ProtohypericinCAS# 548-03-8 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 548-03-8 | SDF | Download SDF |
| PubChem ID | 5489488 | Appearance | Powder |
| Formula | C30H18O8 | M.Wt | 506.46 |
| Type of Compound | Quinones | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
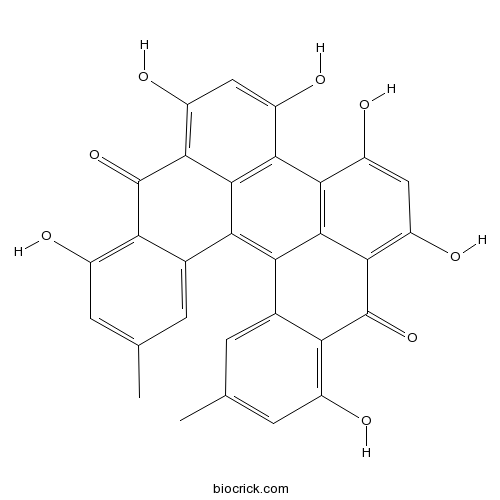
| SMILES | CC1=CC(=C2C(=C1)C3=C4C5=CC(=CC(=C5C(=O)C6=C(C=C(C(=C46)C7=C3C(=C(C=C7O)O)C2=O)O)O)O)C)O | ||
| Standard InChIKey | YLILOANQCQKPOD-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C30H18O8/c1-9-3-11-19(13(31)5-9)29(37)25-17(35)7-15(33)23-24-16(34)8-18(36)26-28(24)22(21(11)27(23)25)12-4-10(2)6-14(32)20(12)30(26)38/h3-8,31-36H,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 1. Protohypericin exhibits photocytotoxicity. |

Protohypericin Dilution Calculator

Protohypericin Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 1.9745 mL | 9.8724 mL | 19.7449 mL | 39.4898 mL | 49.3622 mL |
| 5 mM | 0.3949 mL | 1.9745 mL | 3.949 mL | 7.898 mL | 9.8724 mL |
| 10 mM | 0.1974 mL | 0.9872 mL | 1.9745 mL | 3.949 mL | 4.9362 mL |
| 50 mM | 0.0395 mL | 0.1974 mL | 0.3949 mL | 0.7898 mL | 0.9872 mL |
| 100 mM | 0.0197 mL | 0.0987 mL | 0.1974 mL | 0.3949 mL | 0.4936 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- Huzhangoside B
Catalog No.:BCN8682
CAS No.:94795-70-7
- Cafestol
Catalog No.:BCN8681
CAS No.:469-83-0
- Perisesaccharide C
Catalog No.:BCN8680
CAS No.:1311473-28-5
- Kudinoside D
Catalog No.:BCN8679
CAS No.:173792-61-5
- Flemiphilippinin A
Catalog No.:BCN8678
CAS No.:140366-64-9
- Magnaldehyde B
Catalog No.:BCN8677
CAS No.:92829-72-6
- Lappaol C
Catalog No.:BCN8676
CAS No.:64855-00-1
- 13-Methylberberine
Catalog No.:BCN8675
CAS No.:54260-72-9
- Aurantio-obtusin beta-D-glucoside
Catalog No.:BCN8674
CAS No.:129025-96-3
- Pomiferin
Catalog No.:BCN8673
CAS No.:572-03-2
- Resveratroloside
Catalog No.:BCN8672
CAS No.:38963-95-0
- Epimagnolin B
Catalog No.:BCN8671
CAS No.:1134188-26-3
- Glabrol
Catalog No.:BCN8684
CAS No.:59870-65-4
- Isolindleyin
Catalog No.:BCN8685
CAS No.:87075-18-1
- Isopropyl ferulate
Catalog No.:BCN8686
CAS No.:59831-94-6
- Licochalcone D
Catalog No.:BCN8687
CAS No.:144506-15-0
- Licochalcone E
Catalog No.:BCN8688
CAS No.:864232-34-8
- Conicasterol
Catalog No.:BCN8689
CAS No.:76758-18-4
- Clematichinenoside AR
Catalog No.:BCN8690
CAS No.:761425-93-8
- Beta-Hydroxyisovalerylshikonin
Catalog No.:BCN8691
CAS No.:7415-78-3
- Notoginsenoside FP2
Catalog No.:BCN8692
CAS No.:1004988-75-3
- Isorhamnetin 3-robinobioside
Catalog No.:BCN8693
CAS No.:53584-69-3
- 3-O-Acetyl-11-hydroxy-beta-boswellic acid
Catalog No.:BCN8694
CAS No.:146019-25-2
- Clitorin
Catalog No.:BCN8695
CAS No.:55804-74-5
In vitro transport and uptake of protohypericin and hypericin in the Caco-2 model.[Pubmed:10528085]
Int J Pharm. 1999 Oct 15;188(1):81-6.
The intestinal absorption characteristics of Protohypericin, a protonaphthodianthrone present in Hypericum extract, were studied and compared with those of hypericin. The Caco-2 model was used as a model of the intestinal mucosa to assess transepithelial transport and cell uptake. The experimental work was performed in specific light conditions that prevented both the photoconversion of Protohypericin into hypericin and the photosensitization of the cells. Following application of the individual compounds (80-200 microM) to the apical side of the monolayers, the appearance in the basolateral compartment was found to be very low (<0.5%/5 h), but was comparable for both compounds. A lag-time of 2-3 h was observed, suggesting gradual saturation of binding sites on the membrane or inside the cells. Uptake experiments of Protohypericin and hypericin by Caco-2 cells revealed a very significant cellular accumulation (4-8%); uptake was characterised by saturation after 3 h. The findings of this study suggest that Protohypericin has comparable absorption characteristics as hypericin and may contribute to the beneficial effect of Hypericum extract after oral dosing.
Radiopharmaceutical evaluation of (131)I-protohypericin as a necrosis avid compound.[Pubmed:25655506]
J Drug Target. 2015 Jun;23(5):417-26.
Hypericin is a necrosis avid agent useful for nuclear imaging and tumor therapy. Protohypericin, with a similar structure to hypericin except poorer planarity, is the precursor of hypericin. In this study, we aimed to investigate the impact of this structural difference on self-assembly, and evaluate the necrosis affinity and metabolism in the rat model of reperfused hepatic infarction. Protohypericin appeared less aggregative in solution compared with hypericin by fluorescence analysis. Biodistribution data of (131)I-Protohypericin showed the percentage of injected dose per gram of tissues (%ID/g) increased with time and reached to the maximum of 7.03 at 24 h in necrotic liver by gamma counting. The maximum ratio of target/non-target tissues was 11.7-fold in necrotic liver at 72 h. Pharmacokinetic parameters revealed that the half-life of (131)I-Protohypericin was 14.9 h, enabling a long blood circulation and constant retention in necrotic regions. SPECT-CT, autoradiography, and histological staining showed high uptake of (131)I-Protohypericin in necrotic tissues. These results suggest that (131)I-Protohypericin is a promising necrosis avid compound with a weaker aggregation tendency compared with hypericin and it may have a broad application in imaging and oncotherapy.
Simultaneous determination of protopseudohypericin, pseudohypericin, protohypericin, and hypericin without light exposure.[Pubmed:16526439]
J AOAC Int. 2005 Nov-Dec;88(6):1607-12.
St. John's wort products are commonly standardized to total naphthodianthrones and hyperforin. Determination of these marker compounds is complicated because of the photochemistry of the naphthodianthrones pseudohypericin and hypericin and the instability of hyperforin in solution. Protopseudohypericin and Protohypericin have been identified as naturally occurring naphthodianthrones and, when exposed to light, they are converted into pseudohypericin and hypericin, respectively. However, exposure to light and the resulting naphthodianthrone free-radical reactions oxidize hyperforin. A mathematical relationship between the response of the proto compound and the resulting naphthodianthrone can be established by comparing the analytical response of the proto compound in a solution protected from light with the increase in the analytical response of naphthodianthrone in the same solution after exposure to light. By mathematically converting the proto compounds to their respective products, exposure to light can be avoided while still including proto compounds in a single assay. The method presented here details the reporting of all significant naphthodianthrones, including protopseudohypericin and Protohypericin, without exposure to light. This approach includes the benefits of improved naphthodianthrone precision and protection of hyperforin from oxidation.
Photocytotoxicity of protohypericin after photoconversion to hypericin.[Pubmed:10630113]
Planta Med. 1999 Dec;65(8):719-22.
In the present study, Protohypericin was synthesised in order to compare its intrinsic photocytotoxicity with that of hypericin. The experimental work was performed in specific filtered light conditions that prevented both an unintended photoconversion of Protohypericin and photosensitization of the cells. Assessing the photocytotoxicity as a function of irradiation time, it was found that the photocytotoxicity of both compounds converged after a long irradiation time (i.e., 15 min), while the difference between the photocytotoxicities was maximal after a short irradiation time (i.e., 1 min). Since this could not be accounted for by a redistribution of Protohypericin during irradiation, and the different irradiation times corresponded to different degrees of photoconversion of Protohypericin into hypericin, the results clearly suggest that Protohypericin exhibits intrinsically a dramatically lower photoactivity as compared to hypericin.


