7,3'-Di-O-methylorobolCAS# 104668-88-4 |
Quality Control & MSDS
3D structure
Package In Stock
Number of papers citing our products

| Cas No. | 104668-88-4 | SDF | Download SDF |
| PubChem ID | 13845970 | Appearance | Powder |
| Formula | C17H14O6 | M.Wt | 314.30 |
| Type of Compound | Flavonoids | Storage | Desiccate at -20°C |
| Solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | ||
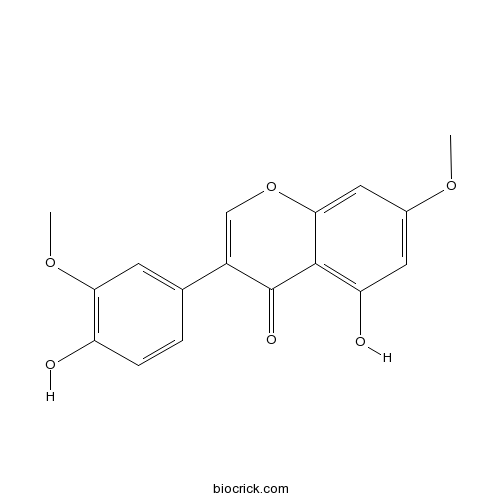
| Chemical Name | 5-hydroxy-3-(4-hydroxy-3-methoxyphenyl)-7-methoxychromen-4-one | ||
| SMILES | COC1=CC(=C2C(=C1)OC=C(C2=O)C3=CC(=C(C=C3)O)OC)O | ||
| Standard InChIKey | NMQZMHHAWZDJOJ-UHFFFAOYSA-N | ||
| Standard InChI | InChI=1S/C17H14O6/c1-21-10-6-13(19)16-15(7-10)23-8-11(17(16)20)9-3-4-12(18)14(5-9)22-2/h3-8,18-19H,1-2H3 | ||
| General tips | For obtaining a higher solubility , please warm the tube at 37 ℃ and shake it in the ultrasonic bath for a while.Stock solution can be stored below -20℃ for several months. We recommend that you prepare and use the solution on the same day. However, if the test schedule requires, the stock solutions can be prepared in advance, and the stock solution must be sealed and stored below -20℃. In general, the stock solution can be kept for several months. Before use, we recommend that you leave the vial at room temperature for at least an hour before opening it. |
||
| About Packaging | 1. The packaging of the product may be reversed during transportation, cause the high purity compounds to adhere to the neck or cap of the vial.Take the vail out of its packaging and shake gently until the compounds fall to the bottom of the vial. 2. For liquid products, please centrifuge at 500xg to gather the liquid to the bottom of the vial. 3. Try to avoid loss or contamination during the experiment. |
||
| Shipping Condition | Packaging according to customer requirements(5mg, 10mg, 20mg and more). Ship via FedEx, DHL, UPS, EMS or other couriers with RT, or blue ice upon request. | ||
| Description | 7,3'-Di-O-methylorobol is a natural product from Sophora japonica L. |
| In vitro | Flavonoids and other compounds from Ouratea ferruginea (Ochnaceae) as anticancer and chemopreventive agents.[Pubmed: 22759912 ]Molecules. 2012 Jul 3;17(7):7989-8000.
|
| Structure Identification | J Asian Nat Prod Res. 2002 Mar;4(1):1-5.A new coumaronochromone from Sophora japonica.[Pubmed: 11991186 ]
|

7,3'-Di-O-methylorobol Dilution Calculator

7,3'-Di-O-methylorobol Molarity Calculator
| 1 mg | 5 mg | 10 mg | 20 mg | 25 mg | |
| 1 mM | 3.1817 mL | 15.9084 mL | 31.8167 mL | 63.6335 mL | 79.5418 mL |
| 5 mM | 0.6363 mL | 3.1817 mL | 6.3633 mL | 12.7267 mL | 15.9084 mL |
| 10 mM | 0.3182 mL | 1.5908 mL | 3.1817 mL | 6.3633 mL | 7.9542 mL |
| 50 mM | 0.0636 mL | 0.3182 mL | 0.6363 mL | 1.2727 mL | 1.5908 mL |
| 100 mM | 0.0318 mL | 0.1591 mL | 0.3182 mL | 0.6363 mL | 0.7954 mL |
| * Note: If you are in the process of experiment, it's necessary to make the dilution ratios of the samples. The dilution data above is only for reference. Normally, it's can get a better solubility within lower of Concentrations. | |||||

Calcutta University

University of Minnesota

University of Maryland School of Medicine

University of Illinois at Chicago

The Ohio State University

University of Zurich

Harvard University

Colorado State University

Auburn University

Yale University

Worcester Polytechnic Institute

Washington State University

Stanford University

University of Leipzig

Universidade da Beira Interior

The Institute of Cancer Research

Heidelberg University

University of Amsterdam

University of Auckland

TsingHua University

The University of Michigan

Miami University

DRURY University

Jilin University

Fudan University

Wuhan University

Sun Yat-sen University

Universite de Paris

Deemed University

Auckland University

The University of Tokyo

Korea University
- H-Ile-NH2.HCl
Catalog No.:BCC2962
CAS No.:10466-56-5
- Dexpramipexole
Catalog No.:BCC1527
CAS No.:104632-28-2
- Dexpramipexole dihydrochloride
Catalog No.:BCC1528
CAS No.:104632-27-1
- Pramipexole
Catalog No.:BCC4467
CAS No.:104632-26-0
- Pramipexole dihydrochloride
Catalog No.:BCN2181
CAS No.:104632-25-9
- CGS 15943
Catalog No.:BCC7157
CAS No.:104615-18-1
- Caffeic acid phenethyl ester
Catalog No.:BCN2695
CAS No.:104594-70-9
- p-Menthan-3-one
Catalog No.:BCN3850
CAS No.:10458-14-7
- CAY10603
Catalog No.:BCC5542
CAS No.:1045792-66-2
- EC 23
Catalog No.:BCC6097
CAS No.:104561-41-3
- Bayogenin 3-O-beta-D-glucopyranoside
Catalog No.:BCN7868
CAS No.:104513-86-2
- Testosterone acetate
Catalog No.:BCC9165
CAS No.:1045-69-8
- Iriflophenone 3-C-beta-D-glucopyranoside
Catalog No.:BCN1635
CAS No.:104669-02-5
- Lupiwighteone
Catalog No.:BCN4045
CAS No.:104691-86-3
- Ganoderic acid K
Catalog No.:BCN3039
CAS No.:104700-95-0
- Ganoderol B
Catalog No.:BCN5859
CAS No.:104700-96-1
- Ganoderol A
Catalog No.:BCN5860
CAS No.:104700-97-2
- Ganoderal A
Catalog No.:BCN2451
CAS No.:104700-98-3
- Boc-D-Glu(OtBu)-OH
Catalog No.:BCC3395
CAS No.:104719-63-3
- 8-Phenyloctanol
Catalog No.:BCC8791
CAS No.:10472-97-6
- 6-O-α-Maltosyl-β-cyclodextrin
Catalog No.:BCC8075
CAS No.:104723-60-6
- Mayteine
Catalog No.:BCN3098
CAS No.:104736-05-2
- 4E-Deacetylchromolaenide 4'-O-acetate
Catalog No.:BCN7263
CAS No.:104736-09-6
- Ganoderic acid S
Catalog No.:BCN5861
CAS No.:104759-35-5
Flavonoids and other compounds from Ouratea ferruginea (Ochnaceae) as anticancer and chemopreventive agents.[Pubmed:22759912]
Molecules. 2012 Jul 3;17(7):7989-8000.
The chemical study of the extracts from leaves and stems of Ouratea ferruginea allowed the identification of a new isoflavone, 5-hydroxy-7,3'4'5'-tetramethoxyisoflavone, and twenty two known compounds, including friedelin, 3beta-friedelinol, lupeone, a mixture of sitosterol, stigmasterol and campesterol, sitosteryl- and stigmasteryl-3-O-b-D-glucopyranosides, 5,4'-dihydroxy-7,5',3'-trimethoxyisoflavone, 5,4'-dihydroxy-7,3'-di-methoxyisoflavone (7,3'-di-O-methylorobol), 5,7,4'-trihydroxy-3',5'-dimethoxyisoflavone (piscigenin), 2R,3R-epicatechin, syringic acid, 2,6-dimethoxybenzoquinone, 2,6-dimethoxyhydroquinone, syringic and ferulic aldehyde, a mixture of vanillic acid, 1-hydroxy-2-methoxy-4-(1E-3-hydroxy-1-propenyl)-benzene and 3,5-dimethoxy-4-hydroxy-dihydrocinamaldehyde, besides amenthoflavone and 7-O-methylamenthoflavone (sequoiaflavone) which are considered as chemotaxonomic markers of Ouratea. The structures were identified by IR, (1)H- and (13)C-NMR and GC-MS, HPLC-MS, besides comparison with literature data. The inhibitory effects of 5,4'-dihydroxy-7,5',3'-trimethoxyisoflavone, 7,3'-di-O-methylorobol, piscigenin and 7-O-methylamenthoflavone on cytochrome P450-dependent 7-ethoxycoumarin O-deethylase (ECOD) and glutathione S-transferase (GST) were evaluated in vitro. The 5,4'-dihydroxy-7,5',3'-trimethoxy-isoflavone was the best inhibitor, inhibiting almost 75% of GST activity. Sequoiaflavone was the most potent inhibitor, inhibiting ECOD assay in 75%. These activities allow us to consider both these flavonoids as potential anticancer and chemopreventive agents.
A new coumaronochromone from Sophora japonica.[Pubmed:11991186]
J Asian Nat Prod Res. 2002 Mar;4(1):1-5.
A new coumaronochromone derivative, sophorophenolone (1), along with 13 known compounds, l-maackiain (2), medicagol (3), 7-O-methylpseudobaptigenin (4), pseudobaptigenin (5), 7,3'-di-O-methylorobol (6), genistein (7), prunetin (8), daidzein (9), formononetin (10), Di-O-methyldaidzein (11), quercetin (12), kaempferol (13) and isorhamnetin (14) were isolated from pericarps of Sophorajaponica L. The structure of compound 1 was established by UV, IR, MS, and one-dimensional and two-dimensional NMR spectroscopy, including DEPT, NOESY, 1H-1H COSY, HMQC, and HMBC experiments.


