Paeonilactone BCAS# 98751-78-1 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 98751-78-1 | SDF | Download SDF |
| PubChem ID | 10375422 | Appearance | Powder |
| Formula | C10H12O4 | M.Wt | 196.2 |
| Type of Compound | Monoterpenoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
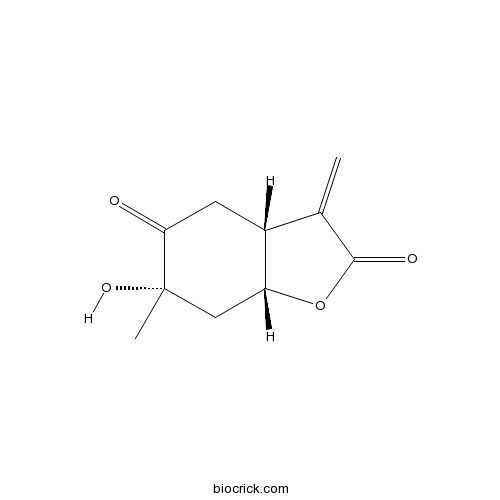
| Chemical Name | (3aR,6S,7aR)-6-hydroxy-6-methyl-3-methylidene-3a,4,7,7a-tetrahydro-1-benzofuran-2,5-dione | ||
| SMILES | CC1(CC2C(CC1=O)C(=C)C(=O)O2)O | ||
| Standard InChIKey | GFSTVWDPRTWASK-XSSZXYGBSA-N | ||
| Standard InChI | InChI=1S/C10H12O4/c1-5-6-3-8(11)10(2,13)4-7(6)14-9(5)12/h6-7,13H,1,3-4H2,2H3/t6-,7-,10+/m1/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Targets | NADPH-oxidase |

Paeonilactone B Dilution Calculator

Paeonilactone B Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 5.0968 mL | 25.4842 mL | 50.9684 mL | 101.9368 mL | 127.421 mL |
| 5 mM | 1.0194 mL | 5.0968 mL | 10.1937 mL | 20.3874 mL | 25.4842 mL |
| 10 mM | 0.5097 mL | 2.5484 mL | 5.0968 mL | 10.1937 mL | 12.7421 mL |
| 50 mM | 0.1019 mL | 0.5097 mL | 1.0194 mL | 2.0387 mL | 2.5484 mL |
| 100 mM | 0.051 mL | 0.2548 mL | 0.5097 mL | 1.0194 mL | 1.2742 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Paeonilactone C
Catalog No.:BCN3964
CAS No.:98751-77-0
- Ropivacaine HCl
Catalog No.:BCC4841
CAS No.:98717-15-8
- ATP disodium salt
Catalog No.:BCC5160
CAS No.:987-65-5
- Dregeoside Ga1
Catalog No.:BCN4548
CAS No.:98665-66-8
- Dregeoside Da1
Catalog No.:BCN4764
CAS No.:98665-65-7
- Ganoderic acid G
Catalog No.:BCN2915
CAS No.:98665-22-6
- Ganolucidic acid A
Catalog No.:BCN2444
CAS No.:98665-21-5
- Ganoderic acid I
Catalog No.:BCN2865
CAS No.:98665-20-4
- Ganoderic acid H
Catalog No.:BCN3038
CAS No.:98665-19-1
- Lucidenic acid D2
Catalog No.:BCN8202
CAS No.:98665-16-8
- Ganoderic Acid J
Catalog No.:BCN8436
CAS No.:100440-26-4
- Ganoderic acid F
Catalog No.:BCN3037
CAS No.:98665-14-6
- Paeonilactone A
Catalog No.:BCN3967
CAS No.:98751-79-2
- Latifoline N-oxide
Catalog No.:BCN1979
CAS No.:98752-06-8
- Isothymusin
Catalog No.:BCN4532
CAS No.:98755-25-0
- Reboxetine mesylate
Catalog No.:BCC4934
CAS No.:98769-84-7
- 3-(4-Hydroxy-3-methoxyphenyl)propyl tetracosanoate
Catalog No.:BCN1292
CAS No.:98770-70-8
- (R)-(+)-Bay K 8644
Catalog No.:BCC7107
CAS No.:98791-67-4
- FLAG tag Peptide
Catalog No.:BCC2562
CAS No.:98849-88-8
- Danshenxinkun D
Catalog No.:BCN2472
CAS No.:98873-76-8
- Pseudolaric acid B-O-beta-D-glucopyranoside
Catalog No.:BCN1291
CAS No.:98891-41-9
- Pseudolaric acid A-O-beta-D-glucopyranoside
Catalog No.:BCN1290
CAS No.:98891-44-2
- 3-Epiursolic acid
Catalog No.:BCN3173
CAS No.:989-30-0
- (-)-Epigallocatechin gallate
Catalog No.:BCN6326
CAS No.:989-51-5
Palbinone, a novel terpenoid from Paeonia albiflora; potent inhibitory activity on 3 alpha-hydroxysteroid dehydrogenase.[Pubmed:8477499]
Chem Pharm Bull (Tokyo). 1993 Mar;41(3):487-90.
Palbinone, a novel terpenoid isolated from the roots of Paeonia albiflora, showed a strong inhibitory activity on the reduced form of nicotinamide adenine dinucleotide phosphate (NADPH)-linked 3 alpha-hydroxysteroid dehydrogenase (3 alpha-HSD) of rat liver cytosol. The structures of palbinone and a known compound, paeonilactone-B isolated from the active fraction of this plant were determined by the use of 2D NMR techniques (1H-1H COSY, 1H-13C COSY, 1H-13C long-range COSY, and HMBC).
Chemical constituents isolated from Paeonia lactiflora roots and their neuroprotective activity against oxidative stress in vitro.[Pubmed:19772486]
J Enzyme Inhib Med Chem. 2009 Oct;24(5):1138-40.
The methanolic extract of Paeonia lactiflora roots significantly protected primary cultures of rat cortical cells exposed to oxidative stress induced by H(2)O(2). Seven monoterpenes, paeonilactone-B (1), paeonilactone-C (2), paeoniflorigenone (3), benzoylpaeoniflorin (4), paeoniflorin (5), oxypaeoniflorin (6) and albiflorin (7), were isolated by bioactivity-guided fractionation and further separation using chromatographic techniques. Among them, compounds 2 and 4 significantly protected primary cultures of rat cortical cells against H(2)O(2)-induced neurotoxicity.


