)F)F.Cl.Cl | ||
| Standard InChIKey | IYJPZTBIYHPSKF-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C20H24F2N4O.2ClH/c1-14-2-3-15(13-26-8-6-23-7-9-26)10-19(14)25-20(27)12-24-16-4-5-17(21)18(22)11-16;;/h2-5,10-11,23-24H,6-9,12-13H2,1H3,(H,25,27);2*1H | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

GW791343 dihydrochloride Dilution Calculator

GW791343 dihydrochloride Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 2.2354 mL | 11.1769 mL | 22.3539 mL | 44.7077 mL | 55.8847 mL |
| 5 mM | 0.4471 mL | 2.2354 mL | 4.4708 mL | 8.9415 mL | 11.1769 mL |
| 10 mM | 0.2235 mL | 1.1177 mL | 2.2354 mL | 4.4708 mL | 5.5885 mL |
| 50 mM | 0.0447 mL | 0.2235 mL | 0.4471 mL | 0.8942 mL | 1.1177 mL |
| 100 mM | 0.0224 mL | 0.1118 mL | 0.2235 mL | 0.4471 mL | 0.5588 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
GW791343 is a P2X7 allosteric modulator. Exhibits species-specific activity and acts as a negative allosteric modulator of human P2X7 (pIC50 = 6.9 - 7.2). A positive allosteric modulator of rat P2X7.
- Zardaverine
Catalog No.:BCC2069
CAS No.:101975-10-4
- Octacosyl (E)-ferulate
Catalog No.:BCN5834
CAS No.:101959-37-9
- S0859
Catalog No.:BCC1914
CAS No.:1019331-10-2
- Calyculin A
Catalog No.:BCC2457
CAS No.:101932-71-2
- Regorafenib monohydrate
Catalog No.:BCC1884
CAS No.:1019206-88-2
- Dabigatran etexilate benzenesulfonate
Catalog No.:BCC8925
CAS No.:1019206-65-5
- sodium 4-pentynoate
Catalog No.:BCC1958
CAS No.:101917-30-0
- LX-4211
Catalog No.:BCC1714
CAS No.:1018899-04-1
- 7-Z-Trifostigmanoside I
Catalog No.:BCN7869
CAS No.:1018898-17-3
- Diclazuril
Catalog No.:BCC8937
CAS No.:101831-37-2
- Butenafine HCl
Catalog No.:BCC4768
CAS No.:101827-46-7
- TG 100801 Hydrochloride
Catalog No.:BCC1997
CAS No.:1018069-81-2
- Acetoacetanilide
Catalog No.:BCC8803
CAS No.:102-01-2
- 3,4-Dihydroxyphenylacetic Acid
Catalog No.:BCC8281
CAS No.:102-32-9
- Phenyethyl 3-methylcaffeate
Catalog No.:BCN8457
CAS No.:71835-85-3
- Sulfaclozine
Catalog No.:BCC9155
CAS No.:102-65-8
- 20,24-Epoxy-24-methoxy-23(24-25)abeo-dammaran-3-one
Catalog No.:BCN1639
CAS No.:1020074-97-8
- SGI-1027
Catalog No.:BCC4588
CAS No.:1020149-73-8
- DCC-2036 (Rebastinib)
Catalog No.:BCC4390
CAS No.:1020172-07-9
- (R)-4-Benzyl-2-oxazolidinone
Catalog No.:BCC8395
CAS No.:102029-44-7
- PF-04457845
Catalog No.:BCC1851
CAS No.:1020315-31-4
- Protosappanin A
Catalog No.:BCN7259
CAS No.:102036-28-2
- Protosappanin B
Catalog No.:BCN2281
CAS No.:102036-29-3
- Tubeimoside I
Catalog No.:BCN1089
CAS No.:102040-03-9
Proposed phase 2/ step 2 in-vitro test on basis of EN 14561 for standardised testing of the wound antiseptics PVP-iodine, chlorhexidine digluconate, polihexanide and octenidine dihydrochloride.[Pubmed:28193164]
BMC Infect Dis. 2017 Feb 13;17(1):143.
BACKGROUND: Currently, there is no agreed standard for exploring the antimicrobial activity of wound antiseptics in a phase 2/ step 2 test protocol. In the present study, a standardised in-vitro test is proposed, which allows to test potential antiseptics in a more realistically simulation of conditions found in wounds as in a suspension test. Furthermore, factors potentially influencing test results such as type of materials used as test carrier or various compositions of organic soil challenge were investigated in detail. METHODS: This proposed phase 2/ step 2 test method was modified on basis of the EN 14561 by drying the microbial test suspension on a metal carrier for 1 h, overlaying the test wound antiseptic, washing-off, neutralization, and dispersion at serial dilutions at the end of the required exposure time yielded reproducible, consistent test results. RESULTS: The difference between the rapid onset of the antiseptic effect of PVP-I and the delayed onset especially of polihexanide was apparent. Among surface-active antimicrobial compounds, octenidine was more effective than chlorhexidine digluconate and polihexanide, with some differences depending on the test organisms. However, octenidine and PVP-I were approximately equivalent in efficiency and microbial spectrum, while polihexanide required longer exposure times or higher concentrations for a comparable antimicrobial efficacy. CONCLUSION: Overall, this method allowed testing and comparing differ liquid and gel based antimicrobial compounds in a standardised setting.
Original research paper. Characterization and taste masking evaluation of microparticles with cetirizine dihydrochloride and methacrylate-based copolymer obtained by spray drying.[Pubmed:28231047]
Acta Pharm. 2017 Mar 1;67(1):113-124.
Taste of a pharmaceutical formulation is an important parameter for the effectiveness of pharmacotherapy. Cetirizine dihydrochloride (CET) is a second-generation antihistamine that is commonly administered in allergy treatment. CET is characterized by extremely bitter taste and it is a great challenge to successfully mask its taste; therefore the goal of this work was to formulate and characterize the microparticles obtained by the spray drying method with CET and poly(butyl methacrylate-co-(2-dimethylaminoethyl) methacrylate-co-methyl methacrylate 1:2:1 copolymer (Eudragit E PO) as a barrier coating. Assessment of taste masking by the electronic tongue has revealed that designed formulations created an effective taste masking barrier. Taste masking effect was also confirmed by the in vivo model and the in vitro release profile of CET. Obtained data have shown that microparticles with a drug/polymer ratio (0.5:1) are promising CET carriers with efficient taste masking potential and might be further used in designing orodispersible dosage forms with CET.
Investigating antimalarial drug interactions of emetine dihydrochloride hydrate using CalcuSyn-based interactivity calculations.[Pubmed:28257497]
PLoS One. 2017 Mar 3;12(3):e0173303.
The widespread introduction of artemisinin-based combination therapy has contributed to recent reductions in malaria mortality. Combination therapies have a range of advantages, including synergism, toxicity reduction, and delaying the onset of resistance acquisition. Unfortunately, antimalarial combination therapy is limited by the depleting repertoire of effective drugs with distinct target pathways. To fast-track antimalarial drug discovery, we have previously employed drug-repositioning to identify the anti-amoebic drug, emetine dihydrochloride hydrate, as a potential candidate for repositioned use against malaria. Despite its 1000-fold increase in in vitro antimalarial potency (ED50 47 nM) compared with its anti-amoebic potency (ED50 26-32 uM), practical use of the compound has been limited by dose-dependent toxicity (emesis and cardiotoxicity). Identification of a synergistic partner drug would present an opportunity for dose-reduction, thus increasing the therapeutic window. The lack of reliable and standardised methodology to enable the in vitro definition of synergistic potential for antimalarials is a major drawback. Here we use isobologram and combination-index data generated by CalcuSyn software analyses (Biosoft v2.1) to define drug interactivity in an objective, automated manner. The method, based on the median effect principle proposed by Chou and Talalay, was initially validated for antimalarial application using the known synergistic combination (atovaquone-proguanil). The combination was used to further understand the relationship between SYBR Green viability and cytocidal versus cytostatic effects of drugs at higher levels of inhibition. We report here the use of the optimised Chou Talalay method to define synergistic antimalarial drug interactivity between emetine dihydrochloride hydrate and atovaquone. The novel findings present a potential route to harness the nanomolar antimalarial efficacy of this affordable natural product.
Pretreatment cognitive and neural differences between sapropterin dihydrochloride responders and non-responders with phenylketonuria.[Pubmed:28271047]
Mol Genet Metab Rep. 2017 Feb 23;12:8-13.
Sapropterin dihydrochloride (BH4) reduces phenylalanine (Phe) levels and improves white matter integrity in a subset of individuals with phenylketonuria (PKU) known as "responders." Although prior research has identified biochemical and genotypic differences between BH4 responders and non-responders, cognitive and neural differences remain largely unexplored. To this end, we compared intelligence and white matter integrity prior to treatment with BH4 in 13 subsequent BH4 responders with PKU, 16 subsequent BH4 non-responders with PKU, and 12 healthy controls. Results indicated poorer intelligence and white matter integrity in non-responders compared to responders prior to treatment. In addition, poorer white matter integrity was associated with greater variability in Phe across the lifetime in non-responders but not in responders. These results underscore the importance of considering PKU as a multi-faceted, multi-dimensional disorder and point to the need for additional research to delineate characteristics that predict response to treatment with BH4.


