Testosterone acetateCAS# 1045-69-8 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 1045-69-8 | SDF | Download SDF |
| PubChem ID | 92145 | Appearance | Powder |
| Formula | C21H30O3 | M.Wt | 330.5 |
| Type of Compound | N/A | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
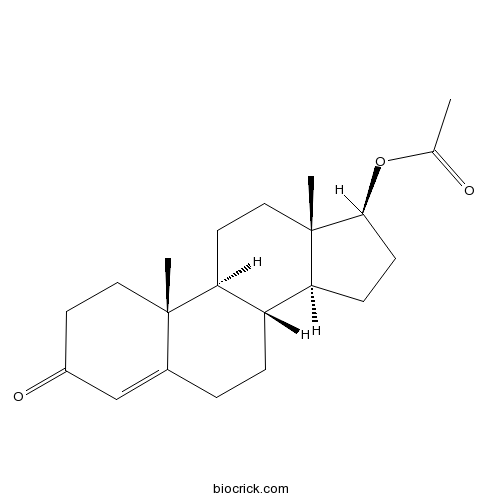
| Chemical Name | [(8R,9S,10R,13S,14S,17S)-10,13-dimethyl-3-oxo-1,2,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-17-yl] acetate | ||
| SMILES | CC(=O)OC1CCC2C1(CCC3C2CCC4=CC(=O)CCC34C)C | ||
| Standard InChIKey | DJPZSBANTAQNFN-PXQJOHHUSA-N | ||
| Standard InChI | InChI=1S/C21H30O3/c1-13(22)24-19-7-6-17-16-5-4-14-12-15(23)8-10-20(14,2)18(16)9-11-21(17,19)3/h12,16-19H,4-11H2,1-3H3/t16-,17-,18-,19-,20-,21-/m0/s1 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||

Testosterone acetate Dilution Calculator

Testosterone acetate Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.0257 mL | 15.1286 mL | 30.2572 mL | 60.5144 mL | 75.643 mL |
| 5 mM | 0.6051 mL | 3.0257 mL | 6.0514 mL | 12.1029 mL | 15.1286 mL |
| 10 mM | 0.3026 mL | 1.5129 mL | 3.0257 mL | 6.0514 mL | 7.5643 mL |
| 50 mM | 0.0605 mL | 0.3026 mL | 0.6051 mL | 1.2103 mL | 1.5129 mL |
| 100 mM | 0.0303 mL | 0.1513 mL | 0.3026 mL | 0.6051 mL | 0.7564 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- RVX-208
Catalog No.:BCC4475
CAS No.:1044870-39-4
- 3'-Methylflavokawin
Catalog No.:BCN3990
CAS No.:1044743-35-2
- Typhaneoside
Catalog No.:BCN4994
CAS No.:104472-68-6
- L803-mts
Catalog No.:BCC5889
CAS No.:1043881-55-5
- RU-SKI 43
Catalog No.:BCC5441
CAS No.:1043797-53-0
- Tetrahydroxysqualene
Catalog No.:BCN5858
CAS No.:1043629-23-7
- Bisoprolol fumarate
Catalog No.:BCC4344
CAS No.:104344-23-2
- IRAK inhibitor 6
Catalog No.:BCC1658
CAS No.:1042672-97-8
- Famciclovir
Catalog No.:BCC4780
CAS No.:104227-87-4
- IRAK inhibitor 1
Catalog No.:BCC1654
CAS No.:1042224-63-4
- Yunnandaphninine G
Catalog No.:BCN5857
CAS No.:1042143-83-8
- Estriol 3,17-dihexanoate
Catalog No.:BCN2238
CAS No.:104202-96-2
- Bayogenin 3-O-beta-D-glucopyranoside
Catalog No.:BCN7868
CAS No.:104513-86-2
- EC 23
Catalog No.:BCC6097
CAS No.:104561-41-3
- CAY10603
Catalog No.:BCC5542
CAS No.:1045792-66-2
- p-Menthan-3-one
Catalog No.:BCN3850
CAS No.:10458-14-7
- Caffeic acid phenethyl ester
Catalog No.:BCN2695
CAS No.:104594-70-9
- CGS 15943
Catalog No.:BCC7157
CAS No.:104615-18-1
- Pramipexole dihydrochloride
Catalog No.:BCN2181
CAS No.:104632-25-9
- Pramipexole
Catalog No.:BCC4467
CAS No.:104632-26-0
- Dexpramipexole dihydrochloride
Catalog No.:BCC1528
CAS No.:104632-27-1
- Dexpramipexole
Catalog No.:BCC1527
CAS No.:104632-28-2
- H-Ile-NH2.HCl
Catalog No.:BCC2962
CAS No.:10466-56-5
- 7,3'-Di-O-methylorobol
Catalog No.:BCN6831
CAS No.:104668-88-4
Bioassay based screening of steroid derivatives in animal feed and supplements.[Pubmed:21742131]
Anal Chim Acta. 2011 Aug 26;700(1-2):183-8.
Receptor binding transcription activation bioassays are valuable tools for the screening of steroid hormones in animal feed and supplements. However, steroid derivatives often lack affinity for their cognate receptor and do not show any direct hormonal activity by themselves. These compounds are thus not detected by these kinds of bioassays and need a bioactivation step in order to become active, both in vivo and in vitro. In this study a comparison was made between different in vitro activation methods for hormone esters and hormone glycosides. Testosterone acetate and testosterone decanoate were chosen as model compounds for the hormone esters, representing the broad range of steroid esters of varying polarities, while genistin was used as a substitute model for the steroid-glycosides. Concerning bioactivation of the steroids esters, the efficiency for alkaline hydrolysis was 90-100% and much better as compared to enzymatic deconjugation by esterase. As a result 1 mug testosterone ester per gram of animal feed could easily be detected by a yeast androgen bioassay. When comparing different enzyme fractions for deglycosilation, genistin was shown to be deconjugated most efficiently by beta-glucuronidase/aryl sulfatase from Helix pomatia, resulting in a significant increase of estrogenic activity as determined by a yeast estrogen bioassay. In conclusion, chemical and enzymatic deconjugation procedures for ester and glycoside conjugates respectively, resulted in a significant increase in hormonal activity as shown by the bioassay readouts and allowed effective screening of these derivatives in animal feed and feed supplements.
Hormone anchored metal complexes. 1. Synthesis, structure, spectroscopy and in vitro antitumor activity of testosterone acetate thiosemicarbazone and its metal complexes.[Pubmed:18472909]
Met Based Drugs. 1999;6(3):177-82.
Testosterone acetate thiosemicarbazone (TATSC, 17-beta-hydroxyandrost-4-one acetate thiosemicarbazone) was synthesized and characterized by single crystal X-ray structure determination. The copper and platinum complexes of this steroid derivative were synthesized and characterized by spectroscopy and electrochemiatry. The in vitro activity of these compounds against human breast cancer cell line MCF-7 was tested. The highest activity was found for the [Pt(TATSC)Cl(1)] followed by [Cu(TATSC)Cl(2)] and the ligand in compariosn with cisplatin.
Induction of androgen receptor activity by norgestimate and norelgestromin in MDA-MB 231 breast cancer cells.[Pubmed:15625768]
Gynecol Endocrinol. 2004 Jul;19(1):18-21.
The function and clinical significance of the androgen receptor (AR) in human breast cancer are still not clear. The synthetic progestins, norgestimate and norelgestromin, were designed to minimize the adverse effects such as acne, hirsuitism and metabolic changes observed with older oral contraceptives while maintaining contraceptive effectiveness and cycle control. AR-mediated effects of these synthetic progestins were studied in an in vitro transactivation assay, employing DNA co-transfection of an AR expression vector and luciferase reporter gene construct in the MDA-MB 231 human breast cancer cell line. Testosterone acetate and 5alpha-dihydrotestosterone induced the reporter gene transcription, whereas incubation of the transfected cells with the natural progestin 17alpha-hydroxyprogesterone did not markedly induce luciferase activity. The progestins norgestimate and norelgestromin exerted a very low androgenic activity. Our data suggest that norgestimate and its metabolite norelgestromin possess weak androgen-like properties. The use of these compounds for clinical application may be of great advantage in the treatment of breast cancer as well as hyperandrogenism in women.


